Actinium (Ac)
actinideSolid
Standard Atomic Weight
[227]Electron configuration
[Rn] 7s2 6d1Melting point
1050.85 °C (1324 K)Boiling point
3197.85 °C (3471 K)Density
1.007000e+4 kg/m³Oxidation states
+3Electronegativity (Pauling)
1.1Ionization energy (1st)
Discovery year
1899Atomic radius
195 pmDetails
Actinium is a radioactive actinide metal and the element that gives the actinide series its name. Natural actinium is present only in minute amounts, chiefly as ²²⁷Ac in uranium decay chains. Its chemistry is dominated by the +3 oxidation state and resembles that of lanthanum more than the later, more strongly 5f-influenced actinides. Because all isotopes are radioactive and scarce, most knowledge comes from tracer-scale chemistry rather than ordinary bulk handling.
Actinium-227, a decay product of uranium-235, is a beta emitter with a 21.6-year half-life. Its principal decay products are thorium-227 (18.5-day half-life), radium-223 (11.4-day half-life), and a number of short-lived products including radon, bismuth, polonium, and lead isotopes. In equilibrium with its decay products, it is a powerful source of alpha particles. Actinium metal has been prepared by the reduction of actinium fluoride with lithium vapor at about 1100 to 1300-degrees C. The chemical behavior of actinium is similar to that of the rare earths, particularly lanthanum. Purified actinium comes into equilibrium with its decay products at the end of 185 days, and then decays according to its 21.6-year half-life. It is about 150 times as active as radium, making it of value in the production of neutrons.
In April of 2012, Los Alamos National Laboratory announced a new medical isotope project that shows promise for rapidly producing major quantities of a new cancer-treatment agent, actinium 225 (Ac-225). Both a press release and a video are available.
Actinium was discovered in 1899 by André-Louis Debierne, a French chemist, while experimenting with new methods of separating rare earth oxides. Friedrich Otto Giesel independently discovered actinium in 1902. Actinium is a rare element that is present in uranium ores in tiny amounts, but it is usually cheaper and easier to create actinium when it is needed by bombarding radium with neutrons in a nuclear reactor.
Actinium's most stable isotope, actinium-227, has a half-life of 21.77 years. It decays into francium-223 through alpha decay or into thorium-227 through beta decay.
