Bismuth (Bi)
post-transition-metalSolid
Standard Atomic Weight
208.9804 uElectron configuration
[Xe] 6s2 4f14 5d10 6p3Melting point
271.4 °C (544.55 K)Boiling point
1563.85 °C (1837 K)Density
9807 kg/m³Oxidation states
−3, −2, −1, 0, +1, +2, +3, +4, +5Electronegativity (Pauling)
2.02Ionization energy (1st)
Discovery year
1753Atomic radius
160 pmDetails
Bismuth is a heavy post-transition metal and the heaviest element with a primordial isotope that is effectively stable on human timescales. Natural bismuth is almost entirely ²⁰⁹Bi, now known to be very weakly radioactive by alpha decay. It has unusually low toxicity for a heavy metal, low thermal conductivity, a low melting point, and a strong tendency to form +3 compounds. These traits make it useful where lead, cadmium, or mercury are undesirable.
It is a white, crystalline, brittle metal with a pinkish tinge. It occurs in a native state. Bismuth is the most diamagnetic of all metals, and the thermal conductivity is lower than any metal, except mercury. It has a high electrical resistance, and has the highest Hall effect of any metal (i.e., greatest increase in electrical resistance when placed in a magnetic field).
The name derives from the German weisse masse for "white mass" from the colour of its oxides. The ancients did not distinguish bismuth from lead. The French chemist Claude-Francois Geoffroy showed that bismuth was distinct from lead in 1753.
Bismuth, which has been known since ancient times, was often confused with lead and tin. Bismuth was first shown to be a distinct element in 1753 by Claude Geoffroy the Younger. Bismuth does occur free in nature and in such minerals as bismuthinite (Bi2S3) and bismite (Bi2O3). The largest deposits of bismuth are found in Bolivia, although bismuth is usually obtained as a by-product of mining and refining lead, copper, tin, silver and gold.
From the German Weisse Masse, meaning white mass; later Wisuth and Bisemutum. In early times bismuth was confused with tin and lead. Claude Geoffroy the Younger showed it to be distinct from lead in 1753.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
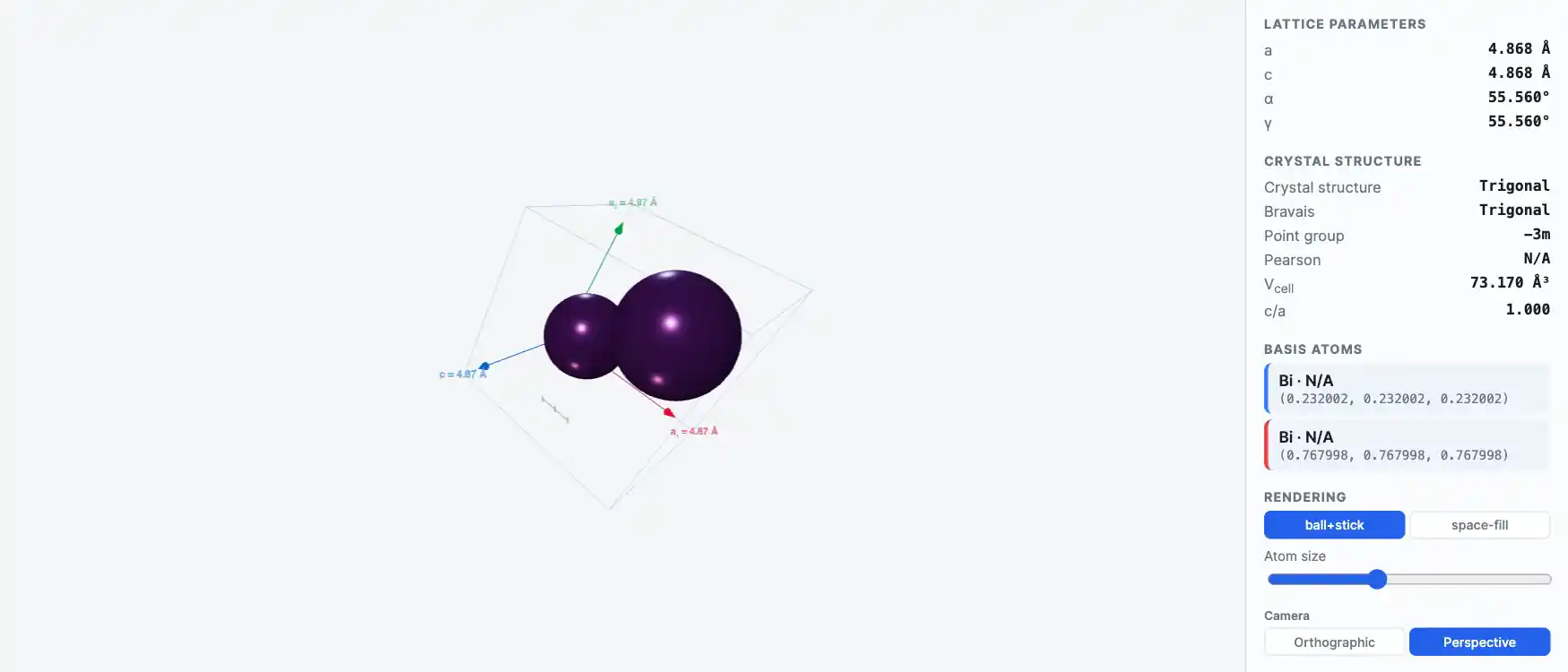
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Bi: 4f¹⁴ 5d¹⁰ 6s² 6p³[Xe] 4f¹⁴ 5d¹⁰ 6s² 6p³1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d¹⁰ 6s² 6p³Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
No stable isotopes.
