Rubidium (Rb)
alkali-metalSolid
Standard Atomic Weight
85.4678 uElectron configuration
[Kr] 5s1Melting point
39.31 °C (312.46 K)Boiling point
687.85 °C (961 K)Density
1530 kg/m³Oxidation states
−1, +1Electronegativity (Pauling)
0.82Ionization energy (1st)
Discovery year
1861Atomic radius
235 pmDetails
Rubidium is a soft alkali metal of group 1, chemically close to potassium and cesium. Natural rubidium is a mixture dominated by stable ⁸⁵Rb with radioactive ⁸⁷Rb, whose very long half-life makes it important in geochronology. The element is not mined as a principal metal; it is obtained from minerals and brines where it substitutes for potassium. Its low ionization energy and convenient atomic transitions make rubidium useful in precision physics.
Rubidium can be liquid at room temperature. It is a soft, silvery-white metallic element of the alkali group and is the second most electropositive and alkaline element. It ignites spontaneously in air and reacts violently in water, setting fire to the liberated hydrogen. As with other alkali metals, it forms amalgams with mercury and it alloys with gold, cesium, sodium, and potassium. It colors a flame yellowish violet. Rubidium metal can be prepared by reducing rubidium chloride with calcium, and by a number of other methods. It must be kept under a dry mineral oil or in a vacuum or inert atmosphere.
The name derives from the Latin rubidus for "deepest red" because of the two deep red lines in its spectra. Rubidium was discovered in the mineral lepidolite by the German chemist Robert Wilhelm Bunsen and the German physicist Gustav-Robert Kirchoff in 1861. Bunsen isolated rubidium in 1863.
Rubidium was discovered by the German chemists Robert Bunsen and Gustav Kirchhoff in 1861 while analyzing samples of the mineral lepidolite (KLi2Al(Al, Si)3O10(F, OH)2) with a device called a spectroscope. The sample produced a set of deep red spectral lines they had never seen before. Bunsen was eventually able to isolate samples of rubidium metal. Today, most rubidium is obtained as a byproduct of refining lithium.
From the Latin word rubidus, deepest red. Discovered in 1861 by Bunsen and Kirchoff in the mineral lepidolite by use of the spectroscope.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
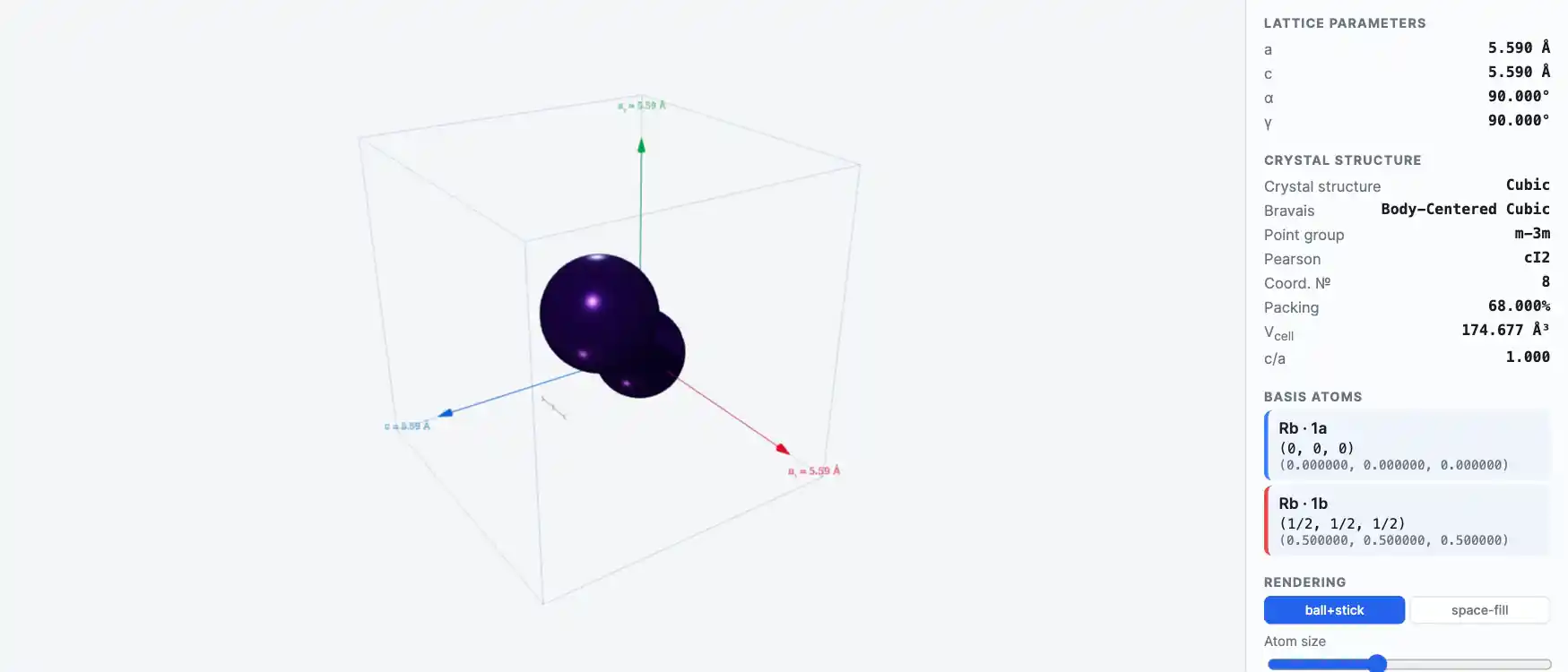
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Rb: 5s¹[Kr] 5s¹1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 5s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 85 Stable | 84.9117897379 ± 0.0000000054 | 72.1700% | Stable |
