Niobium (Nb)
transition-metalSolid
Standard Atomic Weight
92.90637 uElectron configuration
[Kr] 5s1 4d4Melting point
2476.85 °C (2750 K)Boiling point
4743.85 °C (5017 K)Density
8570 kg/m³Oxidation states
−3, −1, 0, +1, +2, +3, +4, +5Electronegativity (Pauling)
1.6Ionization energy (1st)
Discovery year
1801Atomic radius
145 pmDetails
Niobium is a refractory transition metal of group 5, chemically similar to tantalum and commonly occurring with it in oxide minerals. It is valued for its ability to strengthen steel at very small additions and for forming superconducting intermetallic compounds. In most compounds niobium is pentavalent, but lower oxidation states are well established, especially in halides and cluster chemistry.
Niobium is a shiny, white, soft, and ductile metal, and takes on a bluish cast when exposed to air at room temperatures for a long time. The metal starts to oxidize in air at 200°C, and when processed at even moderate temperatures must be placed in a protective atmosphere.
The name derives from the Greek mythological character Niobe, who was the daughter of Tantalus, because the elements niobium and tantalum were originally thought to be identical. Niobium was discovered in a black mineral from America called columbite by the British chemist and manufacturer Charles Hatchett in 1801 and he called the element columbium. In 1809, the English chemist William Hyde Wollaston claimed that columbium and tantalum were identical.
Forty years later, the German chemist and pharmacist, Heinrich Rose, determined that they were two different elements in 1846 and gave the name niobium because it was so difficult to distinguish it from tantalum. The name columbium continued to be used in America and niobium in Europe until IUPAC adopted the name niobium in 1949. Niobium was first isolated by the chemist C. W. Blomstrand in 1846.
The story of niobium's discovery is a bit confusing. The first governor of Connecticut, John Winthrop the Younger, discovered a new mineral around 1734. He named the mineral columbite ((Fe, Mn, Mg)(Nb, Ta)2O6) and sent a sample of it to the British Museum in London, England. The columbite sat in the museum's mineral collection for years until it was analyzed by Charles Hatchett in 1801. Hatchett could tell that there was an unknown element in the columbite, but he was not able to isolate it. He named the new element columbium. The fate of columbium took a drastic turn in 1809 when William Hyde Wollaston, an English chemist and physicist, compared the minerals columbite and tantalite ((Fe, Mn)(Ta, Nb)2O6) and declared that columbium was actually the element tantalum. This confusion arose because tantalum and niobium are similar metals, are always found together and are very difficult to isolate.
Niobium was rediscovered and renamed by Heinrich Rose in 1844 when he produced two new acids, niobic acid and pelopic acid, from samples of columbite and tantalite. These acids are very similar to each other and it took another twenty-two years and a Swiss chemist named Jean Charles Galissard de Marignac to prove that these were two distinct chemicals produced from two different elements. Metallic niobium was finally isolated by the Swedish chemist Christian Wilhelm Blomstrand in 1864. Today, niobium is primarily obtained from the minerals columbite and pyrochlore ((Ca, Na)2Nb2O6(O, OH, F)).
Named after Niobe, the daughter of Tantalu. Discovered in 1801 by Hatchett in an ore sent to England. The metal was first prepared in 1864 by Blomstrand, who reduced the chloride by heating it in a hydrogen atmosphere. The name niobium was adopted by the International Union of Pure and Applied Chemicstry (IUPAC) in 1950 after 100 years of controversy. Many leading chemical societies and government organizations refer to it by this name. Most metallurgists, leading metal societies, and all but one of the leading U.S. commercial producers, however, still refer to the metal as "columbium."
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
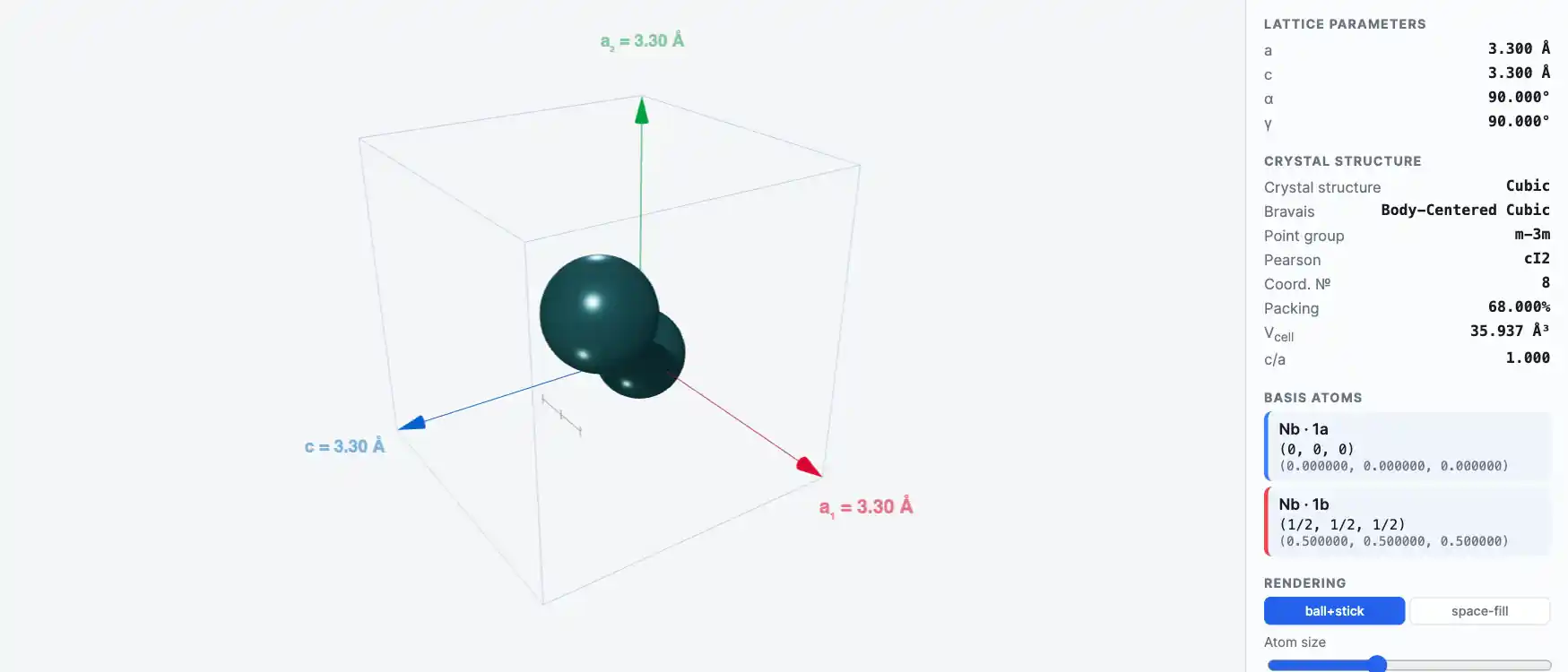
Crystal Structure
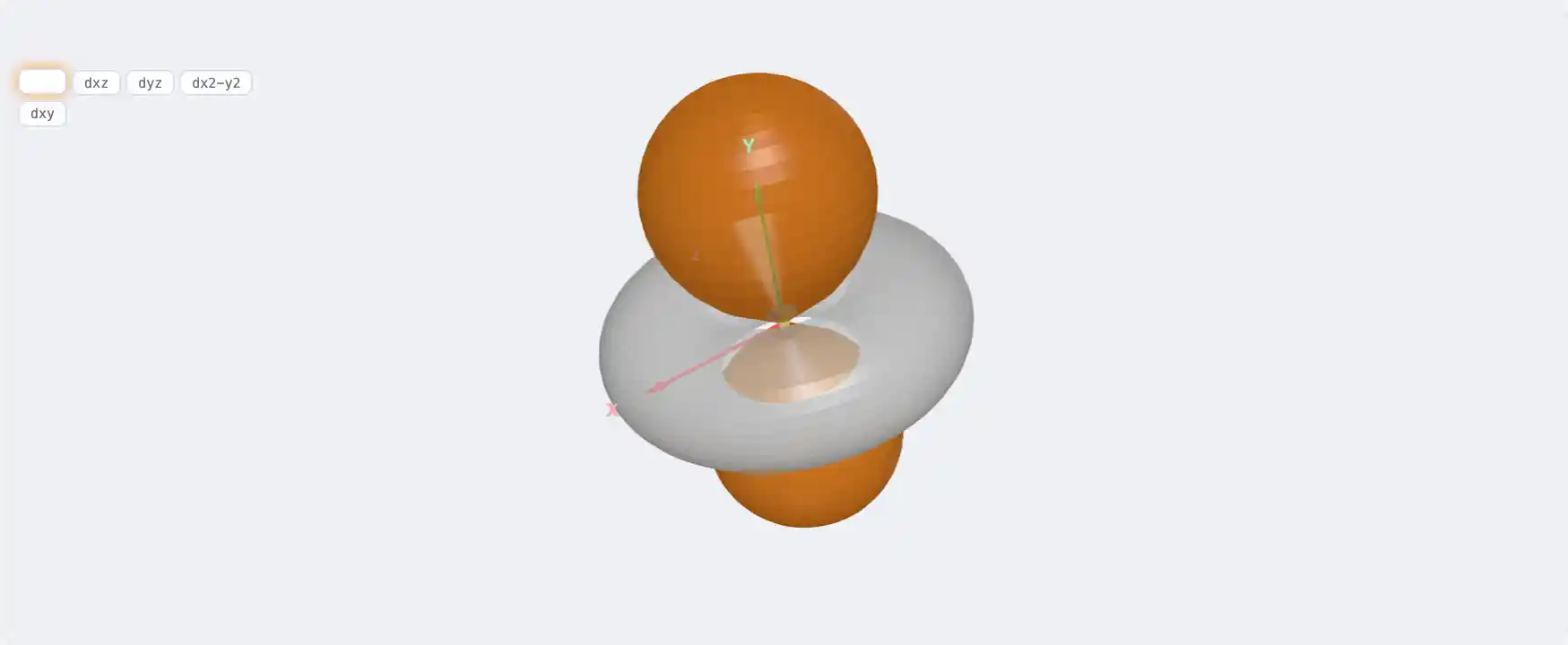
Electronic Structure
Identifiers
Electron Configuration Measured
Nb: 4d⁴ 5s¹[Kr] 4d⁴ 5s¹1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d⁴ 5s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 93 Stable | 92.906373 ± 0.000002 | 100.0000% | Stable |
Phase / State
Reason: 2451.8 °C below melting point (2476.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 41 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Nb I | 0 | 509 | 0 | 0 |
| Nb II | +1 | 150 | 0 | 0 |
| Nb III | +2 | 108 | 0 | 0 |
| Nb IV | +3 | 819 | 819 | 819 |
| Nb V | +4 | 12 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Nb I | 0 | 395 |
| Nb II | +1 | 354 |
| Nb III | +2 | 189 |
| Nb IV | +3 | 183 |
| Nb V | +4 | 31 |
| Nb VI | +5 | 105 |
| Nb VII | +6 | 32 |
| Nb VIII | +7 | 2 |
| Nb IX | +8 | 2 |
| Nb X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 72 pm |
| +4 | 6 | N/A | 68 pm |
| +4 | 8 | N/A | 79 pm |
| +5 | 4 | N/A | 48 pm |
| +5 | 6 | N/A | 64 pm |
| +5 | 7 | N/A | 69 pm |
| +5 | 8 | N/A | 74 pm |
Compounds
Isotopes (1)
Eighteen isotopes of niobium are known. The metal can be isolated from tantalum, and prepared in several ways.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 93 Stable | 92.906373 ± 0.000002 | 100.0000% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 382.5416 nm | 5000 | Nb IV | emission | 4d.5d 3P → 4d.6p 1P* | Measured | NIST | |
| 382.5694 nm | 200000 | Nb IV | emission | 4d.6p 1F* → 4d.6d 3D | Measured | NIST | |
| 382.5875 nm | 250000 | Nb IV | emission | 4d.6p 3P* → 4d.6d 3D | Measured | NIST | |
| 383.106 nm | 15000 | Nb IV | emission | 4d.5d 3D → 4d.6p 1D* | Measured | NIST | |
| 385.2874 nm | 60000 | Nb IV | emission | 4d.6p 3P* → 4d.6d 3D | Measured | NIST | |
| 385.5325 nm | 10000 | Nb IV | emission | 4d.6p 1P* → 4d.6d 3P | Measured | NIST | |
| 386.9546 nm | 8000 | Nb IV | emission | 4d.6p 3P* → 4d.6d 1P | Measured | NIST | |
| 387.5455 nm | 100000 | Nb IV | emission | 4d.5d 3G → 4d.6p 1D* | Measured | NIST | |
| 388.2203 nm | 60000 | Nb IV | emission | 4d.6p 1P* → 4d.6d 1S | Measured | NIST | |
| 389.8028 nm | 100000 | Nb IV | emission | 4d.5d 3S → 4d.6p 3P* | Measured | NIST | |
| 390.0115 nm | 25000 | Nb IV | emission | 4d.6p 3P* → 4d.6d 3D | Measured | NIST | |
| 391.6922 nm | 8000 | Nb IV | emission | 4d.5d 3F → 4d.6p 1F* | Measured | NIST | |
| 392.1878 nm | 5000 | Nb IV | emission | 4d.5d 1P → 4d.6p 3D* | Measured | NIST | |
| 394.057 nm | 25000 | Nb IV | emission | 4d.6p 3P* → 4d.6d 3D | Measured | NIST | |
| 394.3315 nm | 20000 | Nb IV | emission | 4d.5d 3F → 4d.6p 3F* | Measured | NIST | |
| 398.5759 nm | 5000 | Nb IV | emission | 4d.(2D<3/2>).6s 2[3/2] → 4d.6p 1P* | Measured | NIST | |
| 400.1839 nm | 4000 | Nb IV | emission | 4d.6p 3P* → 4d.6d 3D | Measured | NIST | |
| 403.2233 nm | 40000 | Nb IV | emission | 4d.5d 3F → 4d.6p 3D* | Measured | NIST | |
| 404.998 nm | 10000 | Nb IV | emission | 4d.5d 3F → 4d.6p 3F* | Measured | NIST | |
| 405.2616 nm | 15000 | Nb IV | emission | 4d.5d 1P → 4d.6p 1D* | Measured | NIST | |
| 406.3412 nm | 200000 | Nb IV | emission | 4d.5d 3F → 4d.6p 3F* | Measured | NIST | |
| 406.4694 nm | N/A | Nb IV | emission | 4d.6p 1P* → 4d.6d 1D | Measured | NIST | |
| 409.6529 nm | 7000 | Nb IV | emission | 4d.6p 3P* → 4d.6d 3D | Measured | NIST | |
| 459.6 nm | N/A | ID 841 | emission | 2p 2P* → 2s 2S | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 2750.15 K |
