Zirconium (Zr)
transition-metalSolid
Standard Atomic Weight
91.224 uElectron configuration
[Kr] 5s2 4d2Melting point
1854.85 °C (2128 K)Boiling point
4408.85 °C (4682 K)Density
6520 kg/m³Oxidation states
+1, +2, +3, +4Electronegativity (Pauling)
1.33Ionization energy (1st)
Discovery year
1789Atomic radius
155 pmDetails
Zirconium is a lustrous transition metal in group 4, chemically close to hafnium and titanium. It occurs mainly in zircon and related heavy minerals, almost always with hafnium as a companion. The metal is valued for its very low thermal-neutron absorption and its stable, adherent oxide film, which give it a central role in nuclear reactor materials and in corrosion-resistant alloys.
Reactor-grade zirconium is essentially free of hafnium. Zircaloy(R) is an important alloy developed specifically for nuclear applications. Zirconium is exceptionally resistant to corrosion by many common acids and alkalis, by sea water, and by other agents. Alloyed with zinc, zirconium becomes magnetic at temperatures below 35°K.
The name derives from the Arabic zargun for "gold-like". It was discovered in zirconia by the German chemist Martin-Heinrich Klaproth in 1789. Zirconium was first isolated by Swedish chemist Jöns Jacob Berzelius in 1824 in an impure state, and finally by the chemists D. Lely, Jr. and L. Hamburger in a pure state in 1914.
Zirconium was discovered by Martin Heinrich Klaproth, a German chemist, while analyzing the composition of the mineral jargon (ZrSiO4) in 1789. Zirconium was isolated by Jöns Jacob Berzelius, a Swedish chemist, in 1824 and finally prepared in a pure form in 1914. Obtaining pure zirconium is very difficult because it is chemically similar to hafnium, an element which is always found mixed with deposits of zirconium. Today, most zirconium is obtained from the minerals zircon (ZrSiO4) and baddeleyite (ZrO2) through a process known as the Kroll Process.
From the Persian zargun, gold like. Zircon, the primary gemstone of zirconium, is also known as jargon, hyacinth, jacinth, or ligure. This mineral, or its variations, is mentioned in biblical writings. The mineral was not known to contain a new element until Klaproth, in 1789, analyzed a jargon from Ceylon and identified the new element, which Werner named zircon (silex circonius), and which Klaproth called Zirkonertz (zirconia). The impure metal was first isolated by Berzelius in 1824 by heating a mixture of potassium and potassium zirconium fluoride in a small decomposition process they developed.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
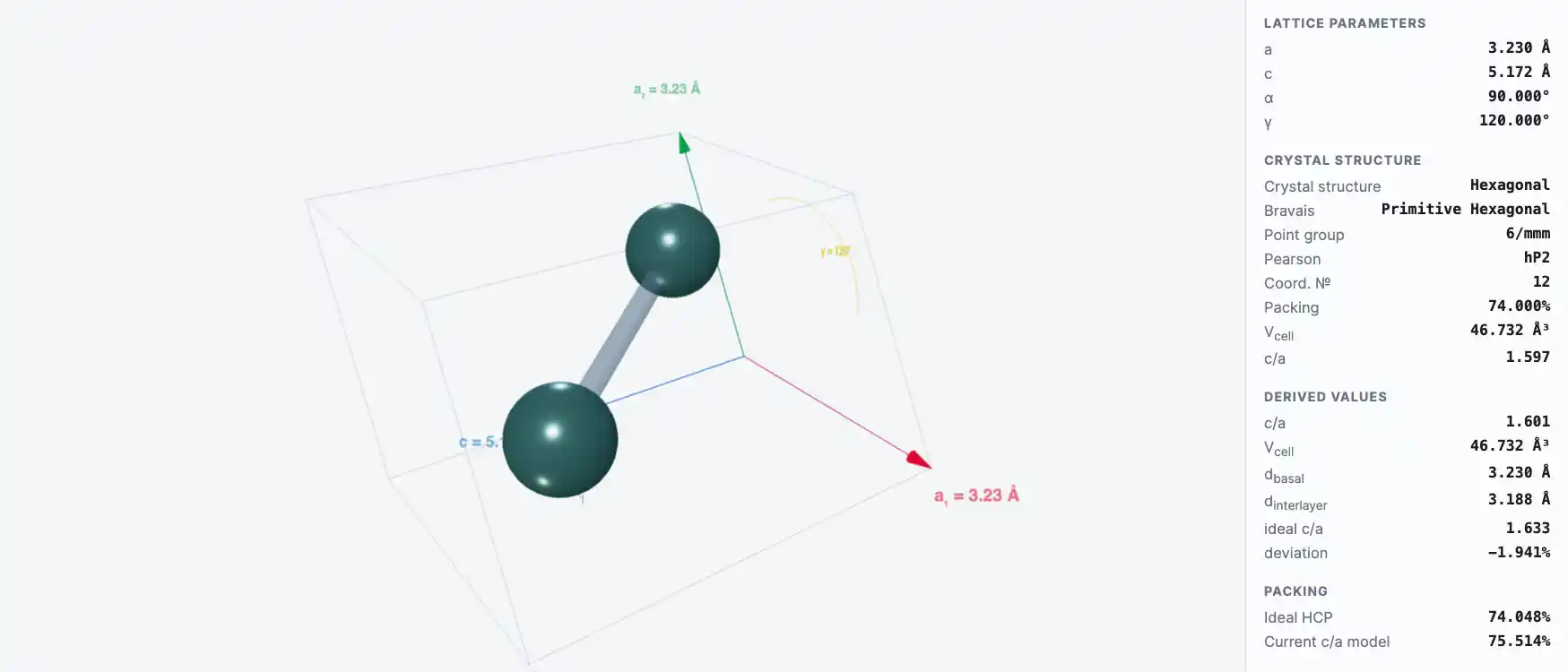
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Zr: 4d² 5s²[Kr] 4d² 5s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d² 5s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 90 Stable | 89.9046977 ± 0.000002 | 51.4500% | Stable |
| 91 Stable | 90.9056396 ± 0.000002 | 11.2200% | Stable |
| 92 Stable | 91.9050347 ± 0.000002 | 17.1500% | Stable |
Phase / State
Reason: 1829.8 °C below melting point (1854.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 40 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Zr I | 0 | 459 | 0 | 0 |
| Zr II | +1 | 207 | 0 | 0 |
| Zr III | +2 | 490 | 490 | 490 |
| Zr IV | +3 | 76 | 0 | 76 |
| Zr V | +4 | 104 | 0 | 0 |
| Zr VI | +5 | 427 | 427 | 427 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Zr I | 0 | 262 |
| Zr II | +1 | 136 |
| Zr III | +2 | 140 |
| Zr IV | +3 | 35 |
| Zr V | +4 | 102 |
| Zr VI | +5 | 97 |
| Zr VII | +6 | 2 |
| Zr VIII | +7 | 2 |
| Zr IX | +8 | 2 |
| Zr X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +4 | 4 | N/A | 59 pm |
| +4 | 5 | N/A | 66 pm |
| +4 | 6 | N/A | 72 pm |
| +4 | 7 | N/A | 78 pm |
| +4 | 8 | N/A | 84 pm |
| +4 | 9 | N/A | 89 pm |
Compounds
Isotopes (3)
Naturally occurring zirconium contains five isotopes. Fifteen other isotopes are known to exist. Zircon, ZrSiO4, the principal ore, is pure ZrO2 in crystalline form having a hafnium content of about 1%. Zirconium also occurs in some 30 other recognized mineral species. Zirconium is produced commercially by reduction of chloride with magnesium (the Kroll Process), and by other methods. It is a grayish-white lustrous metal. When finely divided, the metal may ignite spontaneously in air, especially at elevated temperatures. The solid metal is much more difficult to ignite. The inherent toxicity of zirconium compounds is low. Hafnium is invariably found in zirconium ores, and the separation is difficult.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 90 Stable | 89.9046977 ± 0.000002 | 51.4500% ± 0.4000% | Stable | stable | |
| 91 Stable | 90.9056396 ± 0.000002 | 11.2200% ± 0.0500% | Stable | stable | |
| 92 Stable | 91.9050347 ± 0.000002 | 17.1500% ± 0.0800% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 382.0196 nm | 5 | Zr III | emission | 4d.4f 3G* → 4d.(2D<5/2>).5g 2[7/2] | Measured | NIST | |
| 382.4611 nm | 250 | Zr III | emission | 4d.4f 3H* → 4d.(2D<3/2>).5g 2[9/2] | Measured | NIST | |
| 382.7722 nm | 300 | Zr III | emission | 4d.4f 3F* → 4d.(2D<3/2>).5g 2[7/2] | Measured | NIST | |
| 382.923 nm | 600 | Zr III | emission | 4d.4f 3H* → 4d.(2D<3/2>).5g 2[11/2] | Measured | NIST | |
| 383.0087 nm | 250 | Zr III | emission | 4d.4f 1D* → 4d.(2D<5/2>).5g 2[7/2] | Measured | NIST | |
| 383.7038 nm | 10 | Zr III | emission | 4d.4f 3G* → 4d.(2D<5/2>).5g 2[9/2] | Measured | NIST | |
| 384.2399 nm | 270 | Zr III | emission | 4d.4f 3F* → 4d.(2D<3/2>).5g 2[9/2] | Measured | NIST | |
| 390.7626 nm | 5 | Zr III | emission | 4d.4f 3G* → 4d.(2D<5/2>).5g 2[7/2] | Measured | NIST | |
| 391.0786 nm | 3 | Zr III | emission | 4d.4f 3G* → 4d.(2D<5/2>).5g 2[13/2] | Measured | NIST | |
| 391.6928 nm | 100 | Zr III | emission | 4d.4f 3F* → 4d.(2D<3/2>).5g 2[9/2] | Measured | NIST | |
| 392.0624 nm | 400 | Zr III | emission | 4d.4f 3G* → 4d.(2D<5/2>).5g 2[11/2] | Measured | NIST | |
