Phosphorus (P)
nonmetalSolid
Standard Atomic Weight
30.973762 uElectron configuration
[Ne] 3s2 3p3Melting point
44.15 °C (317.3 K)Boiling point
280.5 °C (553.65 K)Density
1820 kg/m³Oxidation states
−3, −2, −1, 0, +1, +2, +3, +4, +5Electronegativity (Pauling)
2.19Ionization energy (1st)
Discovery year
1669Atomic radius
100 pmDetails
Phosphorus is a reactive nonmetal in group 15 and is essential to life as a component of nucleic acids, phospholipids, and energy-transfer molecules. It does not occur naturally as the free element because it is readily oxidized, but it is abundant in phosphate minerals. Elemental phosphorus is notable for its several allotropes, especially highly reactive white phosphorus and more stable red and black forms.
Phosphorus exists in four or more allotropic forms: white (or yellow), red, and black (or violet). Ordinary phosphorus is a waxy white solid; when pure it is colorless and transparent. White phosphorus has two modifications: alpha and beta with a transition temperature at -3.8°C.
It is insoluble in water, but soluble in carbon disulfide. It takes fire spontaneously in air, burning to the pentoxide.
The name derives from the Greek phosphoros for "bringing light" because it has the property of glowing in the dark. This was also the ancient name for the planet Venus, when it appears before sunrise. Phosphorus was discovered by the German merchant Hennig Brand in 1669.
In what is perhaps the most disgusting method of discovering an element, phosphorus was first isolated in 1669 by Hennig Brand, a German physician and alchemist, by boiling, filtering and otherwise processing as many as 60 buckets of urine. Thankfully, phosphorus is now primarily obtained from phosphate rock (Ca3(PO4)2).
From the Greek phosphoros, light bearing; ancient name for the planet Venus when appearing before sunrise. Brand discovered phosphorus in 1669 by preparing it from urine.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
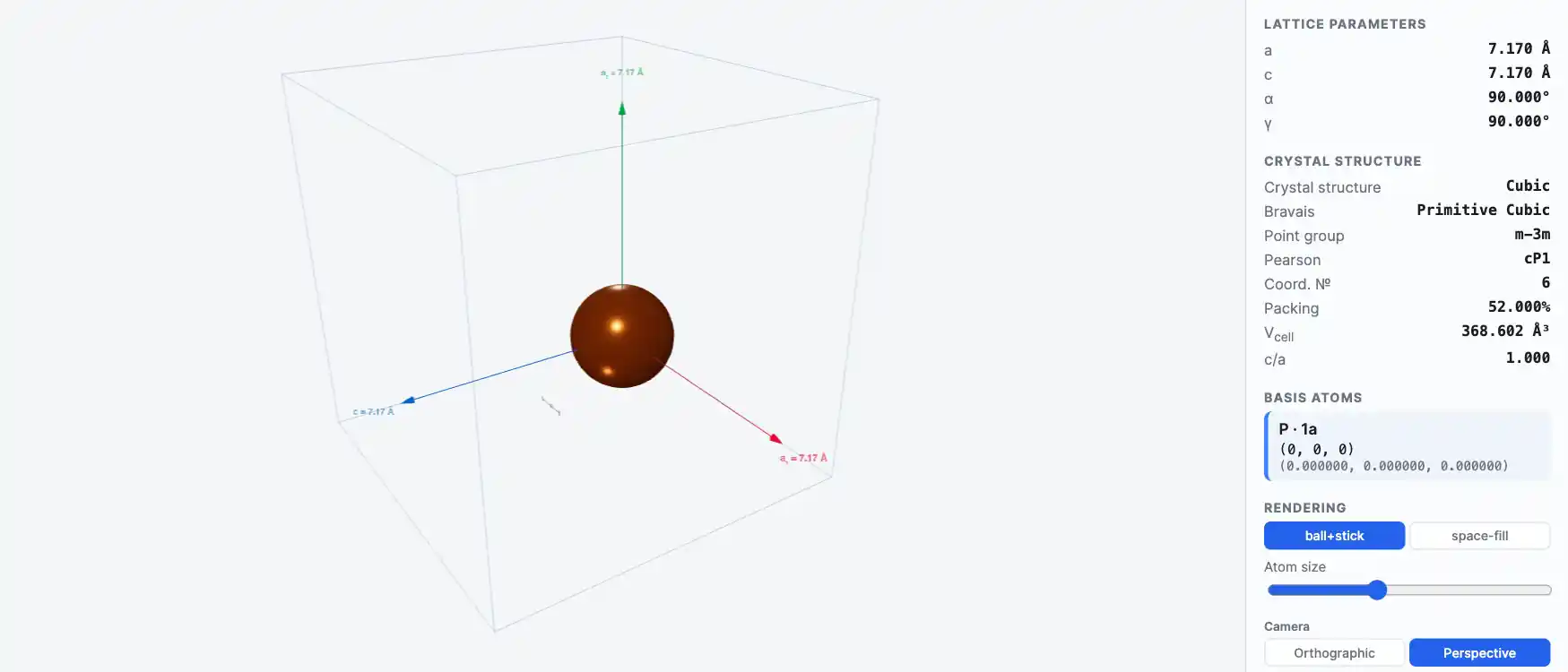
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
P: 3s² 3p³[Ne] 3s² 3p³1s² 2s² 2p⁶ 3s² 3p³Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 31 Stable | 30.97376199842 ± 0.0000000007 | 100.0000% | Stable |
Phase / State
Reason: 19.2 °C below melting point (44.15 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 15 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| P I | 0 | 258 | 132 | 133 |
| P II | +1 | 100 | 73 | 73 |
| P III | +2 | 70 | 23 | 23 |
| P IV | +3 | 129 | 78 | 78 |
| P V | +4 | 48 | 30 | 30 |
| P VI | +5 | 5 | 5 | 5 |
| P VII | +6 | 3 | 3 | 3 |
| P VIII | +7 | 20 | 20 | 20 |
