Silicon (Si)
metalloidSolid
Standard Atomic Weight
28.085 u [28.084, 28.086]Electron configuration
[Ne] 3s2 3p2Melting point
1413.85 °C (1687 K)Boiling point
3264.85 °C (3538 K)Density
2329.6 kg/m³Oxidation states
−4, −3, −2, −1, 0, +1, +2, +3, +4Electronegativity (Pauling)
1.9Ionization energy (1st)
Discovery year
1824Atomic radius
110 pmDetails
Silicon is a hard, brittle metalloid in group 14. It is tetravalent in most compounds and forms strong bonds to oxygen, making silicates the dominant minerals of Earth’s crust. Elemental silicon is central to modern electronics because its oxide, silicon dioxide, can be grown as a stable insulating layer. In bulk chemistry it is less reactive than carbon at ordinary temperatures, but it dissolves or reacts under strongly alkaline, oxidizing, or high-temperature conditions.
Crystalline silicon has a metallic luster and grayish color. Silicon is a relatively inert element, but it is attacked by halogens and dilute alkali. Most acids, except hydrofluoric, do not affect it. Elemental silicon transmits more than 95% of all wavelengths of infrared, from 1.3 to 6.y micro-m.
The name derives from the Latin silex and silicis for "flint". Amorphous silicon was discovered by the Swedish chemist Jöns Jacob Berzelius in 1824. Crystalline silicon was first prepared by the French chemist Henri Sainte-Claire Deville in 1854.
Silicon was discovered by Jöns Jacob Berzelius, a Swedish chemist, in 1824 by heating chips of potassium in a silica container and then carefully washing away the residual by-products. Silicon is the seventh most abundant element in the universe and the second most abundant element in the earth's crust. Today, silicon is produced by heating sand (SiO2) with carbon to temperatures approaching 2200°C.
From the Latin. word silex, silicis, flint. In 1800, Davy thought silica to be a compound and not an element; but in 1811, Gay Lussac and Thenard probably prepared impure amorphous silicon by heating potassium with silicon tetrafluoride.
In 1824 Berzelius, generally credited with the discovery, prepared amorphous silicon by the same general method and purified the product by removing the fluosilicates by repeated washings. Deville in 1854 first prepared crystalline silicon, the second allotropic form of the element.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
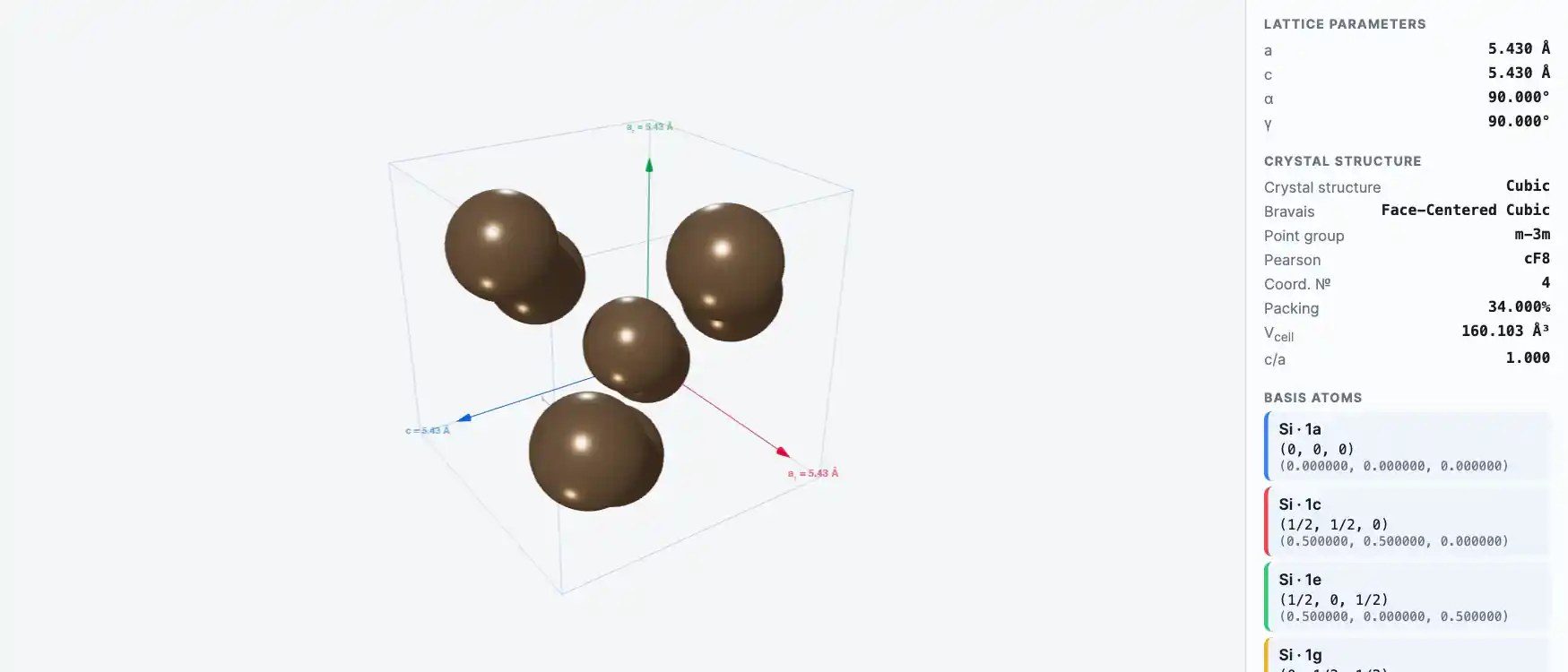
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Si: 3s² 3p²[Ne] 3s² 3p²1s² 2s² 2p⁶ 3s² 3p²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 28 Stable | 27.97692653465 ± 0.00000000044 | 92.2230% | Stable |
| 29 Stable | 28.9764946649 ± 0.00000000052 | 4.6850% | Stable |
| 30 Stable | 29.973770136 ± 0.000000023 | 3.0920% | Stable |
Phase / State
