Platinum (Pt)
transition-metalSolid
Standard Atomic Weight
195.084 uElectron configuration
[Xe] 6s1 4f14 5d9Melting point
1768.4 °C (2041.55 K)Boiling point
3824.85 °C (4098 K)Density
2.146000e+4 kg/m³Oxidation states
−3, −2, −1, 0, +1, +2, +3, +4, +5, +6Electronegativity (Pauling)
2.28Ionization energy (1st)
Discovery year
1735Atomic radius
135 pmDetails
Platinum is a dense, silvery-white transition metal in group 10 and a member of the platinum-group elements. It is exceptionally resistant to corrosion and oxidation, yet it can catalyze many reactions at its surface. The metal is chemically noble in bulk but forms well-defined complexes, especially in the +2 and +4 oxidation states. Its combination of durability, catalytic activity, and electrical stability gives it technological importance disproportionate to its crustal abundance.
Platinum is a beautiful silvery-white metal, when pure, and is malleable and ductile. It has a coefficient of expansion almost equal to that of soda-lime-silica glass, and is therefore used to make sealed electrodes in glass systems. The metal does not oxidize in air at any temperature, but is corroded by halogens, cyanides, sulfur, and caustic alkalis.
It is insoluble in hydrochloric and nitric acid, but dissolves when they are mixed as aqua regia, forming chloroplatinic acid.
The name derives from the Spanish platina for "silver". In 1735, the Spanish astronomer Antonio de Ulloa found platinum in Peru, South America. In 1741, the English metallurgist Charles Wood found platinum from Colombia, South America. In 1750, the English physician William Brownrigg prepared purified platinum metal.
Used by the pre-Columbian Indians of South America, platinum wasn't noticed by western scientists until 1735. Platinum can occur free in nature and is sometimes found in deposits of gold-bearing sands, primarily those found in the Ural mountains, Columbia and the western United States. Platinum, in the form of the mineral sperrylite (PtAs2), is also obtained as a byproduct of the nickel mining operation in the Sudbury region of Ontario, Canada. Credit for the modern rediscovery of platinum is usually given to Antonio de Ulloa.
Discovered in South America by Ulloa in 1735 and by Wood in 1741. The metal was used by pre-Columbian Indians.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
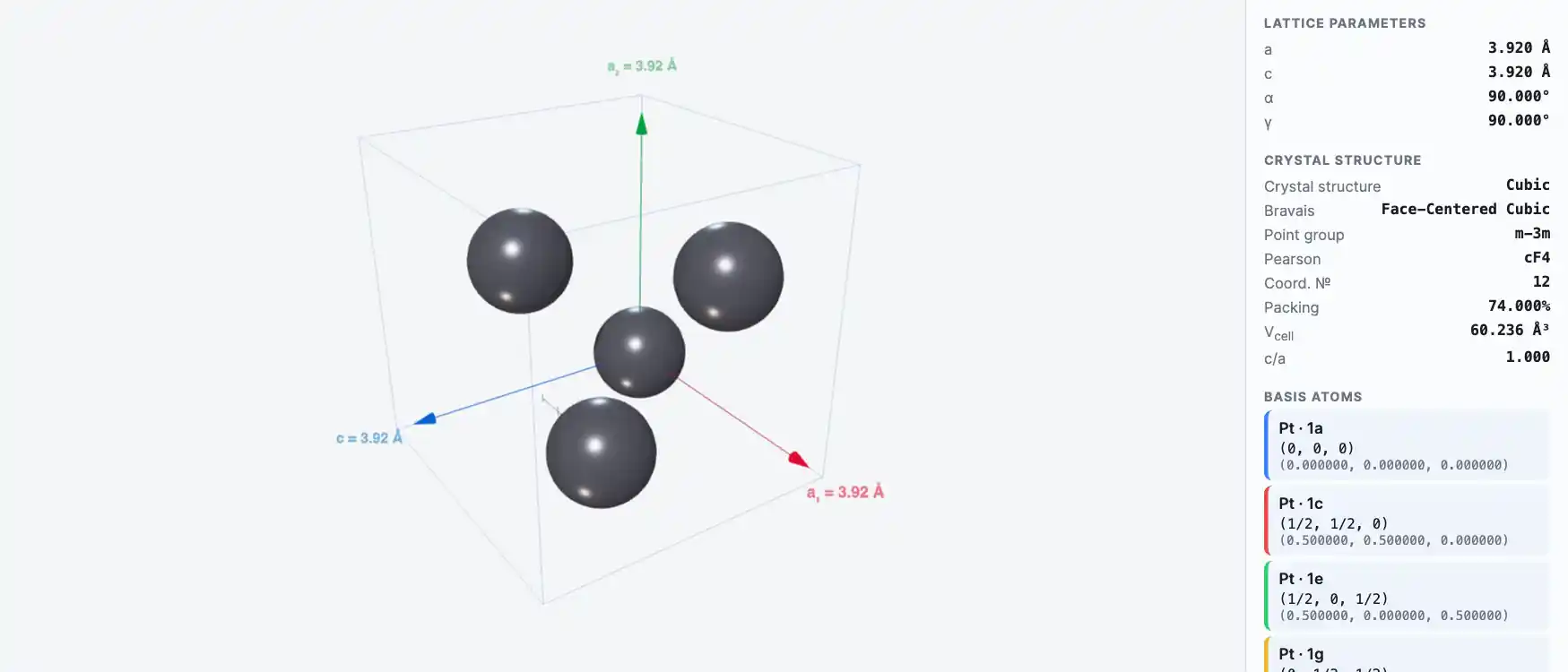
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Pt: 4f¹⁴ 5d⁹ 6s¹[Xe] 4f¹⁴ 5d⁹ 6s¹1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d⁹ 6s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 194 Stable | 193.9626809 ± 0.000001 | 32.8600% | Stable |
| 196 Stable | 195.96495209 ± 0.00000099 | 25.2100% | Stable |
| 198 Stable | 197.9678949 ± 0.0000023 | 7.3560% | Stable |
Phase / State