Phase / State
Reason: 14.3 °C below melting point (39.31 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 37 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Rb I | 0 | 213 | 40 | 213 |
| Rb II | +1 | 699 | 49 | 602 |
| Rb III | +2 | 232 | 0 | 230 |
| Rb IV | +3 | 573 | 0 | 573 |
| Rb V | +4 | 34 | 13 | 34 |
| Rb VI | +5 | 34 | 32 | 34 |
| Rb VII | +6 | 26 | 10 | 26 |
| Rb VIII | +7 | 34 | 26 | 34 |
| Rb IX | +8 | 40 | 17 | 40 |
| Rb X | +9 | 64 | 29 | 64 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Rb I | 0 | 240 |
| Rb II | +1 | 166 |
| Rb III | +2 | 92 |
| Rb IV | +3 | 131 |
| Rb V | +4 | 21 |
| Rb VI | +5 | 20 |
| Rb VII | +6 | 21 |
| Rb VIII | +7 | 25 |
| Rb IX | +8 | 37 |
| Rb X | +9 | 41 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +1 | 6 | N/A | 152 pm |
| +1 | 7 | N/A | 156 pm |
| +1 | 8 | N/A | 161 pm |
| +1 | 9 | N/A | 163 pm |
| +1 | 10 | N/A | 166 pm |
| +1 | 11 | N/A | 169 pm |
| +1 | 12 | N/A | 172 pm |
| +1 | 14 | N/A | 183 pm |
Compounds
Isotopes (1)
Twenty four isotopes of rubidium are known. Naturally occurring rubidium is made of two isotopes, 85Rb and 87Rb. Rubidium-87 is present to the extent of 27.85% in natural rubidium and is a beta emitter with a half-life of 4.9 x 1010 years. Ordinary rubidium is sufficiently radioactive to expose a photographic film in about 30 to 60 days. Rubidium forms four oxides: Rb2O, Rb2O2, Rb2O3, Rb2O4.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 85 Stable | 84.9117897379 ± 0.0000000054 | 72.1700% ± 0.0200% | Stable | stable |
Spectral Lines
Showing 50 of 202 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 424.439 nm | 90000 | Rb II | emission | 4p5.(2P*<3/2>).5s 2[3/2]* → 4p5.(2P*<3/2>).5p 2[5/2] | Measured | NIST | |
| 477.5954 nm | 30000 | Rb II | emission | 4p5.(2P*<3/2>).5s 2[3/2]* → 4p5.(2P*<3/2>).5p 2[1/2] | Measured | NIST | |
| 394.051 nm | 25000 | Rb II | emission | 4p5.(2P*<3/2>).5s 2[3/2]* → 4p5.(2P*<3/2>).5p 2[3/2] | Measured | NIST | |
| 457.1765 nm | 20000 | Rb II | emission | 4p5.(2P*<3/2>).5s 2[3/2]* → 4p5.(2P*<3/2>).5p 2[5/2] | Measured | NIST | |
| 427.3141 nm | 15000 | Rb II | emission | 4p5.(2P*<3/2>).5s 2[3/2]* → 4p5.(2P*<3/2>).5p 2[5/2] | Measured | NIST | |
| 464.8557 nm | 10000 | Rb II | emission | 4p5.4d 3P* → 4p5.(2P*<1/2>).5p 2[3/2] | Measured | NIST | |