Reason: 1388.8 °C below melting point (1413.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 14 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Si I | 0 | 754 | 639 | 640 |
| Si II | +1 | 590 | 474 | 474 |
| Si III | +2 | 1298 | 1288 | 1288 |
| Si IV | +3 | 332 | 314 | 314 |
| Si V | +4 | 151 | 143 | 143 |
| Si VI | +5 | 346 | 346 | 346 |
| Si VII | +6 | 233 | 233 | 233 |
| Si VIII | +7 | 269 | 269 | 269 |
| Si IX | +8 | 366 | 366 | 366 |
| Si X | +9 | 315 | 315 | 315 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Si I | 0 | 542 |
| Si II | +1 | 151 |
| Si III | +2 | 189 |
| Si IV | +3 | 55 |
| Si V | +4 | 99 |
| Si VI | +5 | 72 |
| Si VII | +6 | 65 |
| Si VIII | +7 | 60 |
| Si IX | +8 | 67 |
| Si X | +9 | 55 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +4 | 4 | N/A | 26 pm |
| +4 | 6 | N/A | 40 pm |
Compounds
Isotopes (3)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 28 Stable | 27.97692653465 ± 0.00000000044 | 92.2230% ± 0.0190% | Stable | stable | |
| 29 Stable | 28.9764946649 ± 0.00000000052 | 4.6850% ± 0.0080% | Stable | stable | |
| 30 Stable | 29.973770136 ± 0.000000023 | 3.0920% ± 0.0110% | Stable | stable |
Spectral Lines
Showing 50 of 474 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 504.1024 nm | 1000 | Si II | emission | 3s2.4p 2P* → 3s2.4d 2D | Measured | NIST | |
| 505.5984 nm | 1000 | Si II | emission | 3s2.4p 2P* → 3s2.4d 2D | Measured | NIST | |
| 634.711 nm | 1000 | Si II | emission | 3s2.4s 2S → 3s2.4p 2P* | Measured | NIST | |
| 637.137 nm | 1000 | Si II | emission | 3s2.4s 2S → 3s2.4p 2P* | Measured | NIST | |
| 595.756 nm | 500 | Si II | emission | 3s2.4p 2P* → 3s2.5s 2S | Measured | NIST | |
| 597.893 nm | 500 | Si II | emission | 3s2.4p 2P* → 3s2.5s 2S | Measured | NIST | |
| 390.55231 nm | 300 | Si I | emission | 3s2.3p2 1S → 3s2.3p.4s 1P* | Measured | NIST | |
| 594.8541 nm | 200 | Si I | emission | 3s2.3p.4s 1P* → 3s2.3p.5p 1D | Measured | NIST | |
| 700.3569 nm | 180 | Si I | emission | 3s2.3p.4p 3D → 3s2.3p.6d 3F* | Measured | NIST | |
| 700.588 nm | 180 | Si I | emission | 3s2.3p.4p 3D → 3s2.3p.6d 3F* | Measured | NIST | |
| 570.84 nm | 160 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.5p 3P | Measured | NIST | |
| 462.1722 nm | 150 | Si II | emission | 3s2.4d 2D → 3s2.7f 2F* | Measured | NIST | |
| 568.4484 nm | 120 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.5p 3S | Measured | NIST | |
| 462.1418 nm | 100 | Si II | emission | 3s2.4d 2D → 3s2.7f 2F* | Measured | NIST | |
| 569.0425 nm | 100 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.5p 3P | Measured | NIST | |
| 579.7856 nm | 100 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.5p 3D | Measured | NIST | |
| 667.184 nm | 100 | Si II | emission | 3s.3p.(3P*).4s 4P* → 3s.3p.(3P*).4p 4D | Measured | NIST | |
| 672.1848 nm | 100 | Si I | emission | 3s2.3p.4p 1P → 3s2.3p.6d 1D* | Measured | NIST | |
