Tungsten (W)
transition-metalSolid
Standard Atomic Weight
183.84 uElectron configuration
[Xe] 6s2 4f14 5d4Melting point
3421.85 °C (3695 K)Boiling point
5554.85 °C (5828 K)Density
1.930000e+4 kg/m³Oxidation states
−4, −2, −1, 0, +1, +2, +3, +4, +5, +6Electronegativity (Pauling)
2.36Ionization energy (1st)
Discovery year
1781Atomic radius
135 pmDetails
Tungsten is a dense, refractory transition metal in group 6. It has the highest melting point of any element and retains strength at temperatures where most engineering metals soften. Chemically it is best known for stable high oxidation states, especially +6, and for forming hard carbides and complex oxoanions. Natural tungsten occurs mainly in tungstate minerals rather than as the native metal.
Pure tungsten is a steel-gray to tin-white metal. Very pure tungsten can be cut with a hacksaw, forged, spun, drawn, and extruded. The impure metal is brittle and can be worked only with difficulty. Tungsten has the highest melting point of all metals, and at temperatures over 1650°C has the highest tensile strength. The metal oxidizes in air and must be protected at elevated temperatures. It has excellent corrosion resistance and is attacked only slightly by most mineral acids. The thermal expansion is about the same as borosilicate glass, which makes the metal useful for glass-to-metal seals.
The name derives from the Swedish tungsten for "heavy stone". The symbol W derives from the German wolfram, which was found with tin and interfered with the smelting of tin. It was said to eat up tin like a wolf eats up sheep. The element was discovered by the Swedish pharmacist and chemist Carl-Wilhelm Scheele in 1781. Tungsten metal was first isolated by the Spanish chemists Fausto Elhuyar and his brother Juan José in 1783.
Tungsten was discovered by Juan José and Fausto Elhuyar, Spanish chemists and brothers, in 1783 in samples of the mineral wolframite ((Fe, Mn)WO4). Today, tungsten is primarily obtained from wolframite and scheelite (CaWO4) using the same basic method developed by José and Elhuyar. Tungsten ores are crushed, cleaned and treated with alkalis to form tungsten trioxide (WO3). Tungsten trioxide is then heated with carbon or hydrogen gas (H2), forming tungsten metal and carbon dioxide (CO2) or tungsten metal and water vapor (H2O).
From Swedish, tung sten meanig heavy stone. In 1779 Peter Woulfe examined the mineral now known as wolframite and concluded it must contain a new substance. Scheele, in 1781, found that a new acid could be made from tungsten (a name first applied about 1758 to a mineral now known as scheelite). Scheele and Berman suggested the possibility of obtaining a new metal by reducing this acid. The de Elhuyar brothers found acid in wolframite in 1783 that was identical to the acid of tungsten (tungstic acid) of Scheele, and in that year they succeeded in obtaining the element by reduction of this acid with charcoal. Tungsten occurs in wolframite, scheelite, huebnertie, and ferberite. Important deposits of tungsten occur in California, Colorado, South Korea, Bolivia, Russia, and Portugal. China is reported to have about 75% of the world's tungsten resources. Natural tungsten contains five stable isotopes. Twenty one other unstable isotopes are recognized. The metal is obtained commercially be reducing tungsten oxide with hydrogen or carbon.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