| 515.2081 nm | 10000 | Rb II | emission | 4p5.(2P*<3/2>).5s 2[3/2]* → 4p5.(2P*<3/2>).5p 2[1/2] | Measured | NIST | |
| 645.833 nm | 10000 | Rb II | emission | 4p5.(2P*<1/2>).5s 2[1/2]* → 4p5.(2P*<3/2>).5p 2[1/2] | Measured | NIST | |
| 552.2776 nm | 5000 | Rb II | emission | 4p5.(2P*<1/2>).5s 2[1/2]* → 4p5.(2P*<3/2>).5p 2[3/2] | Measured | NIST | |
| 656.0799 nm | 5000 | Rb II | emission | 4p5.4d 3F* → 4p5.(2P*<1/2>).5p 2[3/2] | Measured | NIST | |
| 419.3079 nm | 3500 | Rb II | emission | 4p5.(2P*<3/2>).5s 2[3/2]* → 4p5.(2P*<3/2>).5p 2[3/2] | Measured | NIST | |
| 453.0333 nm | 3000 | Rb II | emission | 4p5.4d 3P* → 4p5.(2P*<1/2>).5p 2[1/2] | Measured | NIST | |
| 380.1896 nm | 2500 | Rb II | emission | 4p5.(2P*<1/2>).5s 2[1/2]* → 4p5.(2P*<1/2>).5p 2[1/2] | Measured | NIST | |
| 437.7123 nm | 2500 | Rb II | emission | 4p5.(2P*<3/2>).5p 2[5/2] → 4p5.(2P*<3/2>).6s 2[3/2]* | Measured | NIST | |
| 402.9485 nm | 1700 | Rb II | emission | 4p5.(2P*<3/2>).5s 2[3/2]* → 4p5.(2P*<3/2>).5p 2[3/2] | Measured | NIST | |
| 429.3971 nm | 1500 | Rb II | emission | 4p5.(2P*<3/2>).5s 2[3/2]* → 4p5.(2P*<3/2>).5p 2[3/2] | Measured | NIST | |
| 382.66591 nm | 1000 | Rb II | emission | 4p5.(2P*<3/2>).5p 2[3/2] → 4p5.(2P*<3/2>).5d 2[3/2]* | Measured | NIST | |
| 420.18053 nm | 1000 | Rb I | emission | 4p6.5s 2S → 4p6.6p 2P* | Measured | NIST | |
| 434.6961 nm | 1000 | Rb II | emission | 4p5.(2P*<3/2>).5p 2[5/2] → 4p5.(2P*<3/2>).6s 2[3/2]* | Measured | NIST | |
| 446.9475 nm | 1000 | Rb II | emission | 4p5.(2P*<1/2>).5p 2[3/2] → 4p5.(2P*<1/2>).6s 2[1/2]* | Measured | NIST | |
| 473.0454 nm | 1000 | Rb II | emission | 4p5.4d 3P* → 4p5.(2P*<1/2>).5p 2[1/2] | Measured | NIST | |
| 475.5304 nm | 1000 | Rb II | emission | 4p5.(2P*<3/2>).5p 2[3/2] → 4p5.(2P*<3/2>).6s 2[3/2]* | Measured | NIST | |
| 655.5619 nm | 1000 | Rb II | emission | 4p5.4d 3P* → 4p5.(2P*<3/2>).5p 2[3/2] | Measured | NIST | |
| 451.90262 nm | 700 | Rb II | emission | 4p5.4d 1P* → 4p5.(2P*<3/2>).4f 2[3/2] | Measured | NIST | |
| 392.22011 nm | 500 | Rb II | emission | 4p5.(2P*<3/2>).5p 2[3/2] → 4p5.(2P*<3/2>).5d 2[1/2]* | Measured | NIST | |
| 421.5539 nm | 500 | Rb I | emission | 4p6.5s 2S → 4p6.6p 2P* | Measured | NIST | |
| 426.6584 nm | 500 | Rb II | emission | 4p5.(2P*<3/2>).5p 2[5/2] → 4p5.(2P*<3/2>).6s 2[3/2]* | Measured | NIST | |
| 465.9284 nm | 500 | Rb II | emission | 4p5.(2P*<3/2>).5p 2[3/2] → 4p5.(2P*<3/2>).6s 2[3/2]* | Measured | NIST | |