| 564.5613 nm | 90 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.5p 3P | Measured | NIST | |
| 570.1104 nm | 90 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.5p 3P | Measured | NIST | |
| 579.3073 nm | 90 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.5p 3D | Measured | NIST | |
| 479.2324 nm | 80 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.(2P*<3/2>).6p<1/2> (3/2,1/2) | Measured | NIST | |
| 566.5555 nm | 80 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.5p 3P | Measured | NIST | |
| 697.651 nm | 80 | Si I | emission | 3s2.3p.4p 3D → 3s2.3p.6d 3F* | Measured | NIST | |
| 410.29359 nm | 70 | Si I | emission | 3s2.3p2 1S → 3s2.3p.4s 3P* | Measured | NIST | |
| 577.2146 nm | 70 | Si I | emission | 3s2.3p.4s 1P* → 3s2.3p.5p 1S | Measured | NIST | |
| 578.0384 nm | 70 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.5p 3D | Measured | NIST | |
| 719.355 nm | 65 | Si I | emission | 3s2.3p.4p 3P → 3s2.3p.6d 3D* | Measured | NIST | |
| 478.2991 nm | 50 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.(2P*<3/2>).6p<1/2> (3/2,1/2) | Measured | NIST | |
| 682.983 nm | 50 | Si II | emission | 3s2.5p 2P* → 3s2.6d 2D | Measured | NIST | |
| 575.4218 nm | 45 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.5p 3P | Measured | NIST | |
| 633.1956 nm | 45 | Si I | emission | 3s2.3p.4s 1P* → 3s2.3p.5p 1P | Measured | NIST | |
| 655.5463 nm | 45 | Si I | emission | 3s2.3p.4p 3D → 3s2.3p.7d 3F* | Measured | NIST | |
| 500.6059 nm | 40 | Si I | emission | 3s2.3p.4s 1P* → 3s2.3p.(2P*<3/2>).6p<3/2> (3/2,3/2) | Measured | NIST | |
| 479.2213 nm | 35 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.(2P*<1/2>).6p<1/2> (1/2,1/2) | Measured | NIST | |
| 380.6526 nm | 30 | Si III | emission | 3s.4p 3P* → 3s.4d 3D | Measured | NIST | |
| 455.2622 nm | 30 | Si III | emission | 3s.4s 3S → 3s.4p 3P* | Measured | NIST | |
| 494.7607 nm | 30 | Si I | emission | 3s2.3p.4s 1P* → 3s2.3p.(2P*<3/2>).6p<3/2> (3/2,3/2) | Measured | NIST | |
| 562.222 nm | 30 | Si I | emission | 3s2.3p.4s 3P* → 3s2.3p.5p 3S | Measured | NIST | |
| 681.841 nm | 30 | Si II | emission | 3s2.5p 2P* → 3s2.6d 2D | Measured | NIST | |
| 456.784 nm | 25 | Si III | emission | 3s.4s 3S → 3s.4p 3P* | Measured | NIST | |
| 392.4468 nm | 20 | Si III | emission | 3s.4f 1F* → 3s.5g 1G | Measured | NIST | |
| 457.4757 nm | 20 | Si III | emission | 3s.4s 3S → 3s.4p 3P* | Measured | NIST | |
| 573.973 nm | 20 | Si III | emission | 3s.4s 1S → 3s.4p 1P* | Measured | NIST | |
| 669.94 nm | 20 | Si II | emission | 3s.3p.(3P*).4s 4P* → 3s.3p.(3P*).4p 4D | Measured | NIST | |
| 482.895 nm | 18 | Si III | emission | 3s.4f 3F* → 3s.5g 3G | Measured | NIST | |
| 471.6654 nm | 16 | Si III | emission | 3s.4d 1D → 3s.5f 1F* | Measured | NIST | |
| 481.9712 nm | 16 | Si III | emission | 3s.4f 3F* → 3s.5g 3G | Measured | NIST | |
| 481.3333 nm | 15 | Si III | emission | 3s.4f 3F* → 3s.5g 3G | Measured | NIST | |
| 666.503 nm | 15 | Si II | emission | 3s.3p.(3P*).4s 4P* → 3s.3p.(3P*).4p 4D | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Phase Transitions & Allotropes
| Melting point | 1687.15 K |
| Boiling point | 3538.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (5)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.4255 |
| 2 | p | 4.055 |
| 2 | s | 4.98 |
| 3 | p | 9.7148 |
| 3 | s | 9.0968 |
Crystal Radii Detail (2)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 4 | IV | 40 | ||
| 4 | VI | 54 | from r^3 vs V plots, |
Isotope Decay Modes (46)
| Isotope | Mode | Intensity |
|---|---|---|
| 22 | B+ | 100% |
| 22 | B+p | 62% |
| 22 | 2p | 0.7% |
| 23 | B+ | 100% |
| 23 | B+p | 88% |
| 23 | 2p | 3.6% |
| 24 | B+ | 100% |
| 24 | B+p | 34.5% |
| 25 | B+ | 100% |
| 25 | B+p | 35% |
X‑ray Scattering Factors (756)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 3.94851 |
| 10.1617 | — | 3.95531 |
| 10.3261 | — | 3.96212 |
| 10.4931 | — | 3.96894 |
| 10.6628 | — | 3.97577 |
| 10.8353 | — | 3.98262 |
| 11.0105 | — | 3.98948 |
| 11.1886 | — | 3.99635 |
| 11.3696 | — | 4.00322 |
| 11.5535 | — | 4.01012 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
2.82×105 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
2.2 milligrams per liter
References (1)
Sources
Sources of this element.
Silicon is present in the sun and stars and is a principal component of a class of meteorites known as aerolites. It is also a component of tektites, a natural glass of uncertain origin.
Silicon makes up 25.7% of the earth's crust, by weight, and is the second most abundant element, being exceeded only by oxygen. Silicon is not found free in nature, but occurs chiefly as the oxide and as silicates. Sand, quartz, rock crystal, amethyst, agate, flint, jasper, and opal are some of the forms in which the oxide appears. Granite, hornblende, asbestos, feldspar, clay, mica, etc. are but a few of the numerous silicate minerals.
Silicon is prepared commercially by heating silica and carbon in an electric furnace, using carbon electrodes. Several other methods can be used for preparing the element. Amorphous silicon can be prepared as a brown powder, which can be easily melted or vaporized. The Czochralski process is commonly used to produce single crystals of silicon used for solid-state or semiconductor devices. Hyperpure silicon can be prepared by the thermal decomposition of ultra-pure trichlorosilane in a hydrogen atmosphere, and by a vacuum float zone process.
References (1)
- [6] Silicon https://periodic.lanl.gov/14.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Silicon.
The element property data was retrieved from publications.