Rhenium (Re)
transition-metalSolid
Standard Atomic Weight
186.207 uElectron configuration
[Xe] 6s2 4f14 5d5Melting point
3185.85 °C (3459 K)Boiling point
5595.85 °C (5869 K)Density
2.080000e+4 kg/m³Oxidation states
−3, −1, 0, +1, +2, +3, +4, +5, +6, +7Electronegativity (Pauling)
1.9Ionization energy (1st)
Discovery year
1925Atomic radius
135 pmDetails
Rhenium is a very dense, high-melting transition metal in group 7, chemically related to manganese and technetium but far less abundant in the crust. It is notable for retaining strength at extreme temperature and for forming stable high oxidation states, especially +7. Natural rhenium occurs mainly as a trace substitute in molybdenite rather than as separate ores, making it a by-product metal of copper-molybdenum processing.
The element is silvery white with a metallic luster; its density is exceeded only by that of platinum, iridium, and osmium, and its melting point is exceeded only by that of tungsten and carbon.
The usual commercial form of the element is powder, but it can be consolidated by pressing and resistance-sintering in a vacuum or hydrogen atmosphere. This process produces a compact shape in excess of 90 percent of the density of the metal.
Annealed rhenium is very ductile, and can be bent, coiled, or rolled. Rhenium is used as an additive to tungsten and molybdenum -based alloys to impart useful properties.
The name derives from the Latin rhenus for the Rhine river in Germany. Rhenium was discovered by x-ray spectroscopy in 1925 by German chemists Walter Noddack, Ida Tacke, and Otto Berg.
Rhenium was discovered by the German chemists Ida Tacke-Noddack, Walter Noddack and Otto Carl Berg in 1925. They detected rhenium spectroscopically in platinum ores and in the minerals columbite ((Fe, Mn, Mg)(Nb, Ta)2O6), gadolinite ((Ce, La, Nd, Y)2FeBe2Si2O10) and molybdenite (MoS2). Rhenium is present in these materials only in trace amounts. In 1928, Noddack and Berg were able to extract 1 gram of rhenium from 660 kilograms of molybdenite. Today, rhenium is obtained as a byproduct of refining molybdenum and copper.
Discovery of rhenium is generally attributed to Noddack, Tacke, and Berg, who announced in 1925 they had detected the element in platinum ore and columbite. They also found the element in gadolinite and molybdenite. By working up 660 kg of molybdenite in 1928 they were able to extract 1 g of rhenium.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
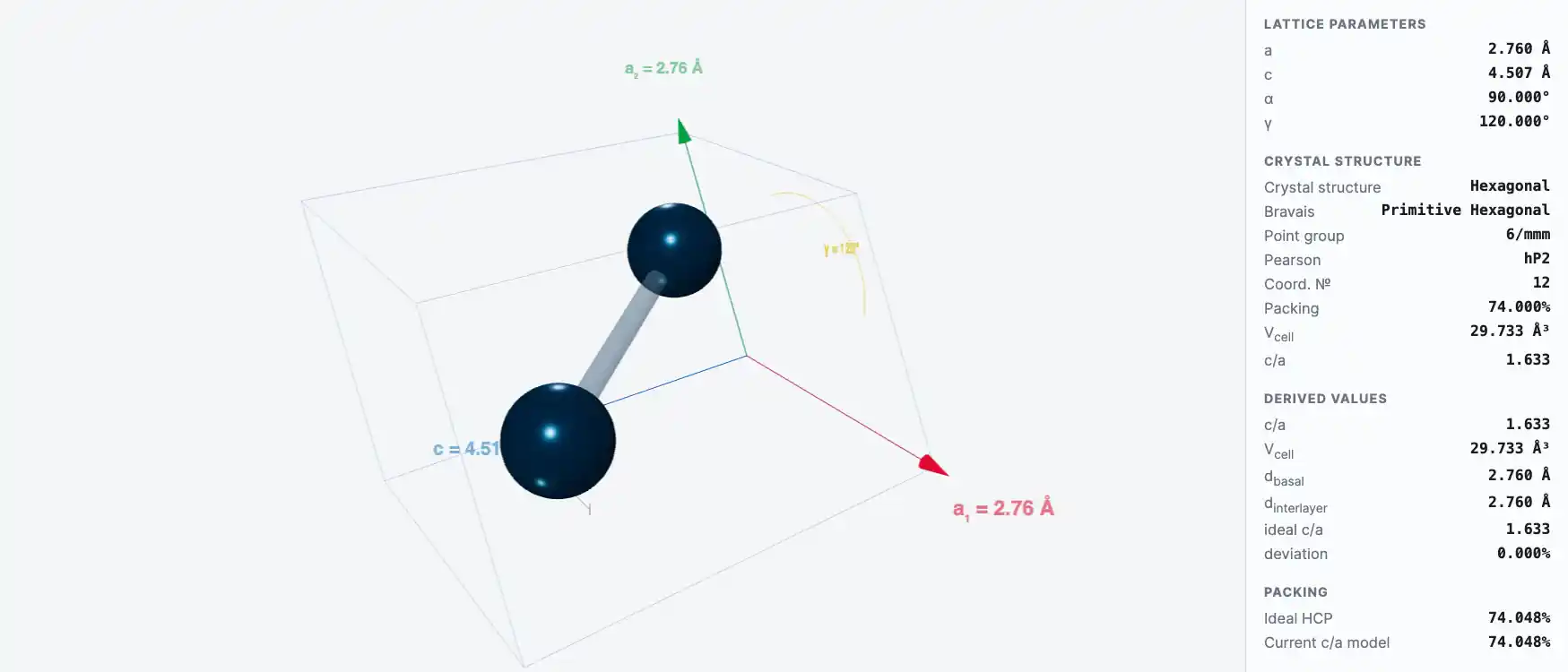
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Re: 4f¹⁴ 5d⁵ 6s²[Xe] 4f¹⁴ 5d⁵ 6s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d⁵ 6s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 185 Stable | 184.9529545 ± 0.0000013 | 37.4000% | Stable |
Phase / State
Reason: 3160.8 °C below melting point (3185.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 75 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Re I | 0 | 432 | 0 | 0 |
| Re II | +1 | 56 | 0 | 0 |
| Re III | +2 | 1381 | 1381 | 1381 |