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 189 Radioactive | 188.989195 ± 0.000022 | N/A | 688 ms |
| 208 Radioactive | 207.9797425 ± 0.0000025 | N/A | 368 ky |
| 195 Radioactive | 194.9806488 ± 0.0000057 | N/A | 183 seconds |
| 201 Radioactive | 200.97701 ± 0.000016 | N/A | 103 minutes |
| 217 Radioactive | 217.009372 ± 0.000019 | N/A | 98.5 seconds |
Phase / State
Reason: 246.4 °C below melting point (271.4 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 83 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Bi I | 0 | 69 | 39 | 63 |
| Bi II | +1 | 111 | 4 | 16 |
| Bi III | +2 | 204 | 204 | 204 |
| Bi IV | +3 | 45 | 0 | 0 |
| Bi V | +4 | 18 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Bi I | 0 | 75 |
| Bi II | +1 | 78 |
| Bi III | +2 | 68 |
| Bi IV | +3 | 38 |
| Bi V | +4 | 15 |
| Bi VI | +5 | 115 |
| Bi VII | +6 | 2 |
| Bi VIII | +7 | 2 |
| Bi IX | +8 | 2 |
| Bi X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 5 | N/A | 96 pm |
| +3 | 6 | N/A | 103 pm |
| +3 | 8 | N/A | 117 pm |
| +5 | 6 | N/A | 76 pm |
Compounds
Isotopes (5)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 189 Radioactive | 188.989195 ± 0.000022 | N/A | 688 ms | α ≈100%β+ ? | |
| 208 Radioactive | 207.9797425 ± 0.0000025 | N/A | 368 ky | β+ =100% | |
| 195 Radioactive | 194.9806488 ± 0.0000057 | N/A | 183 seconds | β+ ≈100%α =0.030±1.2% | |
| 201 Radioactive | 200.97701 ± 0.000016 | N/A | 103 minutes | β+ =100% | |
| 217 Radioactive | 217.009372 ± 0.000019 | N/A | 98.5 seconds | β- =100% |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 384.893 nm | 21 | Bi III | emission | 6s2.6d 2D → 6s2.7p 2P* | Measured | NIST | |
| 392.72 nm | N/A | Bi III | emission | 6s.6p2.(3P) 2P → 6s2.8p 2P* | Measured | NIST | |
| 393.036 nm | N/A | Bi III | emission | 6s.6p2.(1S) 2S → 6s2.8p 2P* | Measured | NIST | |
| 422.469 nm | 350 | Bi III | emission | 6s2.6f 2F* → 6s2.7g 2G | Measured | NIST | |
| 423.421 nm | 280 | Bi III | emission | 6s2.6f 2F* → 6s2.7g 2G | Measured | NIST | |
| 425.9413 nm | N/A | Bi II | emission | 6s2.6p.6d (1/2,5/2)* → 6s2.6p.5f (1/2,7/2) | Measured | NIST | |
| 430.1697 nm | N/A | Bi II | emission | 6s2.6p.6d (1/2,5/2)* → 6s2.6p.5f (1/2,7/2) | Measured | NIST | |
| 430.653 nm | N/A | Bi III | emission | 6s.6p2.(1S) 2S → 6s2.8p 2P* | Measured | NIST | |
| 432.792 nm | 360 | Bi III | emission | 6s2.7p 2P* → 6s2.8s 2S | Measured | NIST | |
| 456.143 nm | N/A | Bi III | emission | 6s2.7s 2S → 6s2.7p 2P* | Measured | NIST | |
| 470.5285 nm | N/A | Bi II | emission | 6s2.6p.7p (1/2,1/2) → 6s2.6p.7d (1/2,3/2)* | Measured | NIST | |
| 472.883 nm | N/A | Bi III | emission | 6s.6p2.(1D) 2D → 6s2.5f 2F* | Measured | NIST | |
| 475.128 nm | N/A | Bi III | emission | 6s.6p2.(1D) 2D → 6s2.5f 2F* | Measured | NIST | |
| 479.742 nm | N/A | Bi III | emission | 6s2.6d 2D → 6s2.7p 2P* | Measured | NIST | |
| 480.9082 nm | N/A | Bi III | emission | 6s2.6p 2P* → 6s2.6p 2P* | Measured | NIST | |
