Yttrium (Y)
transition-metalSolid
Standard Atomic Weight
88.90584 uElectron configuration
[Kr] 5s2 4d1Melting point
1521.85 °C (1795 K)Boiling point
3344.85 °C (3618 K)Density
4470 kg/m³Oxidation states
0, +1, +2, +3Electronegativity (Pauling)
1.22Ionization energy (1st)
Discovery year
1794Atomic radius
180 pmDetails
Yttrium is a silvery transition metal grouped with the rare-earth elements because it commonly occurs with lanthanides and forms predominantly trivalent cations. Its chemistry is close to the heavier lanthanides, especially holmium and erbium, rather than to scandium. Although not itself a lanthanide, yttrium is a key component of phosphors, ceramics, lasers, and high-temperature oxide materials.
Yttrium has a silver-metallic luster and is relatively stable in air. Turnings of the metal, however, ignite in air if their temperature exceeds 400°C. Finely divided yttrium is very unstable in air.
The name derives from the Swedish village of Ytterby where the mineral gadolinite was found. In 1794, the Finnish chemist Johan Gadolin discovered yttrium in the mineral ytterbite, which was later renamed gadolinite for Gadolin. Gadolin originally called the element ytterbium after ytterbite. The name was subsequently shortened to yttrium, and later another element was given the name ytterbium.
Yttrium was discovered by Johan Gadolin, a Finnish chemist, while analyzing the composition of the mineral gadolinite ((Ce, La, Nd, Y)2FeBe2Si2O10) in 1789. Gadolinite, which was named for Johan Gadolin, was discovered several years earlier in a quarry near the town of Ytterby, Sweden. Today, yttrium is primarily obtained through an ion exchange process from monazite sand ((Ce, La, Th, Nd, Y)PO4), a material rich in rare earth elements.
Namded after Ytterby, a village in Sweden near Vauxholm. Yttria earth containing yttrium was discovered by Gadolin in 1794. Ytterby is the site of a quarry which yielded many unusual minerals containing rare earths and other elements. This small town, near Stockholm, bears the honor of giving names to erbium, terbium, and ytterbium as well as yttrium.
In 1843 Mosander showed that yttira could be resolved into the oxides (or earths) of three elements. The name yttria was reserved for the most basic one; the others were named erbia and terbia.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
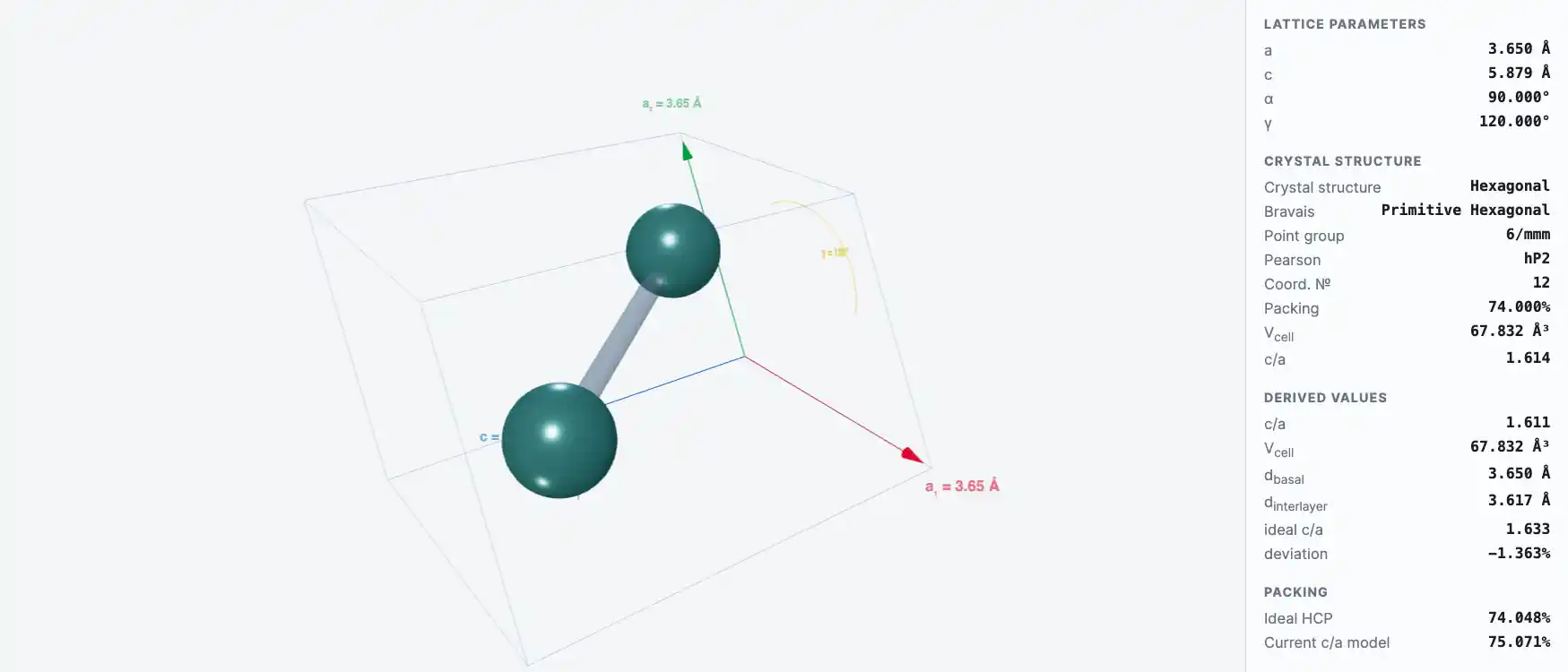
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Y: 4d¹ 5s²[Kr] 4d¹ 5s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹ 5s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 89 Stable | 88.9058403 ± 0.0000024 | 100.0000% | Stable |
Phase / State
Reason: 1496.8 °C below melting point (1521.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 39 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Y I | 0 | 361 | 189 | 351 |
| Y II | +1 | 116 | 66 | 116 |
| Y III | +2 | 113 | 0 | 0 |
| Y IV | +3 | 25 | 0 | 0 |
| Y V | +4 | 632 | 632 | 632 |
| Y VII | +6 | 168 | 168 | 168 |
| Y VIII | +7 | 70 | 70 | 70 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Y I | 0 | 194 |
| Y II | +1 | 249 |
| Y III | +2 | 51 |
| Y IV | +3 | 130 |
| Y V | +4 | 114 |
| Y VI | +5 | 2 |
| Y VII | +6 | 57 |
| Y VIII | +7 | 33 |
| Y IX | +8 | 2 |
| Y X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 90 pm |
| +3 | 7 | N/A | 96 pm |
| +3 | 8 | N/A | 101.89999999999999 pm |
| +3 | 9 | N/A | 107.5 pm |
Compounds
Isotopes (1)
Natural yttrium contains one isotope, 89Y. Nineteen other unstable isotopes have been characterized.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 89 Stable | 88.9058403 ± 0.0000024 | 100.0000% | Stable | stable |
Spectral Lines
