Lead (Pb)
post-transition-metalSolid
Standard Atomic Weight
207.2 u [206.14, 207.94]Electron configuration
[Xe] 6s2 4f14 5d10 6p2Melting point
327.46 °C (600.61 K)Boiling point
1748.85 °C (2022 K)Density
1.134200e+4 kg/m³Oxidation states
−4, −2, −1, 0, +1, +2, +3, +4Electronegativity (Pauling)
2.33Ionization energy (1st)
Discovery year
2021Atomic radius
180 pmDetails
Lead is a dense, soft post-transition metal with atomic number 82. It is chemically characterized by the +2 oxidation state, with +4 less stable except in selected compounds, a pattern influenced by the inert-pair effect. Lead has been used since antiquity because it is easily smelted and worked, but its toxicity now strongly limits dispersive uses. It remains important where high density, corrosion resistance, low melting point, and radiation attenuation are valuable.
Lead is a bluish-white metal of bright luster. It is very soft, highly malleable, ductile, and a poor conductor of electricity. It is very resistant to corrosion; lead pipes bearing the insignia of Roman emperors, used as drains from the baths, are still in service. It is used in containers for corrosive liquids (such as sulfuric acid) and may be toughened by the addition of a small percentage of antimony or other metals.
The name derives from the Anglo-Saxon lead, which is of unknown origin. The element was known from prehistoric times. The chemical symbol Pb is derived from the Latin plumbum.
<!-- --> <p class="caption">For more information about the natural variations of the atomic weight of lead please read IUPAC Technical Report Variation of lead isotopic composition and atomic weight in terrestrial materials (IUPAC Technical Report) <img src="images/pdf.gif" style="width:auto; margin:0; vertical-align:bottom;"> by Z.-K. Zhu et al Pure Appl. Chem. <strong>93</strong>, 155-166 (2021).
Lead has been known since ancient times. It is sometimes found free in nature, but is usually obtained from the ores galena (PbS), anglesite (PbSO4), cerussite (PbCO3) and minum (Pb3O4). Although lead makes up only about 0.0013% of the earth's crust, it is not considered to be a rare element since it is easily mined and refined. Most lead is obtained by roasting galena in hot air, although nearly one third of the lead used in the United States is obtained through recycling efforts.
Long known, mentioned in Exodus. The alchemists believed lead to be the oldest metal and associated with the planet Saturn. Native lead occurs in nature, but is rare.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
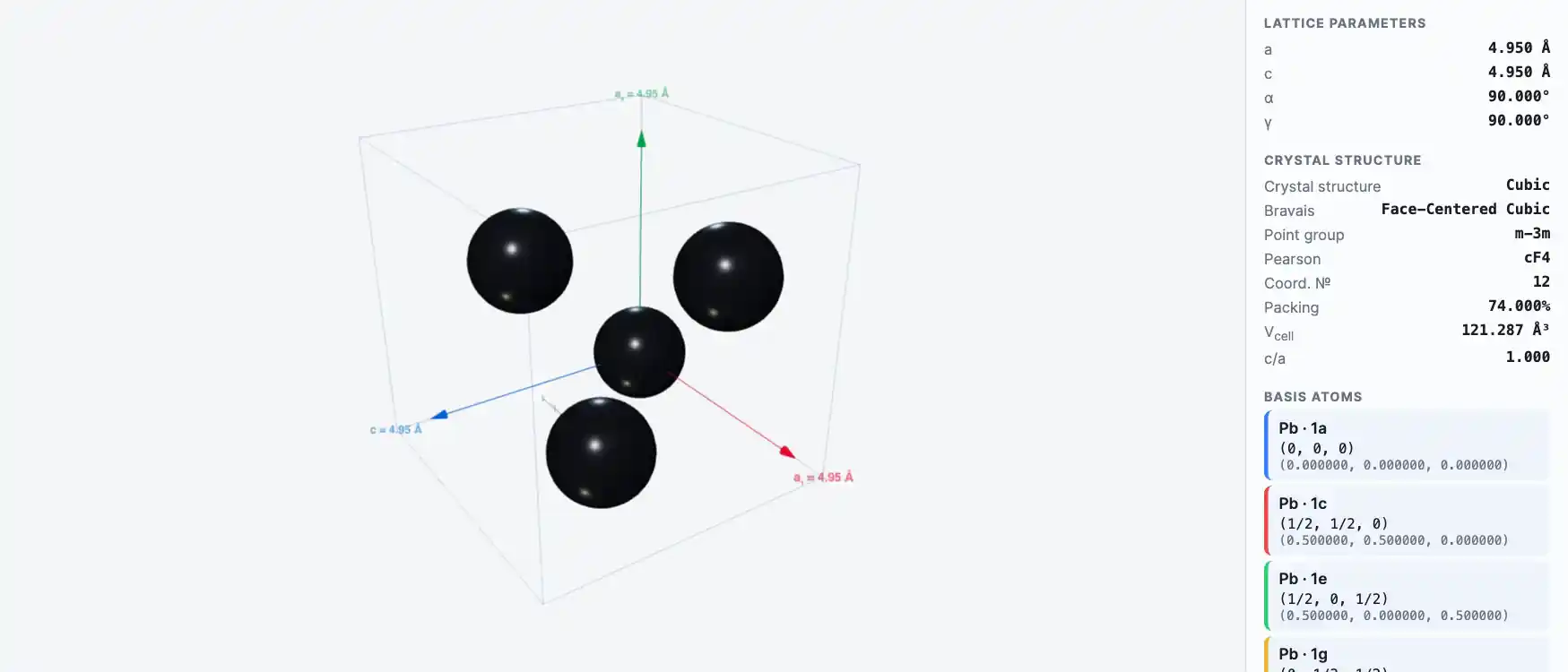
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Pb: 4f¹⁴ 5d¹⁰ 6s² 6p²[Xe] 4f¹⁴ 5d¹⁰ 6s² 6p²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d¹⁰ 6s² 6p²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
No stable isotopes.
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 183 Radioactive | 182.991872 ± 0.00003 | N/A | 535 ms |
| 184 Radioactive | 183.988136 ± 0.000014 | N/A | 490 ms |
