Iridium (Ir)
transition-metalSolid
Standard Atomic Weight
192.217 uElectron configuration
[Xe] 6s2 4f14 5d7Melting point
2445.85 °C (2719 K)Boiling point
4427.85 °C (4701 K)Density
2.256220e+4 kg/m³Oxidation states
−3, −2, −1, +1, +2, +3, +4, +5, +6, +7, +8, +9Electronegativity (Pauling)
2.2Ionization energy (1st)
Discovery year
1803Atomic radius
135 pmDetails
Iridium is a very dense platinum-group transition metal with exceptional resistance to corrosion and high-temperature attack. It occurs naturally mainly with platinum-group minerals and in nickel-copper sulfide ores. Chemically it forms robust complexes, especially in oxidation states +3 and +4, and it is notable for the global iridium anomaly associated with the Cretaceous-Paleogene boundary impact layer.
Iridium, a metal of the platinum family, is white (similar to platinum) but with a slight yellowish cast. Because iridium is very hard and brittle, it is hard to machine, form, or work.
It is the most corrosion-resistant metal known, and was used in making the standard meter bar of Paris, which is a 90 percent platinum and 10 percent iridium alloy. This meter bar was replaced in 1960 as a fundamental unit of length (see Krypton).
Iridium is not attacked by any of the acids nor by aqua regia, but is attacked by molten salts, such as NaCl and NaCN. The specific gravity of iridium is to osmium's specific gravity. Calculations of the densities of iridium and osmium from the space lattices give values of 22.65 and 22.61 g/cm^3, respectively. These values may be more reliable than actual physical measurements for determining which element is heavier.
The name derives from the Latin Iris, the Greek goddess of rainbows, because of the variety of colours in the element's salt solutions. Iridium and osmium were both discovered in a crude platinum ore in 1803 by the English chemist Smithson Tennant. Iridium was discovered independently by the French chemist H. V. Collet-Descotils, who actually published his paper one month before Tennant, but Tennant is given credit for the discovery, perhaps because he alone also found osmium in the ore.
Iridium and osmium were discovered at the same time by the British chemist Smithson Tennant in 1803. Iridium and osmium were identified in the black residue remaining after dissolving platinum ore with aqua regia, a mixture of 25% nitric acid (HNO3) and 75% hydrochloric acid (HCl). Today, iridium is still obtained from platinum ores and as a by-product of mining nickel.
From the Latin word iris meaning rainbow. Tennant discovered iridium in 1803 in the residue left when crude platinum is dissolved by aqua regia. The name iridium is appropriate because its salts are highly colored.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