Showing 50 of 266 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 410.23691 nm | 9900 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(1D).5p y 2F* | Measured | NIST | |
| 407.735998 nm | 9400 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(1D).5p y 2F* | Measured | NIST | |
| 412.829876 nm | 8900 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(1D).5p y 2D* | Measured | NIST | |
| 414.28358 nm | 7500 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(1D).5p y 2D* | Measured | NIST | |
| 404.76281 nm | 2400 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(3D).5p y 2P* | Measured | NIST | |
| 416.750671 nm | 2400 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(1D).5p y 2F* | Measured | NIST | |
| 423.5934 nm | 2200 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(1D).5p y 2D* | Measured | NIST | |
| 408.37033 nm | 2000 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(3D).5p y 2P* | Measured | NIST | |
| 417.41339 nm | 2000 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(3D).5p y 2P* | Measured | NIST | |
| 464.368813 nm | 2000 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(3D).5p z 2F* | Measured | NIST | |
| 467.48486 nm | 2000 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(3D).5p z 2F* | Measured | NIST | |
| 619.17183 nm | 1200 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(3D).5p z 2D* | Measured | NIST | |
| 643.50036 nm | 1000 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(3D).5p z 2D* | Measured | NIST | |
| 403.982219 nm | 940 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(1D).5p y 2D* | Measured | NIST | |
| 452.72342 nm | 890 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p y 4D* | Measured | NIST | |
| 483.9861 nm | 770 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p y 4F* | Measured | NIST | |
| 552.75472 nm | 740 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p z 4G* | Measured | NIST | |
| 546.6464 nm | 710 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p z 4G* | Measured | NIST | |
| 558.18694 nm | 620 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p z 4G* | Measured | NIST | |
| 563.01301 nm | 560 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p z 4G* | Measured | NIST | |
| 484.56655 nm | 550 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p y 4F* | Measured | NIST | |
| 450.59441 nm | 500 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p y 4D* | Measured | NIST | |
| 452.77815 nm | 440 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p y 4D* | Measured | NIST | |
| 476.09753 nm | 410 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(3D).5p z 2F* | Measured | NIST | |
| 485.26766 nm | 410 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p y 4F* | Measured | NIST | |
| 485.98428 nm | 330 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p y 4F* | Measured | NIST | |
| 425.11994 nm | 300 | Y I | emission | 4d.5s.(3D).5p z 4F* → 4d.5s.(3D).5d e 4G | Measured | NIST | |
| 448.74634 nm | 300 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p y 4D* | Measured | NIST | |
| 550.3466 nm | 300 | Y I | emission | 4d2.(3F).5s a 2F → 4d2.(3F).5p x 2F* | Measured | NIST | |
| 622.25784 nm | 300 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(3D).5p z 2D* | Measured | NIST | |
| 543.82242 nm | 190 | Y I | emission | 4d2.(3F).5s a 2F → 4d2.(3F).5p x 2D* | Measured | NIST | |
| 546.62434 nm | 190 | Y I | emission | 4d.5s.(3D).5p z 4F* → 4d.5s.(3D).6s e 4D | Measured | NIST | |
| 679.37029 nm | 190 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(3D).5p z 4F* | Measured | NIST | |
| 524.08001 nm | 181 | Y I | emission | 4d2.(1G).5s a 2G → 4d2.(1G).5p z 2H* | Measured | NIST | |
| 447.69471 nm | 180 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p x 2F* | Measured | NIST | |
| 469.67994 nm | 180 | Y I | emission | 4d2.(1D).5s b 2D → 4d2.(1D).5p w 2F* | Measured | NIST | |
| 479.92999 nm | 180 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p y 4F* | Measured | NIST | |
| 513.51993 nm | 180 | Y I | emission | 4d2.(1G).5s a 2G → 4d2.(1G).5p z 2H* | Measured | NIST | |
| 557.74153 nm | 180 | Y I | emission | 4d2.(3F).5s a 2F → 4d2.(3F).5p z 2G* | Measured | NIST | |
| 447.57178 nm | 170 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p y 4D* | Measured | NIST | |
| 472.8516 nm | 170 | Y I | emission | 5s2.5p z 2P* → 5s2.6s e 2S | Measured | NIST | |
| 478.68762 nm | 170 | Y I | emission | 4d2.(3P).5s a 4P → 4d2.(3P).5p x 4D* | Measured | NIST | |
| 421.77985 nm | 160 | Y I | emission | 5s2.5p z 2P* → 5s2.(2D).5d e 2D | Measured | NIST | |
| 447.74436 nm | 160 | Y I | emission | 4d2.(3F).5s a 4F → 4d2.(3F).5p y 4D* | Measured | NIST | |
| 475.2787 nm | 160 | Y I | emission | 4d2.(3F).5s a 2F → 4d2.(3P).5p x 4D* | Measured | NIST | |
| 570.67133 nm | 160 | Y I | emission | 4d.5s.(3D).5p z 4F* → 4d.5s.(3D).6s e 4D | Measured | NIST | |
| 492.18769 nm | 150 | Y I | emission | 5s2.5p z 2P* → 5s2.6s e 2S | Measured | NIST | |
| 613.84349 nm | 150 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(3D).5p z 4D* | Measured | NIST | |
| 668.75669 nm | 150 | Y I | emission | 4d.5s2 a 2D → 4d.5s.(3D).5p z 4F* | Measured | NIST | |
| 465.37837 nm | 140 | Y I | emission | 4d2.(1D).5s b 2D → 4d2.(3P).5p y 4P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1795.15 K |
| Boiling point | 3618.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (10)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.8244 |
| 2 | p | 3.9968 |
| 2 | s | 10.3778 |
| 3 | d | 13.6029 |
| 3 | p | 15.9075 |
| 3 | s | 15.4485 |
| 4 | d | 23.0416 |
| 4 | p | 26.2544 |
| 4 | s | 24.7364 |
| 5 | s | 32.744 |
Crystal Radii Detail (4)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 104 | from r^3 vs V plots, | |
| 3 | VII | 110 | ||
| 3 | VIII | 115.9 | from r^3 vs V plots, | |
| 3 | IX | 121.5 | from r^3 vs V plots, |
Isotope Decay Modes (60)
| Isotope | Mode | Intensity |
|---|---|---|
| 75 | B+ | — |
| 75 | B+p | — |
| 75 | p | — |
| 76 | B+ | — |
| 76 | p | — |
| 76 | B+p | — |
| 77 | B+ | 100% |
| 77 | B+p | — |
| 77 | p | — |
| 78 | B+ | 100% |
X‑ray Scattering Factors (619)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 2.26036 |
| 10.1617 | — | 2.25621 |
| 10.3261 | — | 2.25207 |
| 10.4931 | — | 2.24793 |
| 10.6628 | — | 2.2438 |
| 10.8353 | — | 2.23968 |
| 11.0105 | — | 2.23344 |
| 11.1886 | — | 2.21122 |
| 11.3696 | — | 2.18921 |
| 11.5535 | — | 2.16742 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
3.3×101 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.3×10-5 milligrams per liter
References (1)
Sources
Sources of this element.
Yttrium occurs in nearly all of the rare-earth minerals. Analysis of lunar rock samples obtained during the Apollo missions show a relatively high yttrium content.
It is recovered commercially from monazite sand, which contains about 3%, and from bastnasite, which contains about 0.2%. Wohler obtained the impure element in 1828 by reduction of the anhydrous chloride with potassium. The metal is now produced commercially by reduction of the fluoride with calcium metal. It can also be prepared by other techniques.
References (1)
- [6] Yttrium https://periodic.lanl.gov/39.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Yttrium.
The element property data was retrieved from publications.