Strontium (Sr)
alkaline-earth-metalSolid
Standard Atomic Weight
87.62 uElectron configuration
[Kr] 5s2Melting point
776.85 °C (1050 K)Boiling point
1381.85 °C (1655 K)Density
2640 kg/m³Oxidation states
+1, +2Electronegativity (Pauling)
0.95Ionization energy (1st)
Discovery year
1792Atomic radius
200 pmDetails
Strontium is an alkaline earth metal below calcium and above barium in group 2. Natural strontium is stable and occurs mainly as the minerals celestine and strontianite rather than as the free metal. Its chemistry is dominated by the Sr²⁺ ion, which closely resembles Ca²⁺ but is larger and more readily forms insoluble sulfate and carbonate salts. Strontium is best known technologically for red pyrotechnic colors, ferrite magnets, glass additives, and the radioactive isotope ⁹⁰Sr.
Strontium is softer than calcium and decomposes in water more vigorously. It does not absorb nitrogen below 380°C. It should be kept under kerosene to prevent oxidation. Freshly cut strontium has a silvery appearance, but rapidly turns a yellowish color with the formation of the oxide. The finely divided metal ignites spontaneously in air. Volatile strontium salts impart a beautiful crimson color to flames, and these salts are used in pyrotechnics and in the production of flares. Natural strontium is a mixture of four stable isotopes.
The name derives from Strontian, a town in Scotland. The mineral strontianite is found in mines in Strontian. The element was discovered in 1792 by the Scottish chemist and physician Thomas Charles Hope, who observed the brilliant red flame colour of strontium. It was first isolated by the English chemist Humphry Davy in 1808.
Strontium was discovered by Adair Crawford, an Irish chemist, in 1790 while studying the mineral witherite (BaCO3). When he mixed witherite with hydrochloric acid (HCl) he did not get the results he expected. He assumed that his sample of witherite was contaminated with an unknown mineral, a mineral he named strontianite (SrCO3). Strontium was first isolated by Sir Humphry Davy, an English chemist, in 1808 through the electrolysis of a mixture of strontium chloride (SrCl2) and mercuric oxide (HgO). Today, strontium is obtained from two of its most common ores, celestite (SrSO4) and strontianite (SrCO3), by treating them with hydrochloric acid, forming strontium chloride. The strontium chloride, usually mixed with potassium chloride (KCl), is then melted and electrolyzed, forming strontium and chlorine gas (Cl2).
Named after Strontian, a town in Scotland. Isolated by Davey by electrolysis in 1808, however, Adair Crawford recognized a new mineral (strontianite) as differing from other barium minerals in 1790.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
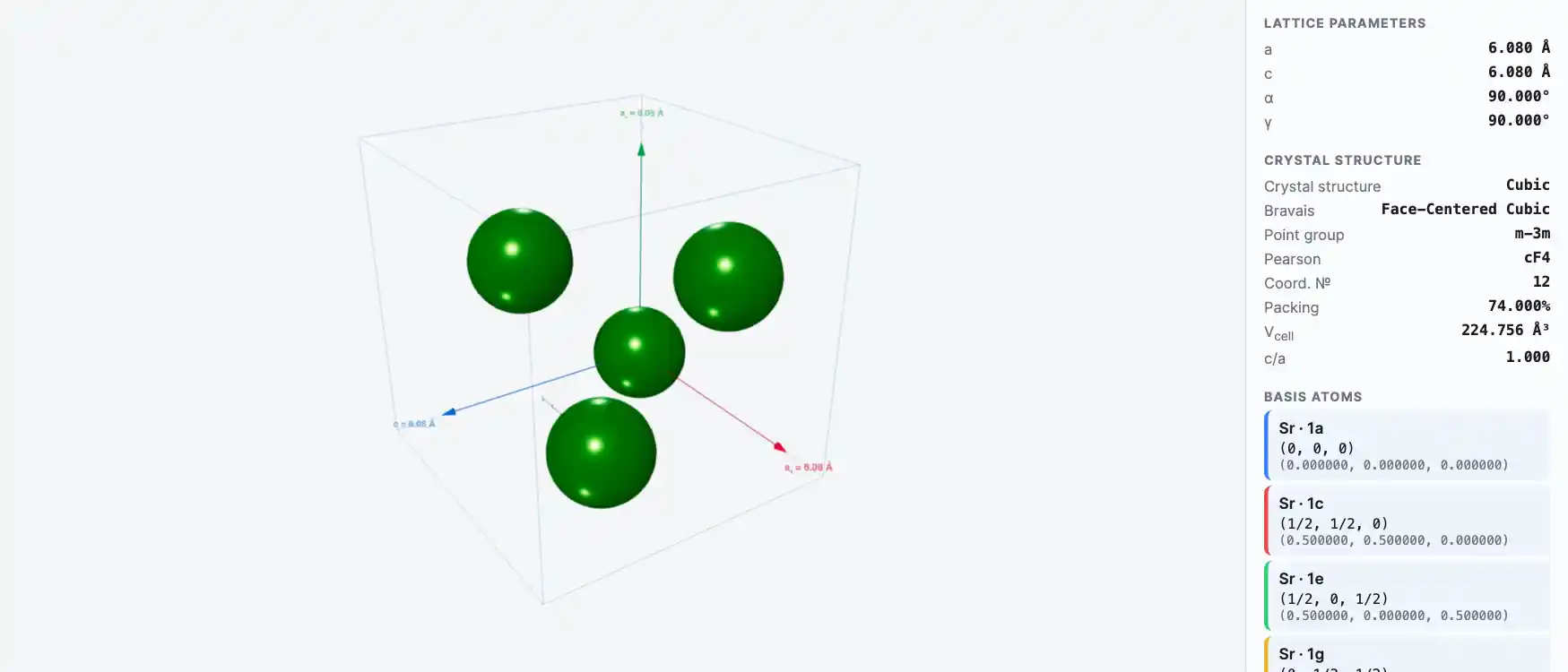
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Sr: 5s²[Kr] 5s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 5s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 84 Stable | 83.9134191 ± 0.0000013 | 0.5600% | Stable |
| 86 Stable | 85.9092606 ± 0.0000012 | 9.8600% | Stable |
| 87 Stable | 86.9088775 ± 0.0000012 | 7.0000% | Stable |
| 88 Stable | 87.9056125 ± 0.0000012 | 82.5800% | Stable |
Phase / State
Reason: 751.9 °C below melting point (776.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 38 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Sr I | 0 | 361 | 86 | 361 |
| Sr II | +1 | 135 | 33 | 135 |
| Sr III | +2 | 613 | 0 | 613 |
| Sr IV | +3 | 1183 | 0 | 1183 |
| Sr V | +4 | 625 | 0 | 625 |
| Sr VI | +5 | 57 | 14 | 57 |
| Sr VII | +6 | 30 | 30 | 30 |
| Sr VIII | +7 | 26 | 24 | 26 |
| Sr IX | +8 | 46 | 28 | 46 |
| Sr X | +9 | 54 | 51 | 54 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Sr I | 0 | 380 |
| Sr II | +1 | 72 |
| Sr III | +2 | 150 |
| Sr IV | +3 | 255 |
| Sr V | +4 | 144 |
| Sr VI | +5 | 22 |
| Sr VII | +6 | 20 |
| Sr VIII | +7 | 21 |
| Sr IX | +8 | 31 |
| Sr X | +9 | 47 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 6 | N/A | 118 pm |
| +2 | 7 | N/A | 121 pm |
| +2 | 8 | N/A | 126 pm |
| +2 | 9 | N/A | 131 pm |
| +2 | 10 | N/A | 136 pm |
| +2 | 12 | N/A | 144 pm |
Compounds
Isotopes (4)
Sixteen other unstable isotopes are known to exist. Of greatest importance is 90Sr with a half-life of 29 years. It is a product of nuclear fallout and presents a health problem. This isotope is one of the best long-lived high-energy beta emitters known, and is used in SNAP (Systems for Nuclear Auxilliary Power) devices. These devices hold promise for use in space vehicles, remote weather stations, navigational buoys, etc., and where a lightweight, long-lived, nuclear-electric power source is needed.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 84 Stable | 83.9134191 ± 0.0000013 | 0.5600% ± 0.0100% | Stable | stable | |
| 86 Stable | 85.9092606 ± 0.0000012 | 9.8600% ± 0.0100% | Stable | stable | |
| 87 Stable | 86.9088775 ± 0.0000012 | 7.0000% ± 0.0100% | Stable | stable | |
| 88 Stable | 87.9056125 ± 0.0000012 | 82.5800% ± 0.0100% | Stable | stable |
Spectral Lines
Showing 50 of 500 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 707.0072 nm | 14000 | Sr I | emission | 5s.5p 3P* → 5s.6s 3S | Measured | NIST | |
| 687.83128 nm | 12000 | Sr I | emission | 5s.5p 3P* → 5s.6s 3S | Measured | NIST | |
| 679.10198 nm | 7000 | Sr I | emission | 5s.5p 3P* → 5s.6s 3S | Measured | NIST | |
| 525.68986 nm | 3400 | Sr I | emission | 5s.4d 3D → 4d.5p 3P* | Measured | NIST | |
| 640.8463 nm | 3100 | Sr I | emission | 5s.4d 3D → 4d.5p 3F* | Measured | NIST | |
| 483.20425 nm | 2900 | Sr I | emission | 5s.5p 3P* → 5s.5d 3D | Measured | NIST | |
| 548.08638 nm | 2700 | Sr I | emission | 5s.4d 3D → 4d.5p 3D* | Measured | NIST | |
| 496.2263 nm | 2500 | Sr I | emission | 5s.5p 3P* → 5s.5d 3D | Measured | NIST | |
| 481.18799 nm | 2300 | Sr I | emission | 5s.5p 3P* → 5p2 3P | Measured | NIST | |
| 689.25894 nm | 2300 | Sr I | emission | 5s2 1S → 5s.5p 3P* | Measured | NIST | |
| 650.3992 nm | 2100 | Sr I | emission | 5s.4d 3D → 4d.5p 3F* | Measured | NIST | |