| 178 Radioactive | 178.003831 ± 0.000026 | N/A | 250 us |
| 215 Radioactive | 215.00474 ± 0.00011 | N/A | 142 seconds |
| 204 Radioactive | 203.973044 ± 0.0000013 | 1.4000% | 140 Py |
Phase / State
Reason: 302.5 °C below melting point (327.46 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 82 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Pb I | 0 | 135 | 28 | 135 |
| Pb II | +1 | 97 | 3 | 12 |
| Pb III | +2 | 41 | 0 | 0 |
| Pb IV | +3 | 92 | 0 | 0 |
| Pb V | +4 | 90 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Pb I | 0 | 136 |
| Pb II | +1 | 95 |
| Pb III | +2 | 124 |
| Pb IV | +3 | 108 |
| Pb V | +4 | 45 |
| Pb VI | +5 | 2 |
| Pb VII | +6 | 2 |
| Pb VIII | +7 | 2 |
| Pb IX | +8 | 2 |
| Pb X | +9 | 2 |
Ionic Radii
Showing 10 of 12 Ionic Radii.
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 4 | N/A | 98 pm |
| +2 | 6 | N/A | 119 pm |
| +2 | 7 | N/A | 123 pm |
| +2 | 8 | N/A | 129 pm |
| +2 | 9 | N/A | 135 pm |
| +2 | 10 | N/A | 140 pm |
| +2 | 11 | N/A | 145 pm |
| +2 | 12 | N/A | 149 pm |
| +4 | 4 | N/A | 65 pm |
| +4 | 5 | N/A | 73 pm |
Compounds
Isotopes (5)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 183 Radioactive | 182.991872 ± 0.00003 | N/A | 535 ms | α ≈100%β+ ? | |
| 184 Radioactive | 183.988136 ± 0.000014 | N/A | 490 ms | α =80±1.1%β+ ? | |
| 178 Radioactive | 178.003831 ± 0.000026 | N/A | 250 us | α ≈100%β+ ? | |
| 215 Radioactive | 215.00474 ± 0.00011 | N/A | 142 seconds | β- =100% | |
| 204 Radioactive | 203.973044 ± 0.0000013 | 1.4000% ± 0.1000% | 140 Py | IS =1.4±0.6%α ? |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 401.96322 nm | 15000 | Pb I | emission | 6s2.6p2 (3/2,3/2) → 6s2.6p.(2P*<1/2>).6d 2[5/2]* | Measured | NIST | |
| 405.780659 nm | 95000 | Pb I | emission | 6s2.6p2 (3/2,1/2) → 6s2.6p.7s (1/2,1/2)* | Measured | NIST | |
| 406.213593 nm | 14000 | Pb I | emission | 6s2.6p2 (3/2,3/2) → 6s2.6p.(2P*<1/2>).6d 2[3/2]* | Measured | NIST | |
| 415.78144 nm | 10 | Pb I | emission | 6s2.6p2 (3/2,3/2) → 6s2.6p.9s (1/2,1/2)* | Measured | NIST | |
| 416.80327 nm | 10000 | Pb I | emission | 6s2.6p2 (3/2,3/2) → 6s2.6p.(2P*<1/2>).6d 2[5/2]* | Measured | NIST | |
| 434.041263 nm | 200 | Pb I | emission | 6s2.6p2 (3/2,3/2) → 6s2.6p.(2P*<1/2>).7d 2[3/2]* | Measured | NIST | |
| 500.54165 nm | 1000 | Pb I | emission | 6s2.6p2 (3/2,3/2) → 6s2.6p.7s (3/2,1/2)* | Measured | NIST | |
| 500.65724 nm | 100 | Pb I | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.9p (1/2,3/2) | Measured | NIST | |
| 507.6322 nm | 10 | Pb I | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.9p (1/2,1/2) | Measured | NIST | |
| 508.94835 nm | 50 | Pb I | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.9p (1/2,3/2) | Measured | NIST | |
| 509.00083 nm | 20 | Pb I | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.9p (1/2,3/2) | Measured | NIST | |
| 510.72427 nm | 10 | Pb I | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.9p (1/2,1/2) | Measured | NIST | |
| 520.14372 nm | 2000 | Pb I | emission | 6s2.6p2 (3/2,3/2) → 6s2.6p.8s (1/2,1/2)* | Measured | NIST | |