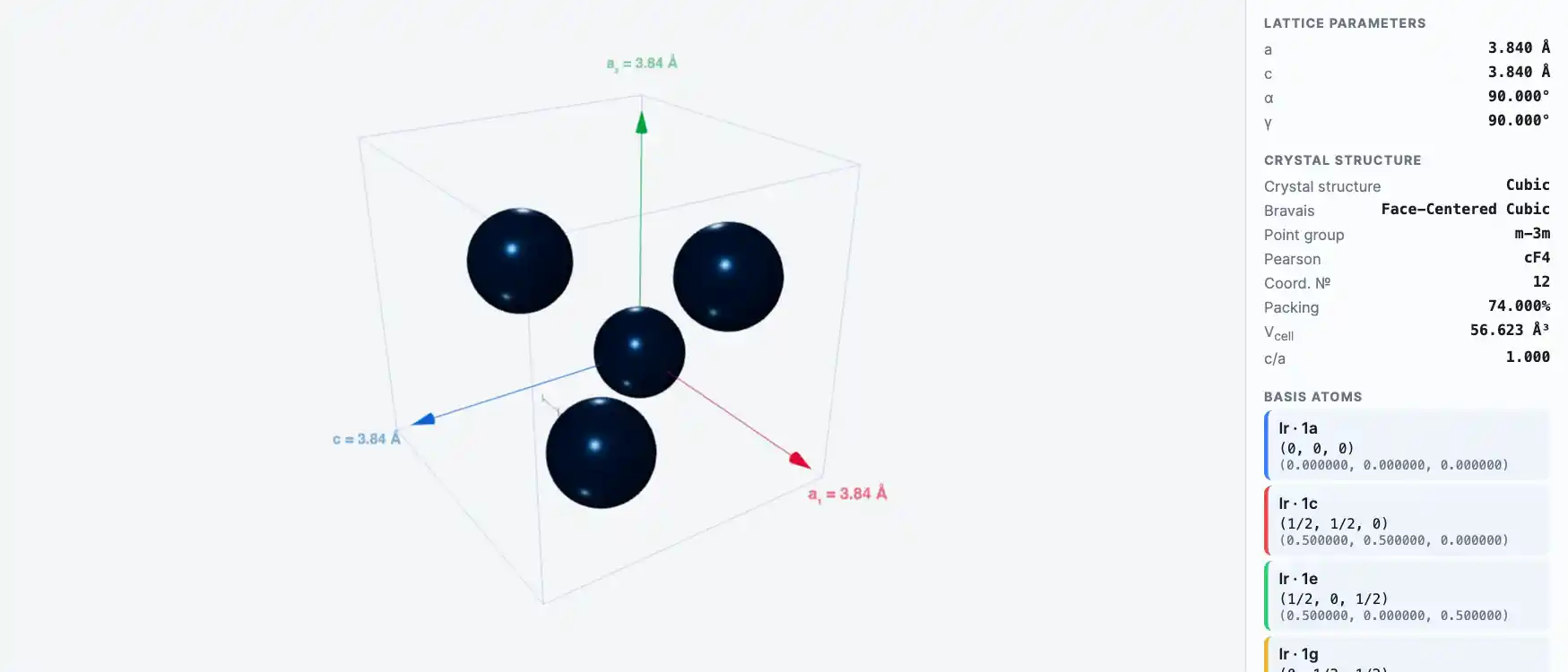
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Ir: 4f¹⁴ 5d⁷ 6s²[Xe] 4f¹⁴ 5d⁷ 6s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d⁷ 6s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 191 Stable | 190.9605893 ± 0.0000021 | 37.3000% | Stable |
| 193 Stable | 192.9629216 ± 0.0000021 | 62.7000% | Stable |
Phase / State
Reason: 2420.8 °C below melting point (2445.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 77 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Ir I | 0 | 402 | 70 | 398 |
| Ir II | +1 | 473 | 129 | 473 |
| Ir IV | +3 | 1374 | 1374 | 1374 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Ir I | 0 | 231 |
| Ir II | +1 | 76 |
| Ir III | +2 | 2 |
| Ir IV | +3 | 224 |
| Ir V | +4 | 2 |
| Ir VI | +5 | 2 |
| Ir VII | +6 | 2 |
| Ir VIII | +7 | 2 |
| Ir IX | +8 | 2 |
| Ir X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 68 pm |
| +4 | 6 | N/A | 62.5 pm |
| +5 | 6 | N/A | 56.99999999999999 pm |
Compounds
Isotopes (2)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 191 Stable | 190.9605893 ± 0.0000021 | 37.3000% ± 0.2000% | Stable | stable | |
| 193 Stable | 192.9629216 ± 0.0000021 | 62.7000% ± 0.2000% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 382.7577 nm | 58 | Ir II | emission | 5d7.(2D2).6s 3D → 5d7.(4F<5/2>).6p (5/2,1/2)* | Measured | NIST | |
| 384.593 nm | N/A | Ir II | emission | 5d7.(2G).6s 1G → 5d7.(4F<9/2>).6p (9/2,3/2)* | Measured | NIST | |
| 387.3624 nm | 9 | Ir II | emission | 5d6.6s2 5D → 5d7.(4P<5/2>).6p (5/2,1/2)* | Measured | NIST | |
| 389.558 nm | N/A | Ir II | emission | 5d6.6s2 5D → 5d7.(4F<7/2>).6p (7/2,1/2)* | Measured | NIST | |
| 395.1973 nm | N/A | Ir II | emission | 5d6.6s2 5D → 5d7.(4F<9/2>).6p (9/2,1/2)* | Measured | NIST | |
| 395.2882 nm | 8 | Ir II | emission | 5d7.(2H).6s 3H → 5d6.6s.(6D<9/2>).6p (9/2,1/2)* | Measured | NIST | |
| 397.882 nm | 6 | Ir II | emission | 5d7.(2F).6s 3F → 5664* | Measured | NIST | |
| 398.6377 nm | 5 | Ir II | emission | 5d6.6s2 5D → 5d7.(4F<3/2>).6p (3/2,1/2)* | Measured | NIST | |
| 399.0389 nm | 6 | Ir II | emission | 5d6.6s2 3H → 6197* | Measured | NIST | |
| 400.1961 nm | 12 | Ir II | emission | 5d7.(2D2).6s 3D → 5d7.(4P<5/2>).6p (5/2,1/2)* | Measured | NIST | |
| 402.5321 nm | 4 | Ir II | emission | 5d7.(2F).6s 3F → 5d6.6s.(6D<5/2>).6p (5/2,1/2)* | Measured | NIST | |
| 402.5399 nm | 29 | Ir II | emission | 5d7.(2D2).6s 3D → 5d7.(4F<7/2>).6p (7/2,1/2)* | Measured | NIST | |
| 404.1381 nm | 45 | Ir II | emission | 5d7.(2H).6s 3H → 5d7.(4F<9/2>).6p (9/2,1/2)* | Measured | NIST | |
| 404.4911 nm | 7 | Ir II | emission | 5d7.(2F).6s 3F → 5d7.(4F<9/2>).6p (9/2,3/2)* | Measured | NIST | |
| 410.8315 nm | 48 | Ir II | emission | 5d7.(2F).6s 3F → 5d7.(4F<5/2>).6p (5/2,1/2)* | Measured | NIST | |
| 411.7209 nm | 3 | Ir II | emission | 5d7.(2G).6s 3G → 5d7.(4F<9/2>).6p (9/2,1/2)* | Measured | NIST | |
| 412.8911 nm | 17 | Ir II | emission | 5d7.(2P).6s 3P → 5d7.(4P<1/2>).6p (1/2,1/2)* | Measured | NIST | |
| 413.91 nm | 21 | Ir II | emission | 5d7.(2P).6s 3P → 5d7.(4F<9/2>).6p (9/2,3/2)* | Measured | NIST | |
| 439.0196 nm | 4 | Ir II | emission | 5d6.6s2 5D → 5d7.(4P<5/2>).6p (5/2,1/2)* | Measured | NIST | |
| 443.3888 nm | N/A | Ir II | emission | 5d7.(2F).6s 3F → 5d6.6s.(6D<7/2>).6p (7/2,1/2)* | Measured | NIST | |
| 454.5672 nm | N/A | Ir II | emission | 5d7.(2H).6s 3H → 5d7.(4F<9/2>).6p (9/2,1/2)* | Measured | NIST | |
| 461.1752 nm | N/A | Ir II | emission | 5d7.(2F).6s 3F → 5d7.(4P<5/2>).6p (5/2,1/2)* | Measured | NIST | |
| 467.5844 nm | 5 | Ir II | emission | 5d7.(2G).6s 3G → 5d7.(4F<9/2>).6p (9/2,1/2)* | Measured | NIST | |
| 479.5262 nm | N/A | Ir II | emission | 5d6.6s2 5D → 5d7.(4F<9/2>).6p (9/2,1/2)* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 2719.15 K |
| Boiling point | 4701.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (14)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.4881 |
| 2 | p | 4.4624 |
| 2 | s | 20.1102 |
| 3 | d | 13.514 |
| 3 | p | 21.9311 |
| 3 | s | 22.7942 |
| 4 | d | 37.2628 |
| 4 | f | 38.6552 |
| 4 | p | 35.086 |
| 4 | s | 34.152 |
Crystal Radii Detail (3)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 82 | estimated, | |
| 4 | VI | 76.5 | from r^3 vs V plots, | |
| 5 | VI | 71 | estimated, from metallic oxides, |
Isotope Decay Modes (64)
| Isotope | Mode | Intensity |
|---|---|---|
| 163 | p | — |
| 164 | p | — |
| 164 | A | — |
| 164 | B+ | — |
| 165 | p | — |
| 165 | A | — |
| 166 | A | 93% |
| 166 | p | 7% |
| 167 | A | 43.5% |
| 167 | p | 38.6% |
X‑ray Scattering Factors (515)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 2.22753 |
| 10.1617 | — | 2.30683 |
| 10.3261 | — | 2.38895 |
| 10.4931 | — | 2.474 |
| 10.6628 | — | 2.56207 |
| 10.8353 | — | 2.65417 |
| 11.0106 | — | 2.75003 |
| 11.1886 | — | 2.84935 |
| 11.3696 | — | 2.94781 |
| 11.5535 | — | 3.0118 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1×10-3 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
Not Applicable
References (1)
Sources
Sources of this element.
Iridium occurs uncombined in nature with platinum and other metals of this family in alluvial deposits. It is recovered as a by-product from the nickel mining industry.
References (1)
- [6] Iridium https://periodic.lanl.gov/77.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Iridium.
The element property data was retrieved from publications.