Arsenic (As)
metalloidSolid
Standard Atomic Weight
74.921595 uElectron configuration
[Ar] 4s2 3d10 4p3Melting point
816.85 °C (1090 K)Boiling point
613.85 °C (887 K)Density
5776 kg/m³Oxidation states
−3, −2, −1, 0, +1, +2, +3, +4, +5Electronegativity (Pauling)
2.18Ionization energy (1st)
Discovery year
1250Atomic radius
115 pmDetails
Arsenic is a metalloid in group 15, chemically related to phosphorus and antimony. It occurs mainly in sulfide minerals and in arsenide or sulfosalt ores, rather than as the native element. Its chemistry is dominated by the +3 and +5 oxidation states, with important differences in mobility and toxicity among species. Arsenic is technologically useful in small quantities, especially in compound semiconductors, but it is better known for the toxicity of many of its inorganic compounds.
The element is a steel gray, very brittle, crystalline, semimetallic solid; it tarnishes in air, and when it is heated it rapidly oxidizes to arsenous oxide, which smells of garlic. Arsenic and its compounds are poisonous.
The name derives from the Latin arsenicium and the Greek arsenikos for "masculine" or "male" because the ancients thought that metals were different sexes. Arsenic was known in prehistoric times for its poisonous sulfides. The German scientist and philosopher, Albert von Bollstadt (Albert the Great or Albertus Magnus) is thought to have obtained the metal around 1250.
Although arsenic compounds were mined by the early Chinese, Greek and Egyptian civilizations, it is believed that arsenic itself was first identified by Albertus Magnus, a German alchemist, in 1250. Arsenic occurs free in nature, but is most often found in the minerals arsenopyrite (FeAsS), realgar (AsS) and orpiment (As2S3). Today, most commercial arsenic is obtained by heating arsenopyrite.