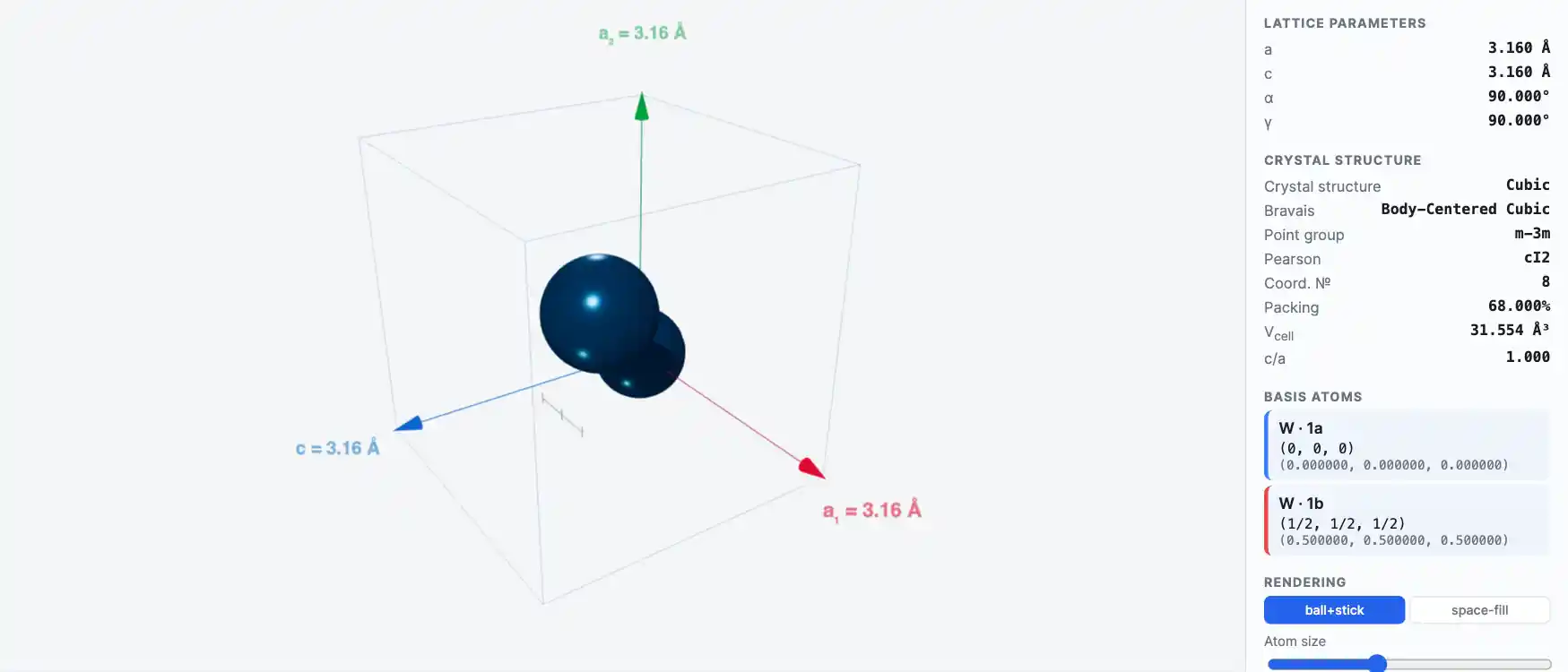
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
W: 4f¹⁴ 5d⁴ 6s²[Xe] 4f¹⁴ 5d⁴ 6s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d⁴ 6s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
No stable isotopes.
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 183 Radioactive | 182.95022275 ± 0.0000009 | 14.3100% | 670 Ey |
| 161 Radioactive | 160.9672 ± 0.00021 | N/A | 409 ms |
| 157 Radioactive | 156.97884 ± 0.00043 | N/A | 275 ms |
| 177 Radioactive | 176.946643 ± 0.00003 | N/A | 132.4 minutes |
| 181 Radioactive | 180.9481978 ± 0.0000051 | N/A | 120.956 days |
Phase / State
Reason: 3396.8 °C below melting point (3421.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 74 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| W I | 0 | 7049 | 522 | 5852 |
| W II | +1 | 2838 | 211 | 2838 |
| W III | +2 | 2644 | 37 | 2644 |
| W IV | +3 | 791 | 0 | 791 |
| W V | +4 | 193 | 0 | 193 |
| W VI | +5 | 17 | 0 | 17 |
| W VII | +6 | 397 | 0 | 397 |
| W VIII | +7 | 193 | 187 | 193 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| W I | 0 | 509 |
| W II | +1 | 264 |
| W III | +2 | 236 |
| W IV | +3 | 106 |
| W V | +4 | 60 |
| W VI | +5 | 15 |
| W VII | +6 | 113 |
| W VIII | +7 | 103 |
| W IX | +8 | 3 |
| W X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +4 | 6 | N/A | 66 pm |
| +5 | 6 | N/A | 62 pm |
| +6 | 4 | N/A | 42 pm |
| +6 | 5 | N/A | 51 pm |
| +6 | 6 | N/A | 60 pm |
Compounds
Isotopes (5)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 183 Radioactive | 182.95022275 ± 0.0000009 | 14.3100% ± 0.0400% | 670 Ey | IS =14.31±0.4%α ? | |
| 161 Radioactive | 160.9672 ± 0.00021 | N/A | 409 ms | α =73±0.3%β+ =27±0.3% | |
| 157 Radioactive | 156.97884 ± 0.00043 | N/A | 275 ms | β+ =100%α =0% | |
| 177 Radioactive | 176.946643 ± 0.00003 | N/A | 132.4 minutes | β+ =100% | |
| 181 Radioactive | 180.9481978 ± 0.0000051 | N/A | 120.956 days | ε =100% |
Spectral Lines
Showing 50 of 2460 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 400.8749 nm | 1000 | W I | emission | 5d5.(6S).6s 7S → 5d5.(6S).6p 7P* | Measured | NIST | |
| 429.4605 nm | 800 | W I | emission | 5d5.(6S).6s 7S → 5d5.(6S).6p 7P* | Measured | NIST | |
| 386.7982 nm | 600 | W I | emission | 5d5.(6S).6s 7S → 5d4.6s.(6D).6p 7D* | Measured | NIST | |
| 407.4357 nm | 600 | W I | emission | 5d5.(6S).6s 7S → 5d5.(6S).6p 7P* | Measured | NIST | |
| 381.7484 nm | 400 | W I | emission | 5d5.(6S).6s 7S → 5d4.6s.(6D).6p 5F* | Measured | NIST | |
| 484.381 nm | 400 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 7D* | Measured | NIST | |
| 505.328 nm | 400 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 7D* | Measured | NIST | |
| 384.6213 nm | 300 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 5F* | Measured | NIST | |
| 525.9338 nm | 300 | W I | emission | 5d5.(6S).6p 7P* → 5d4.6s.(6D).7s 7D | Measured | NIST | |
| 551.4684 nm | 300 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 7D* | Measured | NIST | |
| 383.5052 nm | 250 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 5P* | Measured | NIST | