Oxygen (O)
nonmetalGas
Standard Atomic Weight
15.999 u [15.99903, 15.99977]Electron configuration
[He] 2s2 2p4Melting point
-218.79 °C (54.36 K)Boiling point
-182.95 °C (90.2 K)Density
1.429 kg/m³Oxidation states
−2, −1, 0, +1, +2Electronegativity (Pauling)
3.44Ionization energy (1st)
Discovery year
1771Atomic radius
60 pmDetails
Oxygen is a reactive nonmetal and chalcogen that occurs mainly as the diatomic gas O₂ and, less commonly, as ozone O₃. It is essential to aerobic respiration and is a major constituent of water, silicate minerals, carbonates, and many biological molecules. Its high electronegativity and ability to form strong bonds make oxidation chemistry central to combustion, corrosion, metabolism, and industrial processing.
The gas is colorless, odorless, and tasteless. The liquid and solid forms are a pale blue color and are strongly paramagnetic.
The name derives from the Greek oxys for "acid" and genes for "forming" because the French chemist Antoine-Laurent Lavoisier once thought that oxygen was integral to all acids.
Oxygen was discovered independently by the Swedish pharmacist and chemist Carl-Wilhelm Scheele in 1771, and the English clergyman and chemist Joseph Priestley in 1774. Scheele's Chemical Treatise on Air and Fire was delayed in publication until 1777, so Priestley is credited with the discovery because he published first.
Oxygen had been produced by several chemists prior to its discovery in 1774, but they failed to recognize it as a distinct element. Joseph Priestley and Carl Wilhelm Scheele both independently discovered oxygen, but Priestly is usually given credit for the discovery. They were both able to produce oxygen by heating mercuric oxide (HgO). Priestley called the gas produced in his experiments 'dephlogisticated air' and Scheele called his 'fire air'. The name oxygen was created by Antoine Lavoisier who incorrectly believed that oxygen was necessary to form all acids. Oxygen is the third most abundant element in the universe and makes up nearly 21% of the earth's atmosphere. Oxygen accounts for nearly half of the mass of the earth's crust, two thirds of the mass of the human body and nine tenths of the mass of water. Large amounts of oxygen can be extracted from liquefied air through a process known as fractional distillation. Oxygen can also be produced through the electrolysis of water or by heating potassium chlorate (KClO3).
From the Greek word oxys, acid, and genes, forming. The behavior of oxygen and nitrogen as components of air led to the advancement of the phlogiston theory of combustion, which captured the minds of chemists for a century.
Joseph Priestley is generally credited with its discovery, although Scheele also discovered it independently.
Its atomic weight was used as a standard of comparison for each of the other elements until 1961 when the International Union of Pure and Applied Chemistry adopted carbon 12 as the new basis.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
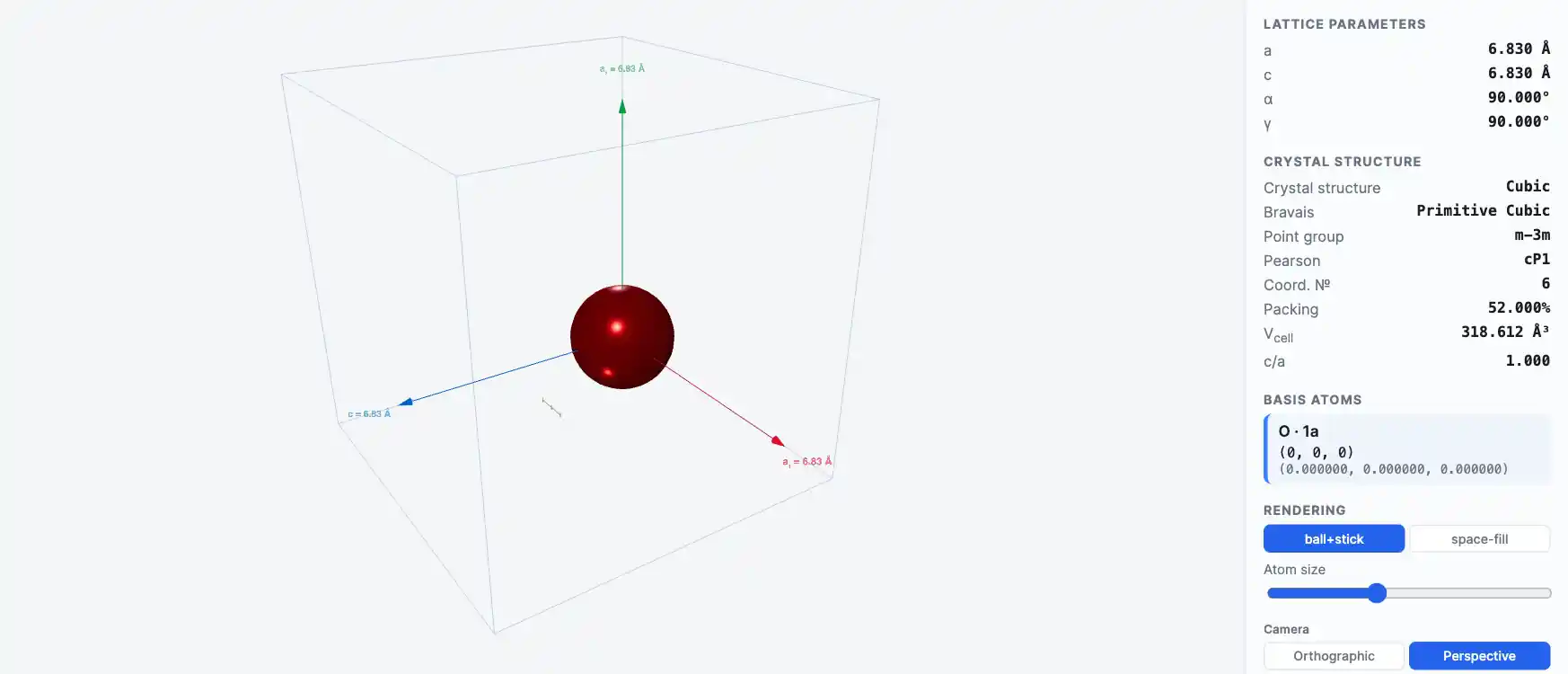
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
O: 2s² 2p⁴[He] 2s² 2p⁴1s² 2s² 2p⁴Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 16 Stable | 15.99491461957 ± 0.00000000017 | 99.7570% | Stable |
| 17 Stable | 16.9991317565 ± 0.00000000069 | 0.0380% | Stable |
| 18 Stable | 17.99915961286 ± 0.00000000076 | 0.2050% | Stable |
Phase / State
Reason: 207.9 °C above boiling point (-182.95 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Density
At standard conditions
Estimated via ideal gas law at current T
Advanced
Atomic Spectra
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| O I | 0 | 910 | 854 | 907 |
| O II | +1 | 1630 | 876 | 1630 |
| O III | +2 | 1005 | 974 | 974 |
| O IV | +3 | 1525 | 1521 | 1523 |
| O V | +4 | 391 | 385 | 385 |
| O VI | +5 | 157 | 126 | 157 |
| O VII | +6 | 189 | 188 | 189 |
| O VIII | +7 | 137 | 137 | 137 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| O I | 0 | 614 |
| O II | +1 | 287 |
| O III | +2 | 188 |
| O IV | +3 | 219 |
| O V | +4 | 172 |
| O VI | +5 | 148 |
| O VII | +6 | 149 |
| O VIII | +7 | 149 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| -2 | 2 | N/A | 135 pm |
| -2 | 3 | N/A | 136 pm |
| -2 | 4 | N/A | 138 pm |
| -2 | 6 | N/A | 140 pm |
| -2 | 8 | N/A | 142 pm |
Compounds
Isotopes (3)
Oxygen has nine isotopes. Natural oxygen is a mixture of three isotopes.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 16 Stable | 15.99491461957 ± 0.00000000017 | 99.7570% ± 0.0160% | Stable | stable | |
| 17 Stable | 16.9991317565 ± 0.00000000069 | 0.0380% ± 0.0010% | Stable | stable | |
| 18 Stable | 17.99915961286 ± 0.00000000076 | 0.2050% ± 0.0140% | Stable | stable |
Spectral Lines
Showing 50 of 1013 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 615.8187 nm | 490 | O I | emission | 2s2.2p3.(4S*).3p 5P → 2s2.2p3.(4S*).4d 5D* | Measured | NIST | |
| 615.6778 nm | 450 | O I | emission | 2s2.2p3.(4S*).3p 5P → 2s2.2p3.(4S*).4d 5D* | Measured | NIST | |
| 700.223 nm | 450 | O I | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).4d 3D* | Measured | NIST | |
| 725.4448 nm | 450 | O I | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).5s 3S* | Measured | NIST | |
| 615.5971 nm | 400 | O I | emission | 2s2.2p3.(4S*).3p 5P → 2s2.2p3.(4S*).4d 5D* | Measured | NIST | |
| 645.5977 nm | 400 | O I | emission | 2s2.2p3.(4S*).3p 5P → 2s2.2p3.(4S*).5s 5S* | Measured | NIST | |
| 725.4154 nm | 400 | O I | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).5s 3S* | Measured | NIST | |
| 645.4444 nm | 360 | O I | emission | 2s2.2p3.(4S*).3p 5P → 2s2.2p3.(4S*).5s 5S* | Measured | NIST | |
| 700.1922 nm | 360 | O I | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).4d 3D* | Measured | NIST | |
| 645.3602 nm | 320 | O I | emission | 2s2.2p3.(4S*).3p 5P → 2s2.2p3.(4S*).5s 5S* | Measured | NIST | |
| 725.4531 nm | 320 | O I | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).5s 3S* | Measured | NIST | |
| 715.6701 nm | 210 | O I | emission | 2s2.2p3.(2D*).3s 1D* → 2s2.2p3.(2D*).3p 1D | Measured | NIST | |
| 396.1573 nm | 200 | O III | emission | 2s2.2p.(2P*).3p 1D → 2s2.2p.(2P*).3d 1F* | Measured | NIST | |
| 533.0741 nm | 190 | O I | emission | 2s2.2p3.(4S*).3p 5P → 2s2.2p3.(4S*).5d 5D* | Measured | NIST | |
| 604.6438 nm | 190 | O I | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).6s 3S* | Measured | NIST | |
| 394.72949 nm | 185 | O I | emission | 2s2.2p3.(4S*).3s 5S* → 2s2.2p3.(4S*).4p 5P | Measured | NIST | |
| 394.74813 nm | 160 | O I | emission | 2s2.2p3.(4S*).3s 5S* → 2s2.2p3.(4S*).4p 5P | Measured | NIST | |
| 532.9681 nm | 160 | O I | emission | 2s2.2p3.(4S*).3p 5P → 2s2.2p3.(4S*).5d 5D* | Measured | NIST | |
| 604.6233 nm | 160 | O I | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).6s 3S* | Measured | NIST | |
| 394.75862 nm | 140 | O I | emission | 2s2.2p3.(4S*).3s 5S* → 2s2.2p3.(4S*).4p 5P | Measured | NIST | |
| 543.6862 nm | 135 | O I | emission | 2s2.2p3.(4S*).3p 5P → 2s2.2p3.(4S*).6s 5S* | Measured | NIST | |
| 559.789 nm | 130 | O V | emission | 1s2.2s.3p 3P* → 1s2.2s.3d 3D | Measured | NIST | |
| 650.024 nm | 130 | O V | emission | 1s2.2p.(2P*<3/2>).3p 3D → 1s2.2p.(2P*<3/2>).3d 3F* | Measured | NIST | |
| 382.34136 nm | 120 | O I | emission | 2s2.2p3.(2D*).3s 3D* → 2s2.2p3.(2P*).3p 3D | Measured | NIST | |
| 557.7339 nm | 120 | O I | emission | 2s2.2p4 1D → 2s2.2p4 1S | Measured | NIST | |
| 543.5775 nm | 110 | O I | emission | 2s2.2p3.(4S*).3p 5P → 2s2.2p3.(4S*).6s 5S* | Measured | NIST | |
| 559.2252 nm | 110 | O III | emission | 2s2.2p.(2P*).3s 1P* → 2s2.2p.(2P*).3p 1P | Measured | NIST | |
| 604.6495 nm | 110 | O I | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(4S*).6s 3S* | Measured | NIST | |
| 395.46067 nm | 100 | O I | emission | 2s2.2p3.(4S*).3p 3P → 2s2.2p3.(2P*).3s 3P* | Measured | NIST | |
| 412.396 nm | 100 | O V | emission | 1s2.2p.(2P*<3/2>).3s 3P* → 1s2.2p.(2P*<3/2>).3p 3D | Measured | NIST | |
| 436.8258 nm | 100 | O I | emission | 2s2.2p3.(4S*).3s 3S* → 2s2.2p3.(4S*).4p 3P | Measured | NIST | |
| 543.5178 nm | 90 | O I | emission | 2s2.2p3.(4S*).3p 5P → 2s2.2p3.(4S*).6s 5S* | Measured | NIST | |
| 423.3274 nm | 80 | O I | emission | 2s2.2p3.(4S*).4p 3P → 2s2.2p3.(2D*<3/2>).3d 3P* | Measured | NIST | |
| 441.4899 nm | 27 | O II | emission | 2s2.2p2.(3P).3s 2P → 2s2.2p2.(3P).3p 2D* | Measured | NIST | |
| 672.1388 nm | 26 | O II | emission | 2s2.2p2.(3P).3s 2P → 2s2.2p2.(3P).3p 2S* | Measured | NIST | |
| 441.6975 nm | 25 | O II | emission | 2s2.2p2.(3P).3s 2P → 2s2.2p2.(3P).3p 2D* | Measured | NIST | |