Reason: 1743.4 °C below melting point (1768.4 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 78 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Pt I | 0 | 995 | 166 | 995 |
| Pt II | +1 | 2268 | 183 | 2268 |
| Pt IV | +3 | 1531 | 1531 | 1531 |
| Pt V | +4 | 1729 | 1729 | 1729 |
| Pt VI | +5 | 1467 | 1467 | 1467 |
| Pt VII | +6 | 786 | 786 | 786 |
| Pt VIII | +7 | 360 | 360 | 360 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Pt I | 0 | 202 |
| Pt II | +1 | 282 |
| Pt III | +2 | 2 |
| Pt IV | +3 | 238 |
| Pt V | +4 | 259 |
| Pt VI | +5 | 251 |
| Pt VII | +6 | 178 |
| Pt VIII | +7 | 80 |
| Pt IX | +8 | 2 |
| Pt X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 4 | N/A | 60 pm |
| +2 | 6 | N/A | 80 pm |
| +4 | 6 | N/A | 62.5 pm |
| +5 | 6 | N/A | 56.99999999999999 pm |
Compounds
Isotopes (3)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 194 Stable | 193.9626809 ± 0.000001 | 32.8600% ± 0.4000% | Stable | stable | |
| 196 Stable | 195.96495209 ± 0.00000099 | 25.2100% ± 0.3400% | Stable | stable | |
| 198 Stable | 197.9678949 ± 0.0000023 | 7.3560% ± 0.1300% | Stable | stable |
Spectral Lines
Showing 50 of 264 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 381.86875 nm | 8300 | Pt I | emission | 5d8.6s2 3F → 5d8.6s.(4F).6p a 5G* | Measured | NIST | |
| 676.00069 nm | 6500 | Pt I | emission | 5d9.6p 3F* → 5d9.7s 3D | Measured | NIST | |
| 530.10143 nm | 3900 | Pt I | emission | 5d8.6s.(4F).6p 5G* → 5d8.(3F<4>).6s.7s.(3S<1>) (4,1) | Measured | NIST | |
| 396.6357 nm | 3400 | Pt I | emission | 5d8.6s2 3F → 5d8.6s.(4F).6p a 5D* | Measured | NIST | |
| 416.45502 nm | 3300 | Pt I | emission | 5d8.6s2 3F → 5d9.6p 3F* | Measured | NIST | |
| 411.86745 nm | 3000 | Pt I | emission | 5d9.6s b 1D → 5d9.6p 3D* | Measured | NIST | |
| 652.34376 nm | 3000 | Pt I | emission | 5d9.6p b 3P* → 5d9.7s 1D | Measured | NIST | |
| 444.25477 nm | 2400 | Pt I | emission | 5d8.6s2 3F → 5d9.6p a 3P* | Measured | NIST | |
| 432.70524 nm | 2300 | Pt I | emission | 5d8.6s.(4F).6p a 5F* → 5d8.(3F<4>).6s.7s.(3S<1>) (4,1) | Measured | NIST | |
| 709.475 nm | 2300 | Pt I | emission | 5d8.6s.(4F).6p 3G* → 5d8.(3F<4>).6s.7s.(1S<0>) (4,0) | Measured | NIST | |
| 711.37244 nm | 2300 | Pt I | emission | 5d8.6s2 3P → 5d9.6p a 3P* | Measured | NIST | |
| 671.03998 nm | 2200 | Pt I | emission | 5d9.6p 3D* → 5d9.7s 1D | Measured | NIST | |
| 522.76459 nm | 2100 | Pt I | emission | 5d9.6s b 1D → 5d9.6p a 3P* | Measured | NIST | |
| 505.94815 nm | 1900 | Pt I | emission | 5d9.6p a 3P* → 5d9.7s 3D | Measured | NIST | |
| 547.57631 nm | 1900 | Pt I | emission | 5d9.6p 3F* → 5d9.7s 3D | Measured | NIST | |
| 584.01269 nm | 1800 | Pt I | emission | 5d8.6s2 3F → 5d9.6p a 3P* | Measured | NIST | |
| 419.24241 nm | 1700 | Pt I | emission | 5d9.6s b 1D → 5d9.6p b 3P* | Measured | NIST | |