| 505.178 nm | N/A | Bi III | emission | 6s2.5g 2G → 6s2.7h 2H* | Measured | NIST | |
| 505.178 nm | N/A | Bi III | emission | 6s2.5g 2G → 6s2.7h 2H* | Measured | NIST | |
| 505.244 nm | 120 | Bi III | emission | 6s2.5g 2G → 6s2.7h 2H* | Measured | NIST | |
| 507.928 nm | N/A | Bi III | emission | 6s2.6d 2D → 6s2.7p 2P* | Measured | NIST | |
| 512.4356 nm | N/A | Bi II | emission | 6s2.6p.7s (3/2,1/2)* → 6s2.6p.7p (3/2,3/2) | Measured | NIST | |
| 514.4507 nm | N/A | Bi II | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.7p (1/2,3/2) | Measured | NIST | |
| 520.9325 nm | N/A | Bi II | emission | 6s2.6p.7s (1/2,1/2)* → 6s2.6p.7p (1/2,3/2) | Measured | NIST | |
| 613.907 nm | 22 | Bi III | emission | 6s2.6f 2F* → 6s2.6g 2G | Measured | NIST | |
| 614.039 nm | 150 | Bi III | emission | 6s2.6f 2F* → 6s2.6g 2G | Measured | NIST | |
| 616.071 nm | 120 | Bi III | emission | 6s2.6f 2F* → 6s2.6g 2G | Measured | NIST | |
| 662.323 nm | 180 | Bi III | emission | 6s2.8p 2P* → 6s2.8d 2D | Measured | NIST | |
| 738.23 nm | N/A | Bi III | emission | 6s.6p2.(1D) 2D → 6s2.7p 2P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 544.55 K |
| Boiling point | 1837.15 K |
| Critical point (temperature) | 4620.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (15)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.6018 |
| 2 | p | 4.533 |
| 2 | s | 21.824 |
| 3 | d | 13.4585 |
| 3 | p | 23.0678 |
| 3 | s | 24.1145 |
| 4 | d | 37.7608 |
| 4 | f | 37.9308 |
| 4 | p | 36.1496 |
| 4 | s | 35.2928 |
Crystal Radii Detail (4)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | V | 110 | calculated, | |
| 3 | VI | 117 | from r^3 vs V plots, | |
| 3 | VIII | 131 | from r^3 vs V plots, | |
| 5 | VI | 90 | estimated, |
Isotope Decay Modes (70)
| Isotope | Mode | Intensity |
|---|---|---|
| 184 | A | 100% |
| 185 | p | — |
| 185 | A | — |
| 186 | A | 100% |
| 186 | B+ | — |
| 186 | B+SF | 0% |
| 187 | A | 100% |
| 188 | A | 100% |
| 188 | B+ | — |
| 188 | B+SF | 0% |
X‑ray Scattering Factors (516)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 5.59475 |
| 10.1617 | — | 5.63587 |
| 10.3261 | — | 5.67729 |
| 10.4931 | — | 5.71901 |
| 10.6628 | — | 5.74574 |
| 10.8353 | — | 5.7564 |
| 11.0106 | — | 5.76707 |
| 11.1886 | — | 5.77776 |
| 11.3696 | — | 5.78847 |
| 11.5535 | — | 5.7834 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
8.5×10-3 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
2×10-5 milligrams per liter
References (1)
Sources
Sources of this element.
The most important ores are bismuthinite or bismuth glance and bismite. Peru, Japan, Mexico, Bolivia, and Canada are major bismuth producers. Much of the bismuth produced in the U.S. is obtained as a by-product in refining lead, copper, tin, silver, and gold ores.
References (1)
- [6] Bismuth https://periodic.lanl.gov/83.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Bismuth.
The element property data was retrieved from publications.