| P IX | +8 | 47 | 47 | 47 |
| P X | +9 | 26 | 26 | 26 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| P I | 0 | 289 |
| P II | +1 | 162 |
| P III | +2 | 129 |
| P IV | +3 | 211 |
| P V | +4 | 68 |
| P VI | +5 | 60 |
| P VII | +6 | 62 |
| P VIII | +7 | 65 |
| P IX | +8 | 48 |
| P X | +9 | 58 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 44 pm |
| +5 | 4 | N/A | 17 pm |
| +5 | 5 | N/A | 28.999999999999996 pm |
| +5 | 6 | N/A | 38 pm |
Compounds
Isotopes (1)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 31 Stable | 30.97376199842 ± 0.0000000007 | 100.0000% | Stable | stable |
Spectral Lines
Showing 50 of 122 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 460.2069 nm | 600 | P II | emission | 3s2.3p.4p 3D → 3s2.3p.4d 3F* | Measured | NIST | |
| 422.2198 nm | 500 | P III | emission | 3s2.4s 2S → 3s2.4p 2P* | Measured | NIST | |
| 458.8032 nm | 500 | P II | emission | 3s2.3p.4p 3D → 3s2.3p.4d 3F* | Measured | NIST | |
| 458.9846 nm | 500 | P II | emission | 3s2.3p.4p 3D → 3s2.3p.4d 3F* | Measured | NIST | |
| 494.3497 nm | 500 | P II | emission | 3s2.3p.4p 3D → 3s2.3p.5s 3P* | Measured | NIST | |
| 602.418 nm | 500 | P II | emission | 3s2.3p.4s 3P* → 3s2.3p.4p 3D | Measured | NIST | |
| 604.308 nm | 500 | P II | emission | 3s2.3p.4s 3P* → 3s2.3p.4p 3D | Measured | NIST | |
| 405.9312 nm | 400 | P III | emission | 3s2.3d 2D → 3s2.4p 2P* | Measured | NIST | |
| 442.0712 nm | 400 | P II | emission | 3s2.3p.4s 1P* → 3s2.3p.4p 1S | Measured | NIST | |
| 529.6077 nm | 400 | P II | emission | 3s2.3p.4s 3P* → 3s2.3p.4p 3S | Measured | NIST | |
| 542.588 nm | 400 | P II | emission | 3s2.3p.4s 3P* → 3s2.3p.4p 3P | Measured | NIST | |
| 545.0709 nm | 400 | P II | emission | 3s2.3p.4p 3P → 3s2.3p.5s 3P* | Measured | NIST | |
| 603.404 nm | 400 | P II | emission | 3s2.3p.4s 3P* → 3s2.3p.4p 3D | Measured | NIST | |
| 424.672 nm | 350 | P III | emission | 3s2.4s 2S → 3s2.4p 2P* | Measured | NIST | |
| 608.784 nm | 350 | P II | emission | 3s2.3p.4s 3P* → 3s2.3p.4p 3D | Measured | NIST | |
| 616.56 nm | 350 | P II | emission | 3s2.3p.4s 3P* → 3s2.3p.4p 3D | Measured | NIST | |
| 395.7641 nm | 300 | P III | emission | 3s.3p.(3P*).4s 4P* → 3s.3p.(3P*).4p 4P | Measured | NIST | |
| 408.0089 nm | 300 | P III | emission | 3s2.3d 2D → 3s2.4p 2P* | Measured | NIST | |
| 424.9655 nm | 300 | P IV | emission | 3s.4s 1S → 3s.4p 1P* | Measured | NIST | |
| 462.6708 nm | 300 | P II | emission | 3s2.3p.4p 3D → 3s2.3p.4d 3F* | Measured | NIST | |
| 465.8309 nm | 300 | P II | emission | 3s2.3p.4p 3D → 3s2.3p.4d 3F* | Measured | NIST | |