| 547.84793 nm | 1500 | Pt I | emission | 5d8.6s.(4F).6p b 5F* → 5d8.(3F<4>).6s.7s.(3S<1>) (4,1) | Measured | NIST | |
| 684.25984 nm | 1500 | Pt I | emission | 5d9.6p 3D* → 5d9.7s 3D | Measured | NIST | |
| 449.8748 nm | 1100 | Pt I | emission | 5d9.6p 3F* → 5d9.6d 3G | Measured | NIST | |
| 539.07754 nm | 1100 | Pt I | emission | 5d9.6p 3F* → 5d9.7s 1D | Measured | NIST | |
| 632.6577 nm | 1100 | Pt I | emission | 5d8.6s2 1G → 5d9.6p 3D* | Measured | NIST | |
| 584.48054 nm | 1000 | Pt I | emission | 5d8.6s.(4F).6p b 5F* → 5d8.(3F<4>).6s.7s.(3S<1>) (4,1) | Measured | NIST | |
| 536.89866 nm | 960 | Pt I | emission | 5d8.6s2 3F → 5d9.6p 3F* | Measured | NIST | |
| 631.83662 nm | 930 | Pt I | emission | 5d9.6p 3P* → 5d9.7s 1D | Measured | NIST | |
| 602.60247 nm | 860 | Pt I | emission | 5d8.6s.(4F).6p 5F* → 5d8.(3F<4>).6s.7s.(3S<1>) (4,1) | Measured | NIST | |
| 695.7507 nm | 800 | Pt I | emission | 5d8.6s.(2F).6p a 3F* → 5d8.(3F<4>).6s.6d b (4,?) | Measured | NIST | |
| 455.24119 nm | 730 | Pt I | emission | 5d8.6s.(4F).6p a 5F* → 5d8.(3F<4>).6s.7s.(3S<1>) (4,1) | Measured | NIST | |
| 428.80508 nm | 680 | Pt I | emission | 5d8.6s2 3F → 5d8.6s.(2D).6p a 3F* | Measured | NIST | |
| 713.16333 nm | 650 | Pt I | emission | 5d8.6s.(2F).6p b 3D* → 5d9.7s 3D | Measured | NIST | |
| 689.67056 nm | 590 | Pt I | emission | 5d8.6s.(2F).6p a 3F* → 5d8.(3F<3>).6s.7s.(3S<1>) (3,1) | Measured | NIST | |
| 409.22515 nm | 580 | Pt I | emission | 5d8.6s.(4F).6p a 5D* → 5d9.6d a 3G | Measured | NIST | |
| 707.8062 nm | 580 | Pt I | emission | 5d8.6s.(4F).6p b 5D* → 5d8.(3F<4>).6s.7s.(3S<1>) (4,1) | Measured | NIST | |
| 401.37143 nm | 570 | Pt II | emission | 5d8.6p 76610* → 5d8.(3P).7s (2,1/2) | Measured | NIST | |
| 628.34779 nm | 570 | Pt I | emission | 5d8.6s.(2F).6p a 3D* → 5d9.7s 3D | Measured | NIST | |
| 683.80564 nm | 560 | Pt I | emission | 5d9.6p 3F* → 5d8.(3F<4>).6s.7s.(3S<1>) (4,1) | Measured | NIST | |
| 452.29919 nm | 550 | Pt I | emission | 5d9.6p 3D* → 5d9.6d 1F | Measured | NIST | |
| 664.83039 nm | 550 | Pt I | emission | 5d9.6p b 3P* → 5d9.7s 3D | Measured | NIST | |
| 703.00606 nm | 540 | Pt I | emission | 5d8.6s.(4P).6p a 5D* → 5d9.7s 3D | Measured | NIST | |
| 448.46871 nm | 520 | Pt I | emission | 5d9.6p 3F* → 5d9.6d 3F | Measured | NIST | |
| 748.60309 nm | 520 | Pt I | emission | 5d8.6s2 1G → 5d8.6s.(4F).6p a 5D* | Measured | NIST | |
| 576.3566 nm | 510 | Pt I | emission | 5d8.6s.(4F).6p a 5D* → 5d9.7s 1D | Measured | NIST | |
| 712.5028 nm | 500 | Pt I | emission | 5d7.(4F).6s2.6p a 3G* → 5d8.(3F<4>).6s.6d b (4,?) | Measured | NIST | |
| 427.3898 nm | 490 | Pt I | emission | 5d8.6s.(2F).6p a 3D* → 5d9.6d b 3G | Measured | NIST | |
| 439.18207 nm | 490 | Pt I | emission | 5d8.6s2 1G → 5d8.6s.(2F).6p a 3D* | Measured | NIST | |