From the Greek aktis, aktinos, meaning beam or ray. Discovered by Andre Debierne in 1899 and independently by F. Giesel in 1902. Occurs naturally in association with uranium minerals.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
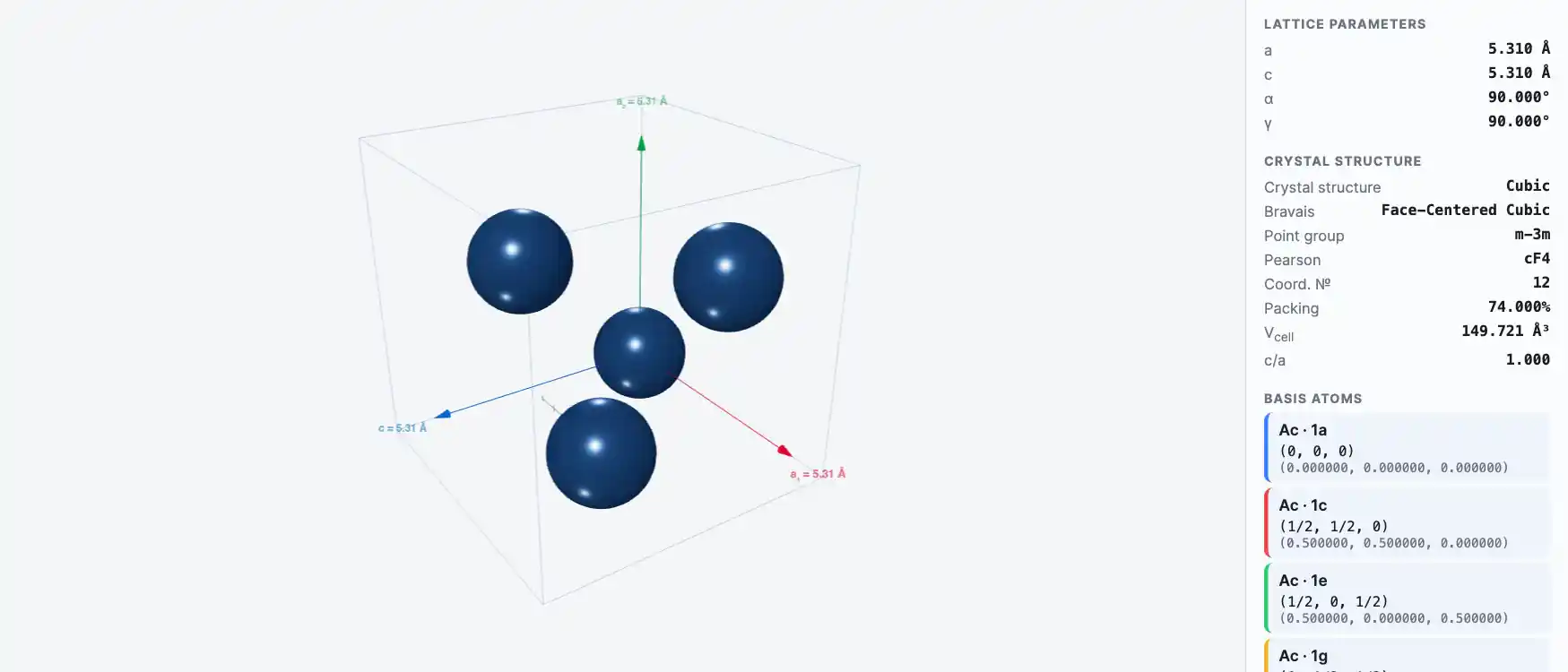
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Ac: 6d¹ 7s²[Rn] 6d¹ 7s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d¹⁰ 6s² 6p⁶ 6d¹ 7s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
No stable isotopes.
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 212 Radioactive | 212.007813 ± 0.000055 | N/A | 895 ms |
| 213 Radioactive | 213.006609 ± 0.000056 | N/A | 738 ms |
| 216 Radioactive | 216.008743 ± 0.000012 | N/A | 440 us |
| 210 Radioactive | 210.009436 ± 0.000062 | N/A | 350 ms |
| 211 Radioactive | 211.007732 ± 0.000057 | N/A | 213 ms |
Phase / State
Reason: 1025.8 °C below melting point (1050.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 89 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Ac I | 0 | 153 | 95 | 113 |
| Ac II | +1 | 345 | 294 | 319 |
| Ac III | +2 | 11 | 11 | 11 |
| Ac IV | +3 | 6 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Ac I | 0 | 53 |
| Ac II | +1 | 84 |
| Ac III | +2 | 8 |
| Ac IV | +3 | 2 |
| Ac V | +4 | 2 |
| Ac VI | +5 | 2 |
| Ac VII | +6 | 2 |
| Ac VIII | +7 | 2 |
| Ac IX | +8 | 2 |
| Ac X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 112.00000000000001 pm |
| +3 | 9 | N/A | 122 pm |
Compounds
Isotopes (5)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 212 Radioactive | 212.007813 ± 0.000055 | N/A | 895 ms | α ≈100%β+ ? | |
| 213 Radioactive | 213.006609 ± 0.000056 | N/A | 738 ms | α ≈100%β+ ? | |
| 216 Radioactive | 216.008743 ± 0.000012 | N/A | 440 us | α =100%β+ ? | |
| 210 Radioactive | 210.009436 ± 0.000062 | N/A | 350 ms | α ≈100%β+ ? | |
| 211 Radioactive | 211.007732 ± 0.000057 | N/A | 213 ms | α ≈100%β+ ? |
Spectral Lines
Showing 50 of 213 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 391.4468 nm | 24000 | Ac II | emission | 6d.7s 3D → 5f.7s 3F* | Measured | NIST | |
| 417.99772 nm | 17000 | Ac I | emission | 6d.7s2 2D → 6d.7s.(1D).7p 2D* | Measured | NIST | |
| 481.2218 nm | 16000 | Ac II | emission | 6d.7s 3D → 7s.7p 3P* | Measured | NIST | |
| 438.64 nm | 15000 | Ac II | emission | 6d.7s 1D → 6d.7p 1D* | Measured | NIST | |
| 397.736 nm | 13000 | Ac II | emission | 6d2 3F → 6d.7p 3P* | Measured | NIST | |
| 445.2188 nm | 12000 | Ac II | emission | 6d.7s 3D → 6d.7p 3F* | Measured | NIST | |
| 388.55592 nm | 9000 | Ac I | emission | 6d.7s2 2D → 6d.7s.(3D).7p 2P* | Measured | NIST | |
| 401.9622 nm | 8700 | Ac II | emission | 6d2 3F → 5f.6d 1G* | Measured | NIST | |