From the Latin word arsenicum, Greek arsenikon. Elemental arsenic occurs in two solid modifications: yellow, and gray or metallic, with specific gravities of 1.97, and 5.73, respectively. It is believed that Albertus Magnus obtained the element in 1250 A.D. In 1649 Schroeder published two methods of preparing the element. Mispickel arsenopyrite, (FeSAs), is the most common mineral from which, on heating, the arsenic sublimes leaving ferrous sulfide.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
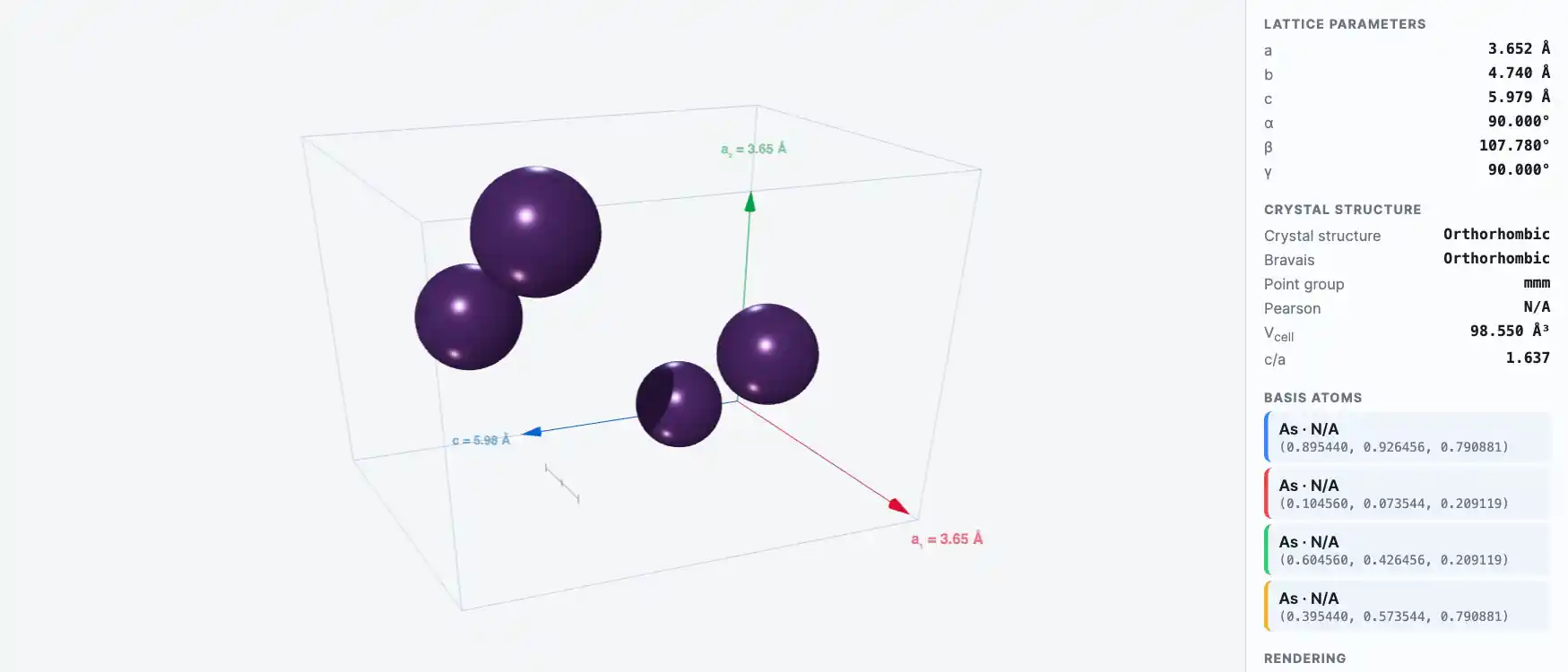
Crystal Structure
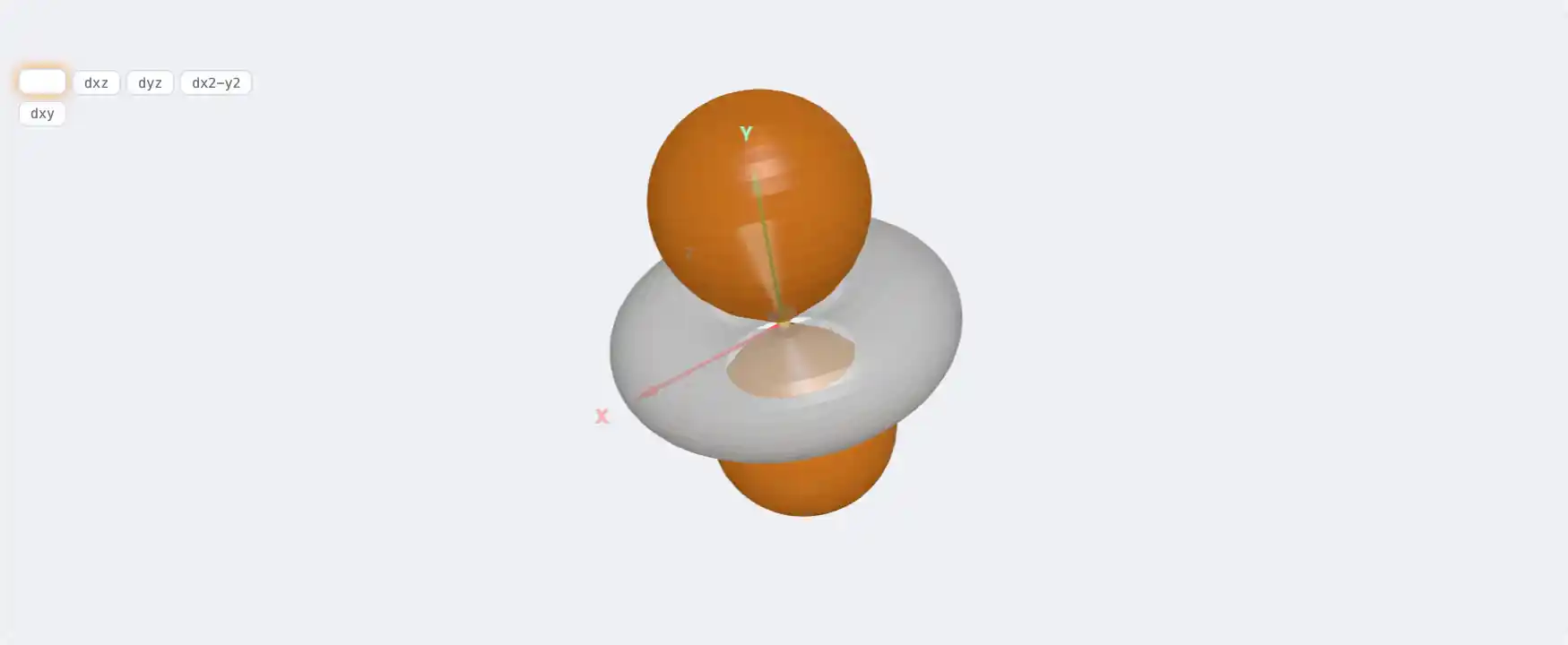
Electronic Structure
Identifiers
Electron Configuration Measured
As: 3d¹⁰ 4s² 4p³[Ar] 3d¹⁰ 4s² 4p³1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p³Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 75 Stable | 74.92159457 ± 0.00000095 | 100.0000% | Stable |
Phase / State
Reason: 588.9 °C below sublimation point (613.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 33 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| As I | 0 | 52 | 14 | 51 |
| As II | +1 | 86 | 0 | 0 |
| As III | +2 | 14 | 0 | 0 |
| As IV | +3 | 8 | 0 | 0 |
| As V | +4 | 9 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| As I | 0 | 116 |
| As II | +1 | 167 |
| As III | +2 | 22 |
| As IV | +3 | 34 |
| As V | +4 | 9 |
| As VI | +5 | 44 |
| As VII | +6 | 50 |
| As VIII | +7 | 2 |
| As IX | +8 | 2 |
| As X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 57.99999999999999 pm |
| +5 | 4 | N/A | 33.5 pm |
| +5 | 6 | N/A | 46 pm |