| 392.53348 nm | 480 | Pt I | emission | 5d8.6s2 3F → 5d8.6s.(4F).6p 5G* | Measured | NIST | |
| 712.2889 nm | 480 | Pt I | emission | 5d8.6s.(4P).6p 5D* → 5d9.7d 3P | Measured | NIST | |
| 386.84222 nm | 470 | Pt I | emission | 5d8.6s.(4F).6p b 5F* → 5d8.(3F<4>).6s.6d b (4,?) | Measured | NIST | |
| 387.57161 nm | 470 | Pt I | emission | 5d8.6s.(4F).6p b 5F* → 5d8.(3F<4>).6s.6d b (4,?) | Measured | NIST | |
| 420.12097 nm | 470 | Pt I | emission | 5d9.6p 3P* → 5d9.7s 3D | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 2041.35 K |
| Boiling point | 4098.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (14)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.506 |
| 2 | p | 4.4746 |
| 2 | s | 20.3702 |
| 3 | d | 13.5027 |
| 3 | p | 22.1139 |
| 3 | s | 23.0157 |
| 4 | d | 37.37 |
| 4 | f | 38.494 |
| 4 | p | 35.2696 |
| 4 | s | 34.3612 |
Crystal Radii Detail (4)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | IVSQ | 74 | ||
| 2 | VI | 94 | Ahrens (1952) ionic radius, | |
| 4 | VI | 76.5 | from r^3 vs V plots, | |
| 5 | VI | 71 | estimated, from r^3 vs V plots, |
Isotope Decay Modes (67)
| Isotope | Mode | Intensity |
|---|---|---|
| 165 | A | 100% |
| 166 | A | 100% |
| 167 | A | 100% |
| 168 | A | 100% |
| 168 | B+ | — |
| 169 | A | 100% |
| 169 | B+ | — |
| 170 | A | 100% |
| 170 | B+ | — |
| 171 | A | 86% |
X‑ray Scattering Factors (945)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 0.1 | -0.0027 | 0.00979 |
| 0.13 | -0.0033 | 0.01359 |
| 0.15 | -0.0035 | 0.01538 |
| 0.17 | -0.0037 | 0.01722 |
| 0.2 | -0.0043 | 0.02103 |
| 0.22 | -0.0045 | 0.02279 |
| 0.25 | -0.0051 | 0.02656 |
| 0.28 | -0.0056 | 0.0304 |
| 0.3 | -0.0058 | 0.03194 |
| 0.32 | -0.0058 | 0.03342 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
5×10-3 milligrams per kilogram
References (1)
- [5] Platinum https://education.jlab.org/itselemental/ele078.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
Not Applicable
References (1)
- [5] Platinum https://education.jlab.org/itselemental/ele078.html
Sources
Sources of this element.
Platinum occurs natively, accompanied by small quantities of iridium, osmium, palladium, ruthenium, and rhodium, all belonging to the same group of metals. These are found in the alluvial deposits of the Ural mountains, of Columbia, and of certain western American states. Sperrylite, occurring with the nickel-bearing deposits of Sudbury, Ontario, is the source of a considerable amount of metal.
The large production of nickel makes up for the fact that is only one part of the platinum metals in two million parts of ore.
References (1)
- [6] Platinum https://periodic.lanl.gov/78.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Platinum.
The element property data was retrieved from publications.