Thallium (Tl)
post-transition-metalSolid
Standard Atomic Weight
204.38 u [204.382, 204.385]Electron configuration
[Xe] 6s2 4f14 5d10 6p1Melting point
303.85 °C (577 K)Boiling point
1472.85 °C (1746 K)Density
1.180000e+4 kg/m³Oxidation states
−5, −2, −1, +1, +2, +3Electronegativity (Pauling)
1.62Ionization energy (1st)
Discovery year
1861Atomic radius
190 pmDetails
Thallium is a soft post-transition metal in group 13. It is chemically notable for the stability of the +1 oxidation state, which reflects the inert-pair effect and makes many thallium(I) salts resemble alkali-metal salts in size and solubility behavior. The element is rare in ores and is usually recovered as a by-product of processing sulfide minerals. Its severe toxicity has greatly reduced former consumer and agricultural uses.
When freshly exposed to air, thallium exhibits a metallic luster, but soon develops a bluish-gray tinge, resembling lead in appearance. A heavy oxide builds up on thallium if left in air, and in the presence of water the hydride is formed. The metal is very soft and malleable. It can be cut with a knife. Twenty five isotopic forms of thallium, with atomic masses ranging from 184 to 210 are recognized. Natural thallium is a mixture of two isotopes. A mercury-thallium alloy, which forms a eutectic at 8.5% thallium, is reported to freeze at -60C, some 20 degrees below the freezing point of mercury.
The name derives from the Greek thallos for "green shoot" or "twig" because of the bright green line in its spectrum. Thallium was discovered by the English physicist and chemist William Crookes in 1861. Metallic thallium was first isolated by the French chemist Claude-Auguste Lamy in 1862.
Thallium was discovered spectroscopically by Sir William Crookes, an English chemist, in 1861. Crooks had obtained the sludge left over from the production of sulfuric acid (H2SO4) from a friend. After removing all of the selenium from the sludge, he inspected it with a device known as a spectroscope to look for signs of tellurium. Rather than seeing the yellow spectral lines produced by tellurium, he observed a bright green line that no one had ever seen before. He named the new element that was producing the green line thallium, after the greek word for 'green twig', thallos. He isolated samples of thallium the next year. Thallium is found in the minerals crooksite (CuThSe), lorandite (TlAsS2) and hutchinsonite ((Pb, Tl)2As5S9), but is usually obtained as a byproduct of the production of sulfuric acid or as a byproduct of refining zinc or lead.
From Greek thallos, meanin a green shoot or twig. Thallium was discovered spectroscopically in 1861 by Crookes. The element was named after the beautiful green spectral line, which identified the element. The metal was isolated both by Crookes and by Lamy in 1862 at about the same time.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
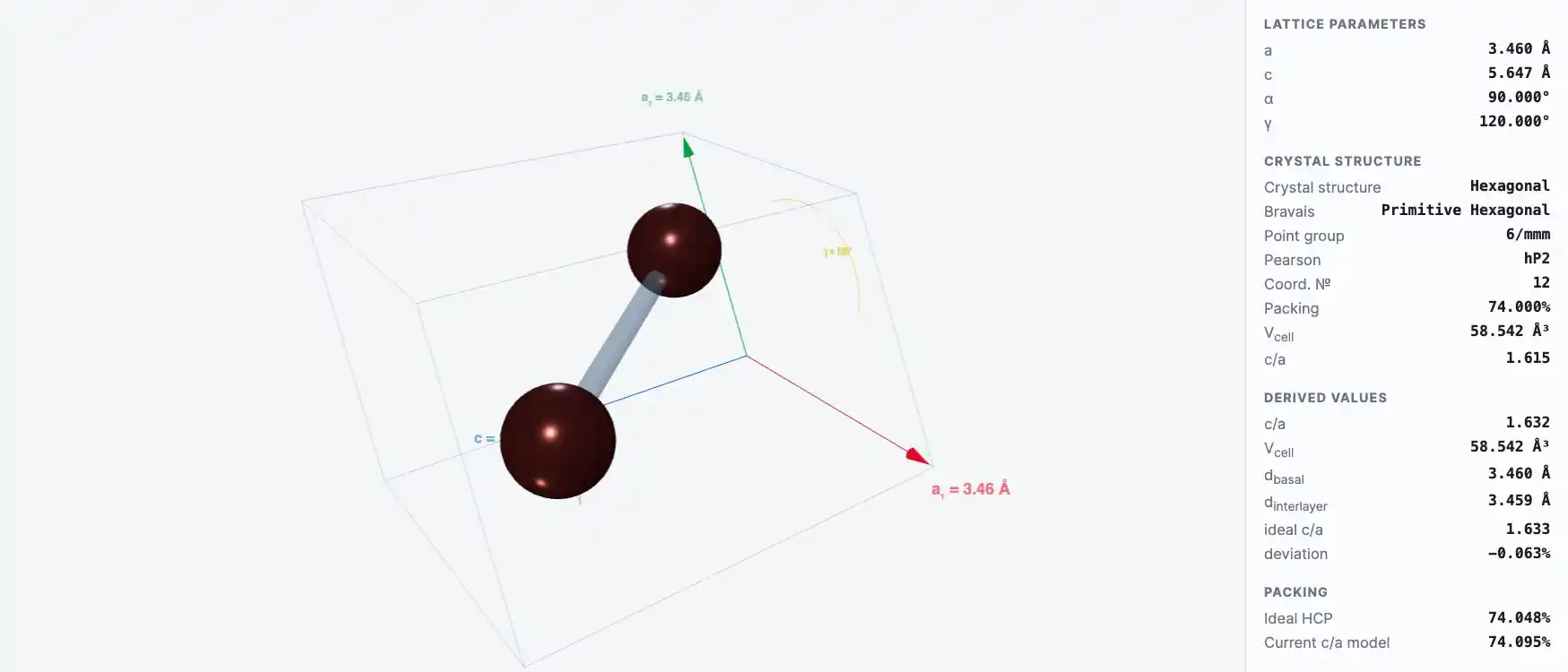
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Tl: 4f¹⁴ 5d¹⁰ 6s² 6p¹[Xe] 4f¹⁴ 5d¹⁰ 6s² 6p¹1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d¹⁰ 6s² 6p¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 203 Stable | 202.9723446 ± 0.0000014 | 29.5200% | Stable |