| 495.4367 nm | 300 | P II | emission | 3s2.3p.4p 3D → 3s2.3p.5s 3P* | Measured | NIST | |
| 496.9701 nm | 300 | P II | emission | 3s2.3p.4p 3D → 3s2.3p.5s 3P* | Measured | NIST | |
| 525.3479 nm | 300 | P II | emission | 3s2.3p.4s 1P* → 3s2.3p.4p 1D | Measured | NIST | |
| 534.4729 nm | 300 | P II | emission | 3s2.3p.4s 3P* → 3s2.3p.4p 3P | Measured | NIST | |
| 538.6895 nm | 300 | P II | emission | 3s2.3p.4s 3P* → 3s2.3p.4p 3P | Measured | NIST | |
| 531.6055 nm | 250 | P II | emission | 3s2.3p.4s 3P* → 3s2.3p.4p 3P | Measured | NIST | |
| 537.8192 nm | 250 | P II | emission | 3s2.3p.4p 3P → 3s2.3p.5s 3P* | Measured | NIST | |
| 558.8301 nm | 250 | P II | emission | 3s2.3p.4p 3S → 3s2.3p.5s 3P* | Measured | NIST | |
| 605.55 nm | 250 | P II | emission | 3s2.3p.4p 1D → 3s2.3p.5s 1P* | Measured | NIST | |
| 390.4811 nm | 200 | P III | emission | 3s.3p.(3P*).4s 4P* → 3s.3p.(3P*).4p 4P | Measured | NIST | |
| 405.7449 nm | 200 | P III | emission | 3s2.3d 2D → 3s2.4p 2P* | Measured | NIST | |
| 438.5393 nm | 200 | P II | emission | 3s2.3p.4p 1P → 3s2.3p.5s 1P* | Measured | NIST | |
| 447.527 nm | 200 | P II | emission | 3s2.3p.4p 3P → 3s2.3p.4d 3D* | Measured | NIST | |
| 449.923 nm | 200 | P II | emission | 3s2.3p.4p 1D → 3s2.3p.4d 1F* | Measured | NIST | |
| 486.4426 nm | 200 | P II | emission | 3s2.3p.4p 3D → 3s2.3p.5s 3P* | Measured | NIST | |
| 540.9722 nm | 200 | P II | emission | 3s2.3p.4s 3P* → 3s2.3p.4p 3P | Measured | NIST | |
| 548.3519 nm | 200 | P II | emission | 3s2.3p.4p 3P → 3s2.3p.5s 3P* | Measured | NIST | |
| 549.9697 nm | 200 | P II | emission | 3s2.3p.4s 3P* → 3s2.3p.4p 3P | Measured | NIST | |
| 550.7174 nm | 200 | P II | emission | 3s2.3p.4p 3P → 3s2.3p.5s 3P* | Measured | NIST | |
| 554.1139 nm | 200 | P II | emission | 3s2.3p.4p 3P → 3s2.3p.5s 3P* | Measured | NIST | |
| 558.3235 nm | 200 | P II | emission | 3s2.3p.4p 3P → 3s2.3p.5s 3P* | Measured | NIST | |
| 534.5854 nm | 180 | P I | emission | 3s2.3p2.(3P).4s 2P → 3s2.3p2.(3P).5p 2P* | Measured | NIST | |
| 547.7672 nm | 180 | P I | emission | 3s2.3p2.(3P).4s 2P → 3s2.3p2.(3P).5p 2D* | Measured | NIST | |
| 716.547 nm | 180 | P I | emission | 3s2.3p2.(3P).4p 4D* → 3s2.3p2.(3P).5d 4F | Measured | NIST | |
| 717.666 nm | 180 | P I | emission | 3s2.3p2.(3P).4p 4D* → 3s2.3p2.(3P).5d 4F | Measured | NIST | |
| 492.7197 nm | 150 | P II | emission | 3s2.3p.4p 3D → 3s2.3p.5s 3P* | Measured | NIST | |
| 519.1393 nm | 150 | P II | emission | 3s2.3p.4s 3P* → 3s2.3p.4p 3S | Measured | NIST | |
| 510.9625 nm | 140 | P I | emission | 3s2.3p2.(3P).4s 4P → 3s2.3p2.(3P).5p 4P* | Measured | NIST | |