| 392.5804 nm | 200 | Zr III | emission | 4d.4f 3G* → 4d.(2D<5/2>).5g 2[9/2] | Measured | NIST | |
| 392.694 nm | 120 | Zr III | emission | 4d.4f 3G* → 4d.(2D<5/2>).5g 2[9/2] | Measured | NIST | |
| 393.1478 nm | 100 | Zr III | emission | 5s.5p 3P* → 4d.5d 3S | Measured | NIST | |
| 396.3178 nm | 500 | Zr III | emission | 4d.4f 3G* → 4d.(2D<5/2>).5g 2[11/2] | Measured | NIST | |
| 396.5231 nm | 10 | Zr III | emission | 4d.4f 3G* → 4d.(2D<5/2>).5g 2[11/2] | Measured | NIST | |
| 397.1691 nm | 200 | Zr III | emission | 4d.4f 3G* → 4d.(2D<5/2>).5g 2[9/2] | Measured | NIST | |
| 397.3984 nm | 220 | Zr III | emission | 4d.4f 1D* → 4d.(2D<3/2>).5g 2[5/2] | Measured | NIST | |
| 398.854 nm | 10 | Zr III | emission | 4d.4f 3D* → 4d.(2D<5/2>).5g 2[5/2] | Measured | NIST | |
| 401.632 nm | 20 | Zr III | emission | 4d.4f 3G* → 4d.(2D<3/2>).5g 2[7/2] | Measured | NIST | |
| 401.6949 nm | 35 | Zr III | emission | 4d.4f 3G* → 4d.(2D<3/2>).5g 2[7/2] | Measured | NIST | |
| 401.7561 nm | 3 | Zr III | emission | 4d.4f 1F* → 4d.(2D<5/2>).5g 2[7/2] | Measured | NIST | |
| 401.8142 nm | 140 | Zr III | emission | 4d.4f 1F* → 4d.(2D<5/2>).5g 2[7/2] | Measured | NIST | |
| 403.2482 nm | 400 | Zr III | emission | 4d.4f 3G* → 4d.(2D<3/2>).5g 2[9/2] | Measured | NIST | |
| 403.3591 nm | 180 | Zr III | emission | 4d.4f 3D* → 4d.(2D<5/2>).5g 2[7/2] | Measured | NIST | |
| 403.6779 nm | 200 | Zr III | emission | 4d.4f 1F* → 4d.(2D<5/2>).5g 2[9/2] | Measured | NIST | |
| 408.0264 nm | 5 | Zr III | emission | 4d.4f 3G* → 4d.(2D<3/2>).5g 2[11/2] | Measured | NIST | |
| 408.7114 nm | 150 | Zr III | emission | 4d.4f 3D* → 4d.(2D<5/2>).5g 2[5/2] | Measured | NIST | |
| 412.5432 nm | 200 | Zr III | emission | 4d.4f 3G* → 4d.(2D<3/2>).5g 2[11/2] | Measured | NIST | |
| 412.6379 nm | 400 | Zr III | emission | 4d.4f 3D* → 4d.(2D<5/2>).5g 2[7/2] | Measured | NIST | |
| 413.2087 nm | 200 | Zr III | emission | 4d.4f 3G* → 4d.(2D<3/2>).5g 2[9/2] | Measured | NIST | |
| 413.7442 nm | 500 | Zr IV | emission | 4p6.5d 2D → 4p6.6p 2P* | Measured | NIST | |
| 414.6654 nm | 20 | Zr III | emission | 4d.4f 3D* → 4d.(2D<5/2>).5g 2[9/2] | Measured | NIST | |
| 415.3368 nm | 2 | Zr III | emission | 4d.4f 3P* → 4d.(2D<5/2>).5g 2[3/2] | Measured | NIST | |
| 416.0827 nm | 250 | Zr III | emission | 4d.4f 3D* → 4d.(2D<3/2>).5g 2[5/2] | Measured | NIST | |
| 416.5293 nm | 15 | Zr III | emission | 4d.4f 3P* → 4d.(2D<5/2>).5g 2[5/2] | Measured | NIST | |
| 417.1353 nm | 20 | Zr III | emission | 4d.4f 3P* → 4d.(2D<5/2>).5g 2[3/2] | Measured | NIST | |
| 417.2872 nm | 300 | Zr III | emission | 4d.4f 3P* → 4d.(2D<5/2>).5g 2[5/2] | Measured | NIST | |
| 419.3504 nm | 275 | Zr III | emission | 4d.4f 3D* → 4d.(2D<3/2>).5g 2[5/2] | Measured | NIST | |
| 419.7309 nm | 15 | Zr III | emission | 4d.4f 3P* → 4d.(2D<5/2>).5g 2[3/2] | Measured | NIST | |
| 419.8266 nm | 3000 | Zr IV | emission | 4p6.5d 2D → 4p6.6p 2P* | Measured | NIST | |