Compounds
Isotopes (1)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 75 Stable | 74.92159457 ± 0.00000095 | 100.0000% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1090.15 K |
| Boiling point | 889.15 K |
| Critical point (temperature) | 1673.15 K |
| Critical point (pressure) | 22.3 MPa |
| Triple point (temperature) | 1090.15 K |
| Triple point (pressure) | 3700 kPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (8)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.7217 |
| 2 | p | 3.9264 |
| 2 | s | 8.873 |
| 3 | d | 15.6216 |
| 3 | p | 15.1503 |
| 3 | s | 14.4045 |
| 4 | p | 25.5508 |
| 4 | s | 24.056 |
Crystal Radii Detail (3)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 72 | Ahrens (1952) ionic radius, | |
| 5 | IV | 47.5 | from r^3 vs V plots, | |
| 5 | VI | 60 | calculated, |
Isotope Decay Modes (50)
| Isotope | Mode | Intensity |
|---|---|---|
| 60 | p | — |
| 61 | p | — |
| 62 | p | — |
| 63 | p | — |
| 64 | B+ | 100% |
| 64 | B+p | — |
| 65 | B+ | 100% |
| 65 | B+p | — |
| 66 | B+ | 100% |
| 67 | B+ | 100% |
X‑ray Scattering Factors (506)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 4.62596 |
| 10.1617 | — | 4.67742 |
| 10.3261 | — | 4.72945 |
| 10.4931 | — | 4.78206 |
| 10.6628 | — | 4.83525 |
| 10.8353 | — | 4.88904 |
| 11.0106 | — | 4.94342 |
| 11.1886 | — | 4.99841 |
| 11.3696 | — | 5.05401 |
| 11.5535 | — | 5.11023 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.8 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
3.7-3 milligrams per liter
References (1)
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Arsenic.
The element property data was retrieved from publications.