| 544.6367 nm | 8700 | Ac II | emission | 6d2 3F → 6d.7p 3D* | Measured | NIST | |
| 435.9118 nm | 7300 | Ac II | emission | 6d.7s 3D → 7s.7p 3P* | Measured | NIST | |
| 446.27307 nm | 6900 | Ac I | emission | 6d.7s2 2D → 6d.7s.(1D).7p 2F* | Measured | NIST | |
| 471.65807 nm | 6900 | Ac I | emission | 6d.7s2 2D → 6d.7s.(1D).7p 2F* | Measured | NIST | |
| 495.8233 nm | 6800 | Ac II | emission | 6d.7s 1D → 6d.7p 1P* | Measured | NIST | |
| 384.304 nm | 6600 | Ac I | emission | 6d.7s2 2D → 6d.7s.(3D).7p 2P* | Measured | NIST | |
| 392.0101 nm | 6500 | Ac II | emission | 6d2 3P → 5f.6d 1D* | Measured | NIST | |
| 418.31199 nm | 6400 | Ac I | emission | 6d.7s2 2D → 6d.7s.(3D).7p 2F* | Measured | NIST | |
| 439.67158 nm | 5800 | Ac I | emission | 6d.7s2 2D → 6d.7s.(3D).7p 2F* | Measured | NIST | |
| 419.43971 nm | 5300 | Ac I | emission | 6d.7s2 2D → 6d.7s.(1D).7p 2P* | Measured | NIST | |
| 494.5181 nm | 5300 | Ac II | emission | 6d2 3F → 6d.7p 3D* | Measured | NIST | |
| 496.0869 nm | 4900 | Ac II | emission | 6d2 3F → 6d.7p 3D* | Measured | NIST | |
| 461.39285 nm | 4000 | Ac I | emission | 6d.7s2 2D → 6d.7s.(3D).7p 2F* | Measured | NIST | |
| 527.15603 nm | 3800 | Ac I | emission | 6d.7s2 2D → 6d.7s.(1D).7p 2F* | Measured | NIST | |
| 406.31064 nm | 3700 | Ac I | emission | 6d.7s2 2D → 6d.7s.(1D).7p 2D* | Measured | NIST | |
| 407.8693 nm | 3700 | Ac II | emission | 6d.7s 3D → 6d.7p 1P* | Measured | NIST | |
| 403.4629 nm | 3500 | Ac I | emission | 6d.7s2 2D → 6d2.(3F).7p 4G* | Measured | NIST | |
| 420.9682 nm | 3500 | Ac II | emission | 6d.7s 3D → 5f.7s 1F* | Measured | NIST | |
| 669.5231 nm | 3300 | Ac II | emission | 6d2 3F → 6d.7p 3F* | Measured | NIST | |
| 474.0522 nm | 2600 | Ac II | emission | 6d2 1G → 6d.7p 1F* | Measured | NIST | |
| 515.6541 nm | 2100 | Ac II | emission | 6d2 3F → 5f.7s 3F* | Measured | NIST | |
| 383.53206 nm | 1900 | Ac I | emission | 6d.7s2 2D → 6d.7s.(1D).7p 2P* | Measured | NIST | |
| 488.9102 nm | 1800 | Ac II | emission | 6d2 3F → 5f.7s 3F* | Measured | NIST | |
| 480.7843 nm | 1700 | Ac II | emission | 6d.7s 1D → 6d.7p 3F* | Measured | NIST | |
| 534.47384 nm | 1700 | Ac I | emission | 6d2.(3F).7s 4F → 6d2.(3F).7p 4G* | Measured | NIST | |
| 400.5469 nm | 1500 | Ac II | emission | 6d2 1G → 5f.6d 3G* | Measured | NIST | |
| 536.2615 nm | 1400 | Ac II | emission | 6d2 3F → 6d.7p 1D* | Measured | NIST | |
| 522.8309 nm | 1300 | Ac I | emission | 6d.7s2 2D → 6d.7s.(3D).7p 4D* | Measured | NIST | |
| 454.408 nm | 1200 | Ac II | emission | 6d2 1D → 7s.7p 1P* | Measured | NIST | |