| 523.85479 nm | 2000 | Sr I | emission | 5s.4d 3D → 4d.5p 3P* | Measured | NIST | |
| 550.4181 nm | 2000 | Sr I | emission | 5s.4d 3D → 4d.5p 3D* | Measured | NIST | |
| 496.5585 nm | 1900 | Sr I | emission | 5s.5p 1P* → 5s.7d 1D | Measured | NIST | |
| 516.5486 nm | 1800 | Sr I | emission | 5s.5p 1P* → 5s.8s 1S | Measured | NIST | |
| 478.43198 nm | 1700 | Sr I | emission | 5s.5p 3P* → 5p2 3P | Measured | NIST | |
| 552.1768 nm | 1700 | Sr I | emission | 5s.4d 3D → 4d.5p 3D* | Measured | NIST | |
| 730.94166 nm | 1700 | Sr I | emission | 5s.4d 1D → 4d.5p 1D* | Measured | NIST | |
| 472.22769 nm | 1600 | Sr I | emission | 5s.5p 3P* → 5p2 3P | Measured | NIST | |
| 474.19221 nm | 1600 | Sr I | emission | 5s.5p 3P* → 5p2 3P | Measured | NIST | |
| 478.3782 nm | 1500 | Sr I | emission | 5s.5p 1P* → 5s.9s 1S | Measured | NIST | |
| 487.249 nm | 1500 | Sr I | emission | 5s.5p 3P* → 5s.5d 3D | Measured | NIST | |
| 489.198 nm | 1500 | Sr I | emission | 5s.4d 3D → 5s.4f 3F* | Measured | NIST | |
| 581.67702 nm | 1500 | Sr I | emission | 5s.4d 1D → 4d.5p 3P* | Measured | NIST | |
| 468.8546 nm | 1400 | Sr I | emission | 5s.5p 1P* → 5s.8d 1D | Measured | NIST | |
| 522.21992 nm | 1400 | Sr I | emission | 5s.4d 3D → 4d.5p 3P* | Measured | NIST | |
| 522.51079 nm | 1400 | Sr I | emission | 5s.4d 3D → 4d.5p 3P* | Measured | NIST | |
| 522.92697 nm | 1400 | Sr I | emission | 5s.4d 3D → 4d.5p 3P* | Measured | NIST | |
| 555.6375 nm | 1400 | Sr I | emission | 5s.5p 1P* → 5s.6d 3D | Measured | NIST | |
| 634.57265 nm | 1400 | Sr I | emission | 5s.4d 3D → 5s.6p 3P* | Measured | NIST | |
| 655.0244 nm | 1400 | Sr I | emission | 5s.5p 1P* → 4d2 1D | Measured | NIST | |
| 495.6274 nm | 1300 | Sr I | emission | 5s.5p 1P* → 5s.7d 3D | Measured | NIST | |
| 638.64581 nm | 1300 | Sr I | emission | 5s.4d 3D → 5s.6p 3P* | Measured | NIST | |
| 485.50448 nm | 1200 | Sr I | emission | 5s.4d 3D → 5s.4f 3F* | Measured | NIST | |
| 486.87005 nm | 1200 | Sr I | emission | 5s.4d 3D → 5s.4f 3F* | Measured | NIST | |
| 487.60745 nm | 1200 | Sr I | emission | 5s.5p 3P* → 5s.5d 3D | Measured | NIST | |
| 496.7942 nm | 1200 | Sr I | emission | 5s.5p 3P* → 5s.5d 3D | Measured | NIST | |
| 559.8159 nm | 1200 | Sr I | emission | 5s.4d 1D → 4d.5p 1F* | Measured | NIST | |
| 458.29879 nm | 1100 | Sr I | emission | 5s.5p 1P* → 5s.10s 1S | Measured | NIST | |
| 486.91724 nm | 1100 | Sr I | emission | 5s.4d 3D → 5s.4f 3F* | Measured | NIST | |
| 489.2642 nm | 1100 | Sr I | emission | 5s.4d 3D → 5s.4f 3F* | Measured | NIST | |
| 661.72651 nm | 1100 | Sr I | emission | 5s.4d 3D → 4d.5p 3F* | Measured | NIST | |
| 403.03772 nm | 1000 | Sr I | emission | 5s.5p 3P* → 5s.6d 3D | Measured | NIST | |
| 443.8043 nm | 1000 | Sr I | emission | 5s.5p 3P* → 5s.7s 3S | Measured | NIST | |
| 446.32981 nm | 1000 | Sr I | emission | 5s.5p 1P* → 5s.11s 1S | Measured | NIST | |
| 453.2375 nm | 1000 | Sr I | emission | 5s.5p 1P* → 5s.9d 1D | Measured | NIST | |
| 471.2151 nm | 1000 | Sr I | emission | 5s.4d 1D → 5s.5f 3F* | Measured | NIST | |