| 569.23465 nm | 40 | Pb I | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.(2P*<1/2>).5f 2[5/2] | Measured | NIST | |
| 589.56245 nm | 200 | Pb I | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.8p (1/2,3/2) | Measured | NIST | |
| 600.18624 nm | 2000 | Pb I | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.8p (1/2,3/2) | Measured | NIST | |
| 601.16667 nm | 500 | Pb I | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.8p (1/2,3/2) | Measured | NIST | |
| 605.93556 nm | 500 | Pb I | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.8p (1/2,1/2) | Measured | NIST | |
| 611.05203 nm | 50 | Pb I | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.8p (1/2,1/2) | Measured | NIST | |
| 623.52656 nm | 100 | Pb I | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.8p (1/2,1/2) | Measured | NIST | |
| 689.2117 nm | 10 | Pb I | emission | 6s2.6p.7p (1/2,1/2) → 6s2.6p.(2P*<1/2>).10d 2[5/2]* | Measured | NIST | |
| 712.893 nm | 5 | Pb I | emission | 6s2.6p.7p (1/2,1/2) → 6s2.6p.11s (1/2,1/2)* | Measured | NIST | |
| 722.89658 nm | 20000 | Pb I | emission | 6s2.6p2 (3/2,3/2) → 6s2.6p.7s (1/2,1/2)* | Measured | NIST | |
| 730.46753 nm | 5 | Pb I | emission | 6s2.6p.7p (1/2,1/2) → 6s2.6p.(2P*<1/2>).9d 2[3/2]* | Measured | NIST | |
| 733.0146 nm | 8 | Pb I | emission | 6s2.6p2 (3/2,1/2) → 6s2.6p2 (3/2,3/2) | Measured | NIST | |
| 734.6676 nm | 10 | Pb I | emission | 6s2.6p.7p (1/2,1/2) → 6s2.6p.(2P*<1/2>).9d 2[5/2]* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 600.61 K |
| Boiling point | 2022.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (15)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.5805 |
| 2 | p | 4.5234 |
| 2 | s | 21.57 |
| 3 | d | 13.4533 |
| 3 | p | 22.8505 |
| 3 | s | 23.8477 |
| 4 | d | 37.6804 |
| 4 | f | 38.0312 |
| 4 | p | 35.9664 |
| 4 | s | 35.1072 |
Crystal Radii Detail (12)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | IVPY | 112 | calculated, | |
| 2 | VI | 133 | ||
| 2 | VII | 137 | calculated, | |
| 2 | VIII | 143 | calculated, | |
| 2 | IX | 149 | calculated, | |
| 2 | X | 154 | calculated, | |
| 2 | XI | 159 | calculated, | |
| 2 | XII | 163 | ||
| 4 | IV | 79 | estimated, | |
| 4 | V | 87 | estimated, |
Isotope Decay Modes (59)
| Isotope | Mode | Intensity |
|---|---|---|
| 178 | A | 100% |
| 178 | B+ | — |
| 179 | A | 100% |
| 180 | A | 100% |
| 181 | A | 100% |
| 181 | B+ | — |
| 182 | A | 100% |
| 182 | B+ | — |
| 183 | A | 100% |
| 183 | B+ | — |
X‑ray Scattering Factors (516)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 4.6699 |
| 10.1617 | — | 4.72735 |
| 10.3261 | — | 4.78551 |
| 10.4931 | — | 4.84439 |
| 10.6628 | — | 4.83957 |
| 10.8353 | — | 4.83203 |
| 11.0106 | — | 4.82451 |
| 11.1886 | — | 4.817 |
| 11.3696 | — | 4.7889 |
| 11.5535 | — | 4.7596 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.4×101 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
3×10-5 milligrams per liter
References (1)
Sources
Sources of this element.
