Selenium (Se)
nonmetalSolid
Standard Atomic Weight
78.971 uElectron configuration
[Ar] 4s2 3d10 4p4Melting point
220.5 °C (493.65 K)Boiling point
684.85 °C (958 K)Density
4809 kg/m³Oxidation states
−2, −1, 0, +1, +2, +3, +4, +5, +6Electronegativity (Pauling)
2.55Ionization energy (1st)
Discovery year
1817Atomic radius
115 pmDetails
Selenium is a chalcogen between sulfur and tellurium, with chemistry that commonly parallels both. It is a trace element required by many organisms because it is incorporated into selenoproteins, yet the margin between nutritional requirement and toxicity is narrow. Industrially, selenium is valued for its semiconductor and photoconductive behavior, its red color in glass, and its role in metallurgy and specialty chemicals.
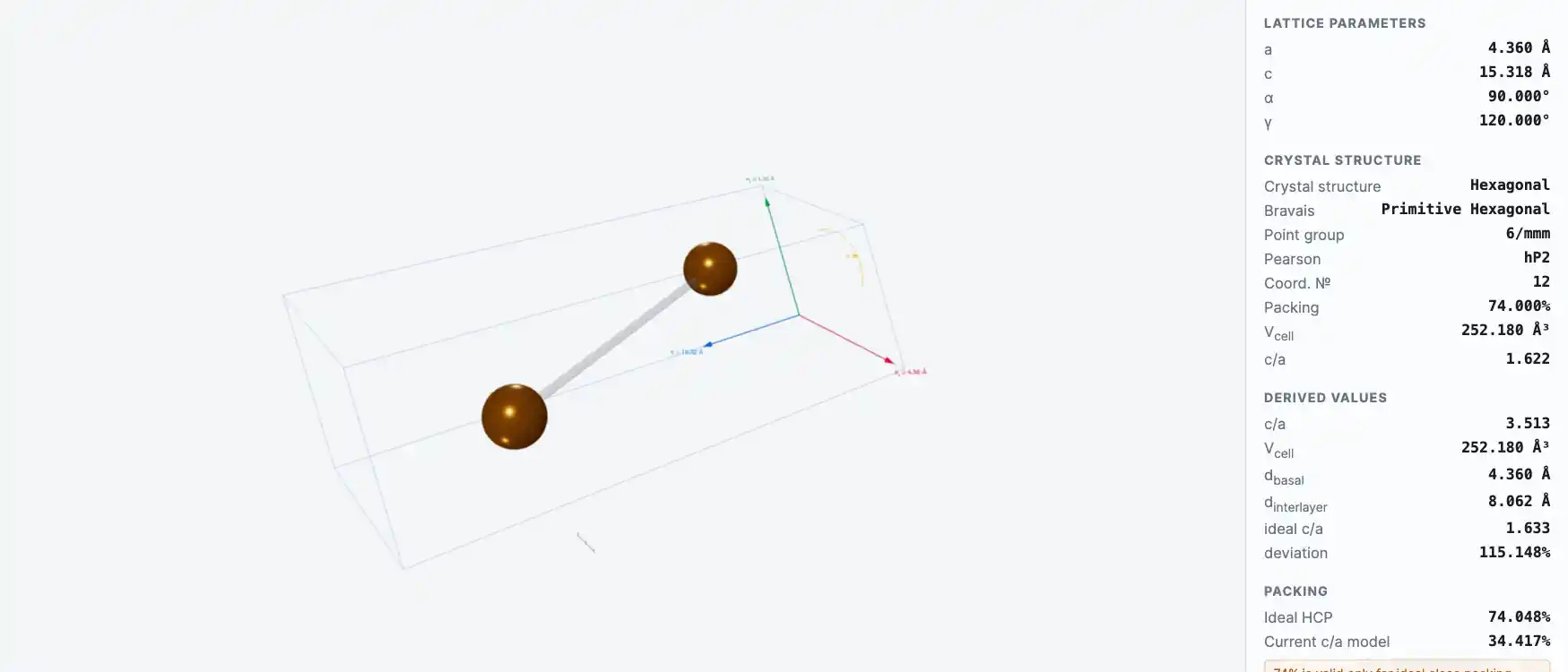
Selenium exists in several allotropic forms, although three are generally recognized. Selenium can be prepared with either an amorphous or a crystalline structure. The color of amorphous selenium is either red (in powder form) or black (in vitreous form). Crystalline monoclinic selenium is a deep red; crystalline hexagonal selenium, the most stable variety, is a metallic gray.
Selenium exhibits both photovoltaic action, where light is converted directly into electricity, and photoconductive action, where the electrical resistance decreases with increased illumination. These properties make selenium useful in the production of photocells and exposure meters for photographic use, as well as solar cells. Selenium is also able to convert a.c. electricity to d.c., and is extensively used in rectifiers. Below its melting point, selenium is a p-type semiconductor and has many uses in electronic and solid-state applications.
Elemental selenium has been said to be practically nontoxic and is considered to be an essential trace element; however, hydrogen selenide and other selenium compounds are extremely toxic, and resemble arsenic in their physiological reactions.
The name derives from the Greek Selene, who was the Greek goddess of the Moon because the element is chemically found with tellurium (Tellus was the Roman goddess of the Earth). Selenium was discovered by the Swedish chemist Jöns Jacob Berzelius in 1817, while trying to isolate tellurium in an impure sample.