| 397.3256 nm | 24 | O II | emission | 2s2.2p2.(3P).3s 2P → 2s2.2p2.(3P).3p 2P* | Measured | NIST | |
| 407.58617 nm | 24 | O II | emission | 2s2.2p2.(3P).3p 4D* → 2s2.2p2.(3P).3d 4F | Measured | NIST | |
| 464.91347 nm | 24 | O II | emission | 2s2.2p2.(3P).3s 4P → 2s2.2p2.(3P).3p 4D* | Measured | NIST | |
| 664.1031 nm | 24 | O II | emission | 2s2.2p2.(3P).3s 2P → 2s2.2p2.(3P).3p 2S* | Measured | NIST | |
| 407.21525 nm | 23 | O II | emission | 2s2.2p2.(3P).3p 4D* → 2s2.2p2.(3P).3d 4F | Measured | NIST | |
| 434.9426 nm | 23 | O II | emission | 2s2.2p2.(3P).3s 4P → 2s2.2p2.(3P).3p 4P* | Measured | NIST | |
| 411.92165 nm | 22 | O II | emission | 2s2.2p2.(3P).3p 4P* → 2s2.2p2.(3P).3d 4D | Measured | NIST | |
| 459.0974 nm | 22 | O II | emission | 2s2.2p2.(1D).3s 2D → 2s2.2p2.(1D).3p 2F* | Measured | NIST | |
| 464.18103 nm | 22 | O II | emission | 2s2.2p2.(3P).3s 4P → 2s2.2p2.(3P).3p 4D* | Measured | NIST | |
| 689.5102 nm | 22 | O II | emission | 2s2.2p2.(3P).3d 4F → 2s2.2p2.(3P).4p 4D* | Measured | NIST | |
| 406.98819 nm | 21 | O II | emission | 2s2.2p2.(3P).3p 4D* → 2s2.2p2.(3P).3d 4F | Measured | NIST | |
| 435.126 nm | 21 | O II | emission | 2s2.2p2.(1D).3s 2D → 2s2.2p2.(1D).3p 2D* | Measured | NIST | |
| 466.16324 nm | 21 | O II | emission | 2s2.2p2.(3P).3s 4P → 2s2.2p2.(3P).3p 4D* | Measured | NIST | |
| 470.5346 nm | 21 | O II | emission | 2s2.2p2.(3P).3p 2D* → 2s2.2p2.(3P).3d 2F | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Phase Transitions & Allotropes
| Melting point | 54.36 K |
| Boiling point | 90.19 K |
| Critical point (temperature) | 154.58 K |
| Critical point (pressure) | 5.04 MPa |
| Triple point (temperature) | 54.36 K |
| Triple point (pressure) | 0.15 kPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (3)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.3421 |
| 2 | p | 3.5468 |
| 2 | s | 3.5084 |
Crystal Radii Detail (5)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| -2 | II | 121 | ||
| -2 | III | 122 | ||
| -2 | IV | 124 | ||
| -2 | VI | 126 | ||
| -2 | VIII | 128 |
Isotope Decay Modes (22)
| Isotope | Mode | Intensity |
|---|---|---|
| 11 | 2p | 100% |
| 12 | 2p | 100% |
| 13 | B+ | 100% |
| 13 | B+p | 10.9% |
| 14 | B+ | 100% |
| 15 | B+ | 100% |
| 19 | B- | 100% |
| 20 | B- | 100% |
| 21 | B- | 100% |
| 21 | B-n | — |
X‑ray Scattering Factors (502)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.70328 |
| 10.1617 | — | 0.70723 |
| 10.3261 | — | 0.70738 |
| 10.4931 | — | 0.70753 |
| 10.6628 | — | 0.70768 |
| 10.8353 | — | 0.70783 |
| 11.0106 | — | 0.70798 |
| 11.1886 | — | 0.70813 |
| 11.3696 | — | 0.70828 |
| 11.5535 | — | 0.70843 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
4.61×105 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
8.57×105 milligrams per liter
References (1)