| 205 Stable | 204.9744278 ± 0.0000014 | 70.4800% | Stable |
Phase / State
Reason: 278.9 °C below melting point (303.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 81 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Tl I | 0 | 65 | 25 | 11 |
| Tl II | +1 | 82 | 3 | 7 |
| Tl III | +2 | 22 | 0 | 0 |
| Tl IV | +3 | 35 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Tl I | 0 | 70 |
| Tl II | +1 | 82 |
| Tl III | +2 | 109 |
| Tl IV | +3 | 44 |
| Tl V | +4 | 2 |
| Tl VI | +5 | 2 |
| Tl VII | +6 | 2 |
| Tl VIII | +7 | 2 |
| Tl IX | +8 | 2 |
| Tl X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +1 | 6 | N/A | 150 pm |
| +1 | 8 | N/A | 159 pm |
| +1 | 12 | N/A | 170 pm |
| +3 | 4 | N/A | 75 pm |
| +3 | 6 | N/A | 88.5 pm |
| +3 | 8 | N/A | 98 pm |
Compounds
Isotopes (2)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 203 Stable | 202.9723446 ± 0.0000014 | 29.5200% ± 0.0100% | Stable | stable | |
| 205 Stable | 204.9744278 ± 0.0000014 | 70.4800% ± 0.0100% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Phase Transitions & Allotropes
| Melting point | 577.15 K |
| Boiling point | 1746.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (15)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.5591 |
| 2 | p | 4.5138 |
| 2 | s | 21.3158 |
| 3 | d | 13.4658 |
| 3 | p | 22.6335 |
| 3 | s | 23.5809 |
| 4 | d | 37.6112 |
| 4 | f | 38.1324 |
| 4 | p | 35.7832 |
| 4 | s | 34.9212 |
Crystal Radii Detail (6)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 1 | VI | 164 | from r^3 vs V plots, | |
| 1 | VIII | 173 | from r^3 vs V plots, | |
| 1 | XII | 184 | from r^3 vs V plots, estimated, | |
| 3 | IV | 89 | ||
| 3 | VI | 102.5 | from r^3 vs V plots, | |
| 3 | VIII | 112 | calculated, |
Isotope Decay Modes (69)
| Isotope | Mode | Intensity |
|---|---|---|
| 176 | p | 100% |
| 176 | A | — |
| 176 | B+ | — |
| 177 | A | 73% |
| 177 | p | — |
| 178 | A | 62% |
| 178 | B+ | 38% |
| 178 | B+SF | 0.1% |
| 179 | A | 60% |
| 179 | B+ | — |
X‑ray Scattering Factors (516)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.80392 |
| 10.1617 | — | 1.75555 |
| 10.3261 | — | 1.69589 |
| 10.4931 | — | 1.59546 |
| 10.6628 | — | 1.46625 |
| 10.8353 | — | 1.39341 |
| 11.0106 | — | 1.31349 |
| 11.1886 | — | 1.20783 |
| 11.3696 | — | 1.13261 |
| 11.5535 | — | 1.07387 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
8.5×10-1 milligrams per kilogram
References (1)
- [5] Thallium https://education.jlab.org/itselemental/ele081.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.9×10-5 milligrams per liter
References (1)
- [5] Thallium https://education.jlab.org/itselemental/ele081.html
Sources
Sources of this element.
Thallium occurs in crooksite, lorandite, and hutchinsonite. It is also present in pyrites and is recovered from the roasting of this ore in connection with the production of sulfuric acid. It is also obtained from the smelting of lead and zinc ores. Extraction is somewhat complex and depends on the source of the thallium. Manganese nodules, found on the ocean floor, contain thallium.
References (1)
- [6] Thallium https://periodic.lanl.gov/81.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Thallium.
The element property data was retrieved from publications.