| 545.08373 nm | 1000 | Sr I | emission | 5s.4d 3D → 4d.5p 3D* | Measured | NIST | |
| 548.6135 nm | 1000 | Sr I | emission | 5s.4d 3D → 4d.5p 3D* | Measured | NIST | |
| 553.4799 nm | 1000 | Sr I | emission | 5s.4d 3D → 4d.5p 3D* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1050.15 K |
| Boiling point | 1650.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (9)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.8089 |
| 2 | p | 3.9696 |
| 2 | s | 10.0982 |
| 3 | d | 15.2738 |
| 3 | p | 15.8324 |
| 3 | s | 15.3362 |
| 4 | p | 26.068 |
| 4 | s | 24.5556 |
| 5 | s | 31.9295 |
Crystal Radii Detail (6)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | VI | 132 | ||
| 2 | VII | 135 | ||
| 2 | VIII | 140 | ||
| 2 | IX | 145 | ||
| 2 | X | 150 | calculated, | |
| 2 | XII | 158 | calculated, |
Isotope Decay Modes (54)
| Isotope | Mode | Intensity |
|---|---|---|
| 73 | B+ | 100% |
| 73 | B+p | 63% |
| 74 | B+ | 100% |
| 74 | B+p | — |
| 75 | B+ | 100% |
| 75 | B+p | 5.2% |
| 76 | B+ | 100% |
| 76 | B+p | 3.4% |
| 77 | B+ | 100% |
| 77 | B+p | 0.1% |
X‑ray Scattering Factors (508)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.17126 |
| 10.1617 | — | 0.1749 |
| 10.3261 | — | 0.17861 |
| 10.4931 | — | 0.1824 |
| 10.6628 | — | 0.18627 |
| 10.8353 | — | 0.19061 |
| 11.0106 | — | 0.19514 |
| 11.1886 | — | 0.19977 |
| 11.3696 | — | 0.2045 |
| 11.5535 | — | 0.20936 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
3.70×102 milligrams per kilogram
References (1)
- [5] Strontium https://education.jlab.org/itselemental/ele038.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
7.9 milligrams per liter
References (1)
- [5] Strontium https://education.jlab.org/itselemental/ele038.html
Isotopes in Forensic Science and Anthropology
Information on the use of this element's isotopes in forensic science and anthropology.
The isotope-amount ratio n(87Sr)/n(86Sr) is highly variable in rocks, minerals, soils, and waters, and it can be transmitted to plants (Fig. IUPAC.38.1), animals, and manufactured materials. Measurements of n(87Sr)/n(86Sr) ratios are used for forensic applications in food authentication (determining where food came from), archaeology, crime-scene investigation, and human migration [298] [298] B. L. Beard, C. M. Johnson. J. Forensic Sci.45, 1049 (2000).[298] B. L. Beard, C. M. Johnson. J. Forensic Sci.45, 1049 (2000)., [299] [299] K. M. Frei, R. Frei. Appl. Geochem.26, 326 (2011).[299] K. M. Frei, R. Frei. Appl. Geochem.26, 326 (2011)..
References (4)
- [298] B. L. Beard, C. M. Johnson. J. Forensic Sci.45, 1049 (2000).
- [299] K. M. Frei, R. Frei. Appl. Geochem.26, 326 (2011).
- [300] K. Miller, T. B. Coplen, M. Wieser. “Identification of the geographical origin of exotic wood species using 87Sr/86Sr isotope amount ratios”, in Goldschmidt 22nd Conference, Montreal, Quebec, Canada.
- [4] IUPAC Periodic Table of the Elements and Isotopes (IPTEI) https://doi.org/10.1515/pac-2015-0703
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Strontium.
The element property data was retrieved from publications.