| 420.3546 nm | 200 | Zr III | emission | 4d.4f 3P* → 4d.(2D<5/2>).5g 2[5/2] | Measured | NIST | |
| 423.5695 nm | 275 | Zr III | emission | 4d.4f 1F* → 4d.(2D<3/2>).5g 2[7/2] | Measured | NIST | |
| 431.7077 nm | 2000 | Zr IV | emission | 4p6.5d 2D → 4p6.6p 2P* | Measured | NIST | |
| 434.2686 nm | 400 | Zr III | emission | 4d.4f 1H* → 4d.(2D<5/2>).5g 2[13/2] | Measured | NIST | |
| 440.7385 nm | 20 | Zr III | emission | 4d.4f 1H* → 4d.(2D<5/2>).5g 2[11/2] | Measured | NIST | |
| 456.1637 nm | 50 | Zr III | emission | 4d.4f 1P* → 4d.(2D<5/2>).5g 2[3/2] | Measured | NIST | |
| 456.922 nm | 1800 | Zr IV | emission | 4p6.5g 2G → 4p6.6h 2H* | Measured | NIST | |
| 456.927 nm | 1800 | Zr IV | emission | 4p6.5g 2G → 4p6.6h 2H* | Measured | NIST | |
| 460.8973 nm | 60 | Zr III | emission | 4d.4f 1H* → 4d.(2D<3/2>).5g 2[11/2] | Measured | NIST | |
| 500.71 nm | N/A | ID 803 | emission | 2p 2P* → 2s 2S | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 2127.15 K |
| Boiling point | 4679.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (10)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.841 |
| 2 | p | 4.0072 |
| 2 | s | 10.6262 |
| 3 | d | 14.4331 |
| 3 | p | 16.1545 |
| 3 | s | 15.6385 |
| 4 | d | 26.9284 |
| 4 | p | 26.54 |
| 4 | s | 25.0984 |
| 5 | s | 33.5545 |
Crystal Radii Detail (6)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 4 | IV | 73 | from r^3 vs V plots, | |
| 4 | V | 80 | calculated, | |
| 4 | VI | 86 | from r^3 vs V plots, | |
| 4 | VII | 92 | ||
| 4 | VIII | 98 | ||
| 4 | IX | 103 |
Isotope Decay Modes (56)
| Isotope | Mode | Intensity |
|---|---|---|
| 77 | B+ | — |
| 77 | B+p | — |
| 77 | p | — |
| 78 | B+ | — |
| 78 | B+p | — |
| 79 | B+ | 100% |
| 79 | B+p | — |
| 80 | B+ | 100% |
| 81 | B+ | 100% |
| 81 | B+p | 0.1% |
X‑ray Scattering Factors (724)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 1 | — | 0.18706 |
| 1.0149 | — | 0.19051 |
| 1.0299 | — | 0.19402 |
| 1.0452 | — | 0.1976 |
| 1.0608 | — | 0.20124 |
| 1.0765 | — | 0.20499 |
| 1.0925 | — | 0.20885 |
| 1.1087 | — | 0.21277 |
| 1.1252 | — | 0.21677 |
| 1.142 | — | 0.22085 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.65×102 milligrams per kilogram
References (1)
- [5] Zirconium https://education.jlab.org/itselemental/ele040.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
3×10-5 milligrams per liter
References (1)
- [5] Zirconium https://education.jlab.org/itselemental/ele040.html
Sources
Sources of this element.
Zirconium is produced from the mineral zircon (ZrSiO4). It is found in abundance in S-type stars, and has been identified in the sun and meteorites. Analysis of lunar rock samples obtained during the various Apollo missions to the moon show a surprisingly high zirconium oxide content, compared with terrestrial rocks.
References (1)
- [6] Zirconium https://periodic.lanl.gov/40.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Zirconium.
The element property data was retrieved from publications.