| 551.2542 nm | 500 | Rb II | emission | 4p5.4d 3F* → 4p5.(2P*<1/2>).5p 2[3/2] | Measured | NIST | |
| 386.07454 nm | 450 | Rb II | emission | 4p5.(2P*<3/2>).5p 2[1/2] → 4p5.(2P*<3/2>).6s 2[3/2]* | Measured | NIST | |
| 454.0732 nm | 400 | Rb II | emission | 4p5.(2P*<3/2>).5p 2[3/2] → 4p5.(2P*<3/2>).6s 2[3/2]* | Measured | NIST | |
| 444.00924 nm | 300 | Rb II | emission | 4p5.4d 1P* → 4p5.(2P*<3/2>).4f 2[5/2] | Measured | NIST | |
| 516.4575 nm | 300 | Rb II | emission | 4p5.(2P*<1/2>).5s 2[1/2]* → 4p5.(2P*<3/2>).5p 2[3/2] | Measured | NIST | |
| 626.94 nm | 300 | Rb II | emission | 4p5.(2P*<3/2>).4f 2[9/2] → 4p5.(2P*<3/2>).6g 2[11/2]* | Measured | NIST | |
| 390.7292 nm | 250 | Rb II | emission | 4p5.(2P*<3/2>).5p 2[3/2] → 4p5.(2P*<3/2>).5d 2[1/2]* | Measured | NIST | |
| 542.244 nm | 250 | Rb II | emission | 4p5.4d 1P* → 4p5.(2P*<3/2>).6p 2[3/2] | Measured | NIST | |
| 527.0514 nm | 200 | Rb II | emission | 4p5.(2P*<3/2>).5p 2[1/2] → 4p5.(2P*<3/2>).6s 2[3/2]* | Measured | NIST | |
| 573.9645 nm | 200 | Rb II | emission | 4p5.(2P*<3/2>).5d 2[7/2]* → 4p5.(2P*<3/2>).5f 2[9/2] | Measured | NIST | |
| 613.5268 nm | 200 | Rb II | emission | 4p5.(2P*<3/2>).5d 2[5/2]* → 4p5.(2P*<3/2>).5f 2[7/2] | Measured | NIST | |
| 383.78512 nm | 175 | Rb II | emission | 4p5.(2P*<3/2>).5p 2[3/2] → 4p5.(2P*<3/2>).5d 2[1/2]* | Measured | NIST | |
| 740.8171 nm | 150 | Rb I | emission | 4p6.5p 2P* → 4p6.7s 2S | Measured | NIST | |
| 550.0635 nm | 100 | Rb II | emission | 4p5.(2P*<3/2>).6p 2[5/2] → 4p5.(2P*<3/2>).7d 2[7/2]* | Measured | NIST | |
| 627.5697 nm | 100 | Rb II | emission | 4p5.(2P*<3/2>).4f 2[9/2] → 4p5.(2P*<3/2>).6g 2[11/2]* | Measured | NIST | |
| 451.9884 nm | 75 | Rb II | emission | 4p5.(2P*<3/2>).5d 2[5/2]* → 4p5.(2P*<3/2>).6f 2[7/2] | Measured | NIST | |
| 459.989 nm | 75 | Rb II | emission | 4p5.(2P*<3/2>).5d 2[5/2]* → 4p5.(2P*<3/2>).6f 2[7/2] | Measured | NIST | |
| 543.1528 nm | 75 | Rb I | emission | 4p6.5p 2P* → 4p6.8d 2D | Measured | NIST | |
| 589.308 nm | 75 | Rb II | emission | 4p5.(2P*<3/2>).5d 2[7/2]* → 4p5.(2P*<3/2>).5f 2[9/2] | Measured | NIST | |
| 607.0751 nm | 75 | Rb I | emission | 4p6.5p 2P* → 4p6.8s 2S | Measured | NIST | |
| 614.0319 nm | 75 | Rb II | emission | 4p5.4d 3F* → 4p5.(2P*<1/2>).5p 2[3/2] | Measured | NIST | |
| 572.4125 nm | 60 | Rb I | emission | 4p6.5p 2P* → 4p6.7d 2D | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Phase Transitions & Allotropes
| Melting point | 312.45 K |