| 515.4842 nm | 140 | P I | emission | 3s2.3p2.(3P).4s 4P → 3s2.3p2.(3P).5p 4D* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 317.3 K |
| Boiling point | 553.65 K |
| Critical point (temperature) | 994.15 K |
| Melting point | 852.35 K |
| Boiling point | 704.15 K |
| Critical point (temperature) | 994.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (5)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.4422 |
| 2 | p | 4.0388 |
| 2 | s | 5.175 |
| 3 | p | 10.1136 |
| 3 | s | 9.3582 |
Crystal Radii Detail (4)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 58 | Ahrens (1952) ionic radius, | |
| 5 | IV | 31 | ||
| 5 | V | 43 | ||
| 5 | VI | 52 | calculated, |
Isotope Decay Modes (50)
| Isotope | Mode | Intensity |
|---|---|---|
| 24 | p | — |
| 24 | B+ | — |
| 24 | B+p | — |
| 25 | p | — |
| 26 | B+ | 100% |
| 26 | B+p | 35.1% |
| 26 | 2p | 2% |
| 27 | B+ | 100% |
| 27 | B+p | 0.1% |
| 28 | B+ | 100% |
X‑ray Scattering Factors (504)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 8.47738 |
| 10.1617 | — | 8.27092 |
| 10.3261 | — | 8.06949 |
| 10.4931 | — | 7.87297 |
| 10.6628 | — | 7.68123 |
| 10.8353 | — | 7.49416 |
| 11.0106 | — | 7.31165 |
| 11.1886 | — | 7.13359 |
| 11.3696 | — | 6.95985 |
| 11.5535 | — | 6.79035 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.05×103 milligrams per kilogram
References (1)
- [5] Phosphorus https://education.jlab.org/itselemental/ele015.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
6×10-2 milligrams per liter
References (1)
- [5] Phosphorus https://education.jlab.org/itselemental/ele015.html
Sources
Sources of this element.
Never found free in nature, it is widely distributed in combination with minerals. Phosphate rock, which contains the mineral apatite, an impure tri-calcium phosphate, is an important source of the element. Large deposits are found in Russia, in Morocco, and in Florida, Tennessee, Utah, Idaho, and elsewhere.
References (1)
- [6] Phosphorus https://periodic.lanl.gov/15.shtml
Production
Production of this element (from raw materials or other compounds containing the element).
White phosphorus may be made by several methods. By one process, tri-calcium phosphate, the essential ingredient of phosphate rock, is heated in the presence of carbon and silica in an electric furnace or fuel-fired furnace. Elementary phosphorus is liberated as vapor and may be collected under phosphoric acid, an important compound in making super-phosphate fertilizers.
References (1)
- [6] Phosphorus https://periodic.lanl.gov/15.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Phosphorus.
The element property data was retrieved from publications.