Selenium was discovered by Jöns Jacob Berzelius, a Swedish chemist, in 1817 after analyzing an impurity that was contaminating the sulfuric acid (H2SO4) being produced at a particular factory in Sweden. Originally believing the material was tellurium, Berzelius eventually realized that it was actually a previously unknown element. Selenium occurs in minerals such as eucairite (CuAgSe), crooksite (CuThSe) and clausthalite (PbSe), but these minerals are too rare to use as a major source of selenium. Today, most selenium is obtained as a byproduct of refining copper.
From the Greek word Selene, moon. Discovered by Berzelius in 1817, who found it associated with tellurium (named for the earth).
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Se: 3d¹⁰ 4s² 4p⁴[Ar] 3d¹⁰ 4s² 4p⁴1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁴Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 76 Stable | 75.919213704 ± 0.000000017 | 9.3700% | Stable |
| 77 Stable | 76.919914154 ± 0.000000067 | 7.6300% | Stable |
| 78 Stable | 77.91730928 ± 0.0000002 | 23.7700% | Stable |
| 80 Stable | 79.9165218 ± 0.0000013 | 49.6100% | Stable |
Phase / State
Reason: 195.5 °C below melting point (220.5 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 34 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Se I | 0 | 138 | 0 | 0 |
| Se II | +1 | 39 | 0 | 0 |
| Se III | +2 | 39 | 0 | 0 |
| Se IV | +3 | 22 | 0 | 0 |
| Se V | +4 | 17 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Se I | 0 | 173 |
| Se II | +1 | 78 |
| Se III | +2 | 53 |
| Se IV | +3 | 29 |
| Se V | +4 | 15 |
| Se VI | +5 | 7 |
| Se VII | +6 | 45 |
| Se VIII | +7 | 37 |
| Se IX | +8 | 2 |
| Se X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| -2 | 6 | N/A | 198 pm |
| +4 | 6 | N/A | 50 pm |
| +6 | 4 | N/A | 28.000000000000004 pm |
| +6 | 6 | N/A | 42 pm |
Compounds
Isotopes (4)
Naturally selenium contains six stable isotopes. Fifteen other isotopes have been characterized. The element is a member of the sulfur family and resembles sulfur both in its various forms and in its compounds.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 76 Stable | 75.919213704 ± 0.000000017 | 9.3700% ± 0.2900% | Stable | stable | |
| 77 Stable | 76.919914154 ± 0.000000067 | 7.6300% ± 0.1600% | Stable | stable | |
| 78 Stable | 77.91730928 ± 0.0000002 | 23.7700% ± 0.2800% | Stable | stable | |
| 80 Stable | 79.9165218 ± 0.0000013 | 49.6100% ± 0.4100% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Supply Risk & Economics
Phase Transitions & Allotropes
| Transition temperature | 453.15 K |
| Boiling point | 958.15 K |
| Critical point (temperature) | 1766.15 K |
| Melting point | 493.95 K |
| Boiling point | 958.15 K |
| Critical point (temperature) | 1766.15 K |
| Critical point (pressure) | 27.2 MPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (8)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.7378 |
| 2 | p | 3.9348 |
| 2 | s | 9.1116 |
| 3 | d | 15.523 |
| 3 | p | 15.295 |
| 3 | s | 14.5966 |
| 4 | p | 25.7128 |
| 4 | s | 24.2424 |
Crystal Radii Detail (4)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| -2 | VI | 184 | Pauling's (1960) crystal radius, | |
| 4 | VI | 64 | Ahrens (1952) ionic radius, | |
| 6 | IV | 42 | ||
| 6 | VI | 56 | calculated, |
Isotope Decay Modes (52)
| Isotope | Mode | Intensity |
|---|---|---|
| 63 | B+ | 100% |
| 63 | B+p | 89% |
| 63 | 2p | 0.5% |
| 64 | B+ | — |
| 64 | B+p | — |
| 65 | B+ | 100% |
| 65 | B+p | 87% |
| 66 | B+ | 100% |
| 66 | B+p | — |
| 67 | B+ | 100% |
X‑ray Scattering Factors (506)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 5.20241 |
| 10.1617 | — | 5.36005 |
| 10.3261 | — | 5.52247 |
| 10.4931 | — | 5.63017 |
| 10.6628 | — | 5.66221 |
| 10.8353 | — | 5.69443 |
| 11.0106 | — | 5.71762 |
| 11.1886 | — | 5.72709 |
| 11.3696 | — | 5.73659 |
| 11.5535 | — | 5.7461 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
5×10-2 milligrams per kilogram
References (1)
- [5] Selenium https://education.jlab.org/itselemental/ele034.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
2×10-4 milligrams per liter
References (1)
- [5] Selenium https://education.jlab.org/itselemental/ele034.html
Production
Production of this element (from raw materials or other compounds containing the element).
Selenium is found in a few rare minerals such as crooksite and clausthalite. In years past it has been obtained from flue dusts remaining from processing copper sulfide ores, but the anode metal from electrolytic copper refineries now provide the source of most of the world's selenium. Selenium is recovered by roasting the mud with soda or sulfuric acid, or by smelting them with soda and niter.
References (1)
- [6] Selenium https://periodic.lanl.gov/34.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Selenium.
The element property data was retrieved from publications.