Sources
Sources of this element.
Oxygen is the third most abundant element found in the sun, and it plays a part in the carbon-nitrogen cycle, the process once thought to give the sun and stars their energy. Oxygen under excited conditions is responsible for the bright red and yellow-green colors of the Aurora Borealis.
A gaseous element, oxygen forms 21% of the atmosphere by volume and is obtained by liquefaction and fractional distillation. The atmosphere of Mars contains about 0.15% oxygen. The element and its compounds make up 49.2%, by weight, of the earth's crust. About two thirds of the human body and nine tenths of water is oxygen.
In the laboratory it can be prepared by the electrolysis of water or by heating potassium chlorate with manganese dioxide as a catalyst.
References (1)
- [6] Oxygen https://periodic.lanl.gov/8.shtml
Isotopes in Forensic Science and Anthropology
Information on the use of this element's isotopes in forensic science and anthropology.
Measurements of relative 18O abundances have been used to determine the breeding grounds of many species of migrant songbirds. These species of songbirds only grow their feathers before migration, and they grow them on or close to their breeding grounds. Therefore, the isotopic composition of a bird’s feathers correlates to the isotopic signature of the growing season’s precipitation [19] [19] K. A. Hobson. Oecologia120, 314 (1999).[19] K. A. Hobson. Oecologia120, 314 (1999)., [20] [20] K. A. Hobson, L. I. Wassenaar. Oecologia.109, 142 (1996).[20] K. A. Hobson, L. I. Wassenaar. Oecologia.109, 142 (1996)..
Measurements of relative 18O abundances of human hair or nail samples collected at archeological sites have been used to determine the geographic region in which a subject lived based on the oxygen isotopic composition of the water they drank (Fig. IUPAC.8.3). This is possible because hair stores a daily record of oxygen isotopic composition of intake water, which correlates to local meteoric water [92] [92] D. M. O’Brien, M. J. Woller. Rapid Commun. Mass Spectrom.21, 2422 (2007).[92] D. M. O’Brien, M. J. Woller. Rapid Commun. Mass Spectrom.21, 2422 (2007)..
References (7)
- [14] W. Dansgaard. Tellus16, 436 (1964).
- [15] I. D. Clark, P. Fritz. Environmental Isotopes in Hydrogeology, p. 328, Lewis Publishers, New York (1997).
- [19] K. A. Hobson. Oecologia120, 314 (1999).
- [20] K. A. Hobson, L. I. Wassenaar. Oecologia.109, 142 (1996).
- [92] D. M. O’Brien, M. J. Woller. Rapid Commun. Mass Spectrom.21, 2422 (2007).
- [93] I. Fraser, W. Meier-Augenstein, R. M. Kalin. Rapid Commun. Mass Spectrom.20, 1109 (2006).
- [4] IUPAC Periodic Table of the Elements and Isotopes (IPTEI) https://doi.org/10.1515/pac-2015-0703
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Oxygen.
The element property data was retrieved from publications.