Lead is obtained chiefly from galena (PbS) by a roasting process. Anglesite, cerussite, and minim are other common lead minerals.
References (1)
- [6] Lead https://periodic.lanl.gov/82.shtml
Isotopes in Forensic Science and Anthropology
Information on the use of this element's isotopes in forensic science and anthropology.
Different geographic regions may have characteristic terrestrial lead isotopic compositions because of variations in the ages and chemical composition of the rocks and minerals in the local environment. Therefore, lead produced at a particular location can have a unique lead isotopic composition and it is possible to trace the history and origins of pollutants by measuring the relative amounts of the four stable isotopes of lead (208Pb, 207Pb, 206Pb, and 204Pb) (Fig. IUPAC.82.2) [547] [547] I. Renberg, M. L. Brännvall, R. Bindler, O. Emteryd. Ambio29, 150 (2000).[547] I. Renberg, M. L. Brännvall, R. Bindler, O. Emteryd. Ambio29, 150 (2000)., [548] [548] T. J. Chow, J. L. Earl. Science169, 577 (1970).[548] T. J. Chow, J. L. Earl. Science169, 577 (1970).. Using isotopic abundance data, the source of this toxic metal can be identified as it moves through air and water and eventually to living systems [547] [547] I. Renberg, M. L. Brännvall, R. Bindler, O. Emteryd. Ambio29, 150 (2000).[547] I. Renberg, M. L. Brännvall, R. Bindler, O. Emteryd. Ambio29, 150 (2000)., [549] [549] M. K. Reuer, D. J. Weiss. Math. Phys. Eng. Sci.360, 2889 (2002).[549] M. K. Reuer, D. J. Weiss. Math. Phys. Eng. Sci.360, 2889 (2002).. Scientists have analyzed lead in air pollution in California and found that it originated from Asia. Airborne particles from China have relatively higher amounts of 208Pb, which distinguishes the lead isotopic signature between airborne particles from Asia and North America. This knowledge could have implications in understanding the mixing of particles in the atmosphere and how pollutants are transported over vast distances [547] [547] I. Renberg, M. L. Brännvall, R. Bindler, O. Emteryd. Ambio29, 150 (2000).[547] I. Renberg, M. L. Brännvall, R. Bindler, O. Emteryd. Ambio29, 150 (2000)., [549] [549] M. K. Reuer, D. J. Weiss. Math. Phys. Eng. Sci.360, 2889 (2002).[549] M. K. Reuer, D. J. Weiss. Math. Phys. Eng. Sci.360, 2889 (2002)., [550] [550] S. A. Ewing, J. N. Christensen, S. T. Brown, R. A. Vancuren, S. S. Cliff, D. J. Depaolo. Environ. Sci. Technol.44, 8911 (2010).[550] S. A. Ewing, J. N. Christensen, S. T. Brown, R. A. Vancuren, S. S. Cliff, D. J. Depaolo. Environ. Sci. Technol.44, 8911 (2010)., [551] [551] D. Krotz. Lead Isotopes Yield Clues to How Asian Air Pollution Reaches California, Lawrence Berkeley National Laboratory News Center (2014), Feb. 25; http://newscenter.lbl.gov/feature-stories/2010/12/01/lead-isotopes-air-pollution/.[551] D. Krotz. Lead Isotopes Yield Clues to How Asian Air Pollution Reaches California, Lawrence Berkeley National Laboratory News Center (2014), Feb. 25; http://newscenter.lbl.gov/feature-stories/2010/12/01/lead-isotopes-air-pollution/.