| Boiling point | 961.15 K |
| Critical point (temperature) | 2093.15 K |
| Critical point (pressure) | 16 MPa |
| Triple point (temperature) | 312.41 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (9)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.7922 |
| 2 | p | 3.9612 |
| 2 | s | 9.8432 |
| 3 | d | 15.3208 |
| 3 | p | 15.6967 |
| 3 | s | 15.1573 |
| 4 | p | 26.1192 |
| 4 | s | 24.612 |
| 5 | s | 32.0155 |
Crystal Radii Detail (8)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 1 | VI | 166 | ||
| 1 | VII | 170 | ||
| 1 | VIII | 175 | ||
| 1 | IX | 177 | estimated, | |
| 1 | X | 180 | ||
| 1 | XI | 183 | ||
| 1 | XII | 186 | ||
| 1 | XIV | 197 |
Isotope Decay Modes (61)
| Isotope | Mode | Intensity |
|---|---|---|
| 71 | p | — |
| 72 | p | — |
| 73 | B+ | — |
| 73 | p | 100% |
| 74 | B+ | 100% |
| 74 | B+p | — |
| 75 | B+ | 100% |
| 76 | B+ | 100% |
| 76 | B+A | 3.8% |
| 77 | B+ | 100% |
X‑ray Scattering Factors (508)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.06968 |
| 10.1617 | — | 0.07104 |
| 10.3261 | — | 0.07253 |
| 10.4931 | — | 0.07441 |
| 10.6628 | — | 0.07635 |
| 10.8353 | — | 0.07833 |
| 11.0106 | — | 0.08037 |
| 11.1886 | — | 0.083 |
| 11.3696 | — | 0.08599 |
| 11.5535 | — | 0.0891 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
9.0×101 milligrams per kilogram
References (1)
- [5] Rubidium https://education.jlab.org/itselemental/ele037.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.2×10-1 milligrams per liter
References (1)
- [5] Rubidium https://education.jlab.org/itselemental/ele037.html
Sources
Sources of this element.
The element is much more abundant than was thought several years ago. It is now considered to be the 16th most abundant element in the earth's crust. Rubidium occurs in pollucite, leucite, and zinnwaldite, which contains traces up to 1%, in the form of the oxide. It is found in lepidolite to the extent of about 1.5%, and is recovered commercially from this source. Potassium minerals, such as those found at Searles Lake, California, and potassium chloride recovered from the brines in Michigan also contain the element and are commercial sources. It is also found along with cesium in the extensive deposits of pollucite at Bernic Lake, Manitoba.
References (1)
- [6] Rubidium https://periodic.lanl.gov/37.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Rubidium.
The element property data was retrieved from publications.