| Re IV | +3 | 982 | 982 | 982 |
| Re V | +4 | 401 | 401 | 401 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Re I | 0 | 291 |
| Re II | +1 | 140 |
| Re III | +2 | 232 |
| Re IV | +3 | 162 |
| Re V | +4 | 80 |
| Re VI | +5 | 2 |
| Re VII | +6 | 2 |
| Re VIII | +7 | 2 |
| Re IX | +8 | 2 |
| Re X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +4 | 6 | N/A | 63 pm |
| +5 | 6 | N/A | 57.99999999999999 pm |
| +6 | 6 | N/A | 55.00000000000001 pm |
| +7 | 4 | N/A | 38 pm |
| +7 | 6 | N/A | 53 pm |
Compounds
Isotopes (1)
Natural rhenium is a mixture of two stable isotopes. Twenty six other unstable isotopes are recognized.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 185 Stable | 184.9529545 ± 0.0000013 | 37.4000% ± 0.0200% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 3458.15 K |
| Boiling point | 5863.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (14)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.4522 |
| 2 | p | 4.438 |
| 2 | s | 19.5902 |
| 3 | d | 13.5453 |
| 3 | p | 21.5655 |
| 3 | s | 22.3515 |
| 4 | d | 36.9456 |
| 4 | f | 39.0752 |
| 4 | p | 34.6268 |
| 4 | s | 33.6436 |
Crystal Radii Detail (5)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 4 | VI | 77 | from r^3 vs V plots, from metallic oxides, | |
| 5 | VI | 72 | estimated, | |
| 6 | VI | 69 | estimated, | |
| 7 | IV | 52 | ||
| 7 | VI | 67 |
Isotope Decay Modes (54)
| Isotope | Mode | Intensity |
|---|---|---|
| 159 | p | — |
| 159 | A | — |
| 160 | p | 89% |
| 160 | A | 11% |
| 161 | p | 100% |
| 161 | A | — |
| 162 | A | 94% |
| 162 | B+ | — |
| 163 | B+ | — |
| 163 | A | 32% |
X‑ray Scattering Factors (516)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.8209 |
| 10.1617 | — | 1.91145 |
| 10.3261 | — | 2.0065 |
| 10.4931 | — | 2.10629 |
| 10.6628 | — | 2.21103 |
| 10.8353 | — | 2.28753 |
| 11.0106 | — | 2.3602 |
| 11.1886 | — | 2.43518 |
| 11.3696 | — | 2.51255 |
| 11.5535 | — | 2.59237 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
7×10-4 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
4×10-6 milligrams per liter
References (1)
Sources
Sources of this element.
Rhenium does not occur free in nature or as a compound in a distinct mineral species. It is, however, widely spread throughout the earth's crust to the extent of about 0.001 ppm. Commercial rhenium in the U.S. today is obtained from molybdenum roaster-flue dusts obtained from copper-sulfide ores mined in the vicinity of Miami, Arizona and elsewhere in Arizona and in Utah.
Some molybdenum contains from 0.002% to 0.2% rhenium. More than 150,000 troy ounces of rhenium are now being produced yearly in the United States. The total estimated Free World reserve of rhenium metal is 3500 tons. Rhenium metal is prepared by reducing ammonium perrhentate with hydrogen at elevated temperatures.
References (1)
- [6] Rhenium https://periodic.lanl.gov/75.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Rhenium.
The element property data was retrieved from publications.