[551] D. Krotz. Lead Isotopes Yield Clues to How Asian Air Pollution Reaches California, Lawrence Berkeley National Laboratory News Center (2014), Feb. 25; http://newscenter.lbl.gov/feature-stories/2010/12/01/lead-isotopes-air-pollution/.[551] D. Krotz. Lead Isotopes Yield Clues to How Asian Air Pollution Reaches California, Lawrence Berkeley National Laboratory News Center (2014), Feb. 25; http://newscenter.lbl.gov/feature-stories/2010/12/01/lead-isotopes-air-pollution/.. Mapping the distribution of lead pollution by studying 204Pb, 206Pb, 207Pb and 208Pb also allows the identification of those human activities that contribute the highest amounts of lead to the environment [547] [547] I. Renberg, M. L. Brännvall, R. Bindler, O. Emteryd. Ambio29, 150 (2000).[547] I. Renberg, M. L. Brännvall, R. Bindler, O. Emteryd. Ambio29, 150 (2000)., [549] [549] M. K. Reuer, D. J. Weiss. Math. Phys. Eng. Sci.360, 2889 (2002).[549] M. K. Reuer, D. J. Weiss. Math. Phys. Eng. Sci.360, 2889 (2002)., [552] [552] D. Cicchella, B. De Vivo, A. Lima, S. Albanese, R. A. R. McGill, R. R. Parrish. Geochem. Explor. Environ. Anal.8, 103 (2008).[552] D. Cicchella, B. De Vivo, A. Lima, S. Albanese, R. A. R. McGill, R. R. Parrish. Geochem. Explor. Environ. Anal.8, 103 (2008)..
The measurement of the isotopic composition of lead in blood can help to determine the source of this toxic element in the body [553] [553] R. H. Gwiazda, D. R. Smith. Environ. Health Perspect.108, 1091 (2000).[553] R. H. Gwiazda, D. R. Smith. Environ. Health Perspect.108, 1091 (2000).. Lead is stored in bones and teeth. If a person moves to a different geographical region, the isotopic composition of the lead in the teeth is maintained, recording their place of origin. Bone can store lead for long periods of time (about 20 years), and some skeletal lead may be older and have a different isotopic composition than other skeletal lead. These differences reflect exposure to lead of different origins. By studying the isotope-amount ratio n(206Pb)/n(204Pb) and n(207Pb)/n(206Pb) in bone and teeth, it is possible to determine someone’s place of origin. For example, isotopes of lead were analyzed in the teeth and bones of a human mummy, known as the “Iceman”, to help determine his place of origin [554] [554] B. L. Gulson, B. R. Gillings. Environ. Health Perspect.105, 820 (1997).[554] B. L. Gulson, B. R. Gillings. Environ. Health Perspect.105, 820 (1997).[554] B. L. Gulson, B. R. Gillings. Environ. Health Perspect.105, 820 (1997).[554] B. L. Gulson, B. R. Gillings. Environ. Health Perspect.105, 820 (1997)., [555] [555] W. Müller, H. Fricke, A. N. Halliday, M. T. McCulloch, J. A. Wartho. Science302, 862 (2003).[555] W. Müller, H. Fricke, A. N. Halliday, M. T. McCulloch, J. A. Wartho. Science302, 862 (2003).[555] W. Müller, H. Fricke, A. N. Halliday, M. T. McCulloch, J. A. Wartho. Science302, 862 (2003).[555] W. Müller, H. Fricke, A. N. Halliday, M. T. McCulloch, J. A. Wartho. Science302, 862 (2003)..
210Pb is a relatively short-lived radioactive isotope of lead that is constantly produced by the decay of 222Rn in the atmosphere. While living, humans naturally incorporate 210Pb from the environment into bones and tissues. The amount of 210Pb in the body reaches equilibrium such that the 210Pb ingested is in equilibrium with the 210Pb that decays. When a person dies, this incorporation of 210Pb ceases and the relative amount of this isotope in the body decreases. Therefore, measurement of the 210Pb activity in a corpse can help determine time of death [556] [556] D. R. Smith, J. D. Osterloh, A. R. Flegal. Environ. Health Perspect.104, 60 (1996).[556] D. R. Smith, J. D. Osterloh, A. R. Flegal. Environ. Health Perspect.104, 60 (1996).[556] D. R. Smith, J. D. Osterloh, A. R. Flegal. Environ. Health Perspect.104, 60 (1996).[556] D. R. Smith, J. D. Osterloh, A. R. Flegal. Environ. Health Perspect.104, 60 (1996)., [557] [557] P. Rincon. “Isotopes could improve forensics”, in BBC News Online.[557] P. Rincon. “Isotopes could improve forensics”, in BBC News Online..
Lead isotope-amount ratios n(206Pb)/n(204Pb), n(207Pb)/n(204Pb), and n(208Pb)/n(204Pb)) along with isotope-amount ratio of silver, n(107Ag)/n(109Ag), and isotope-amount ratio of copper n(65Cu)/n(63Cu) have been used to determine the origin of European coins and to investigate the flow of goods in the world market over time [237] [237] A. M. Desaulty, P. Telouk, E. Albalat, F. Albarede. Proc. Natl. Acad. Sci.108, 9002 (2011).[237] A. M. Desaulty, P. Telouk, E. Albalat, F. Albarede. Proc. Natl. Acad. Sci.108, 9002 (2011).. Metals from Peru and Mexico and those from European mining have distinct isotopic signatures that enable the origin of the metal to be determined by examining the isotopic compositions of silver, copper, and lead in the coins. Abundant silver sources mined in Mexico and Peru in the 16 th century were used to mint coins, but were not a major influence in the European coin market until the 18 th century [237] [237] A. M. Desaulty, P. Telouk, E. Albalat, F. Albarede. Proc. Natl. Acad. Sci.108, 9002 (2011).[237] A. M. Desaulty, P. Telouk, E. Albalat, F. Albarede. Proc. Natl. Acad. Sci.108, 9002 (2011)..
References (13)
- [237] A. M. Desaulty, P. Telouk, E. Albalat, F. Albarede. Proc. Natl. Acad. Sci.108, 9002 (2011).
- [547] I. Renberg, M. L. Brännvall, R. Bindler, O. Emteryd. Ambio29, 150 (2000).
- [548] T. J. Chow, J. L. Earl. Science169, 577 (1970).
- [549] M. K. Reuer, D. J. Weiss. Math. Phys. Eng. Sci.360, 2889 (2002).
- [550] S. A. Ewing, J. N. Christensen, S. T. Brown, R. A. Vancuren, S. S. Cliff, D. J. Depaolo. Environ. Sci. Technol.44, 8911 (2010).
- [551] D. Krotz. Lead Isotopes Yield Clues to How Asian Air Pollution Reaches California, Lawrence Berkeley National Laboratory News Center (2014), Feb. 25; http://newscenter.lbl.gov/feature-stories/2010/12/01/lead-isotopes-air-pollution/.
- [552] D. Cicchella, B. De Vivo, A. Lima, S. Albanese, R. A. R. McGill, R. R. Parrish. Geochem. Explor. Environ. Anal.8, 103 (2008).
- [553] R. H. Gwiazda, D. R. Smith. Environ. Health Perspect.108, 1091 (2000).
- [554] B. L. Gulson, B. R. Gillings. Environ. Health Perspect.105, 820 (1997).
- [555] W. Müller, H. Fricke, A. N. Halliday, M. T. McCulloch, J. A. Wartho. Science302, 862 (2003).
- [556] D. R. Smith, J. D. Osterloh, A. R. Flegal. Environ. Health Perspect.104, 60 (1996).
- [557] P. Rincon. “Isotopes could improve forensics”, in BBC News Online.
- [4] IUPAC Periodic Table of the Elements and Isotopes (IPTEI) https://doi.org/10.1515/pac-2015-0703
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Lead.
The element property data was retrieved from publications.