| Boiling point | 5014.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (10)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.8577 |
| 2 | p | 4.0178 |
| 2 | s | 10.8748 |
| 3 | d | 14.753 |
| 3 | p | 16.3844 |
| 3 | s | 15.8285 |
| 4 | d | 29.7624 |
| 4 | p | 26.9156 |
| 4 | s | 25.7172 |
| 5 | s | 35.079 |
Crystal Radii Detail (7)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 86 | ||
| 4 | VI | 82 | from r^3 vs V plots, estimated, | |
| 4 | VIII | 93 | ||
| 5 | IV | 62 | calculated, | |
| 5 | VI | 78 | ||
| 5 | VII | 83 | calculated, | |
| 5 | VIII | 88 |
Isotope Decay Modes (67)
| Isotope | Mode | Intensity |
|---|---|---|
| 79 | p | — |
| 79 | B+ | — |
| 79 | B+p | — |
| 80 | p | — |
| 80 | B+ | — |
| 80 | B+p | — |
| 81 | p | — |
| 81 | B+ | — |
| 81 | B+p | — |
| 82 | B+ | 100% |
X‑ray Scattering Factors (757)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 0.5 | — | 0.09113 |
| 0.5079 | — | 0.09258 |
| 0.516 | — | 0.09406 |
| 0.5242 | — | 0.09557 |
| 0.5325 | — | 0.0971 |
| 0.5409 | — | 0.09865 |
| 0.5495 | — | 0.10023 |
| 0.5582 | — | 0.10161 |
| 0.5671 | — | 0.103 |
| 0.5761 | — | 0.10441 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
2.0×101 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1×10-5 milligrams per liter
References (1)
Sources
Sources of this element.
The element is found in niobite (or columbite), niobite-tantalite, parochlore, and euxenite. Large deposits of niobium have been found associated with carbonatites (carbon-silicate rocks), as a constituent of parochlore. Extensive ore reserves are found in Canada, Brazil, Nigeria, Zaire, and in Russia.
References (1)
- [6] Niobium https://periodic.lanl.gov/41.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Niobium.
The element property data was retrieved from publications.