| 461.01055 nm | 1100 | Ac I | emission | 6d.7s2 2D → 6d.7s.(1D).7p 2D* | Measured | NIST | |
| 422.599 nm | 1000 | Ac I | emission | 6d2.(3F).7s 4F → 6d2.(3F).7p 4D* | Measured | NIST | |
| 387.7035 nm | 980 | Ac II | emission | 6d.7p 3P* → 5f.7p 3D | Measured | NIST | |
| 420.89072 nm | 960 | Ac I | emission | 6d2.(3F).7s 4F → 6d2.(3F).7p 4D* | Measured | NIST | |
| 469.05284 nm | 960 | Ac I | emission | 6d2.(3F).7s 4F → 6d2.(3F).7p 4F* | Measured | NIST | |
| 521.5399 nm | 960 | Ac II | emission | 6d2 3P → 6d.7p 3P* | Measured | NIST | |
| 486.88523 nm | 890 | Ac I | emission | 6d2.(3F).7s 4F → 6d2.(3F).7p 4F* | Measured | NIST | |
| 526.4481 nm | 890 | Ac I | emission | 7s2.7p 2P* → 7s2.8s 2S | Measured | NIST | |
| 440.21056 nm | 860 | Ac I | emission | 6d2.(3F).7s 4F → 6d2.(3F).7p 4F* | Measured | NIST | |
| 447.18106 nm | 850 | Ac I | emission | 6d2.(3F).7s 4F → 6d2.(3F).7p 4F* | Measured | NIST | |
| 421.80204 nm | 740 | Ac I | emission | 6d2.(3F).7s 4F → 6d2.(3P).7p 2D* | Measured | NIST | |
| 462.16811 nm | 710 | Ac I | emission | 6d2.(3F).7s 4F → 6d2.(3F).7p 4F* | Measured | NIST | |
| 426.8209 nm | 570 | Ac II | emission | 6d2 3F → 6d.7p 3P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Phase Transitions & Allotropes
| Melting point | 1323.15 K |
| Boiling point | 3473.15 K |
Oxidation State Categories
Advanced Reference Data
Crystal Radii Detail (2)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 126 | from r^3 vs V plots, | |
| 3 | IX | — | 136 |
Isotope Decay Modes (56)
| Isotope | Mode | Intensity |
|---|---|---|
| 205 | A | 100% |
| 205 | B+ | — |
| 206 | A | 100% |
| 206 | B+ | — |
| 207 | A | 100% |
| 208 | A | 100% |
| 208 | B+ | — |
| 209 | A | 100% |
| 209 | B+ | — |
| 210 | A | 100% |
X‑ray Scattering Factors (516)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.19484 |
| 10.1617 | — | 1.15672 |
| 10.3261 | — | 1.11982 |
| 10.4931 | — | 1.0841 |
| 10.6628 | — | 1.04952 |
| 10.8353 | — | 1.01604 |
| 11.0106 | — | 0.98234 |
| 11.1886 | — | 0.94669 |
| 11.3696 | — | 0.91233 |
| 11.5535 | — | 0.88152 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
5.5×10-10 milligrams per kilogram
References (1)
- [5] Actinium https://education.jlab.org/itselemental/ele089.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
Not Applicable
References (1)
- [5] Actinium https://education.jlab.org/itselemental/ele089.html
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Actinium.
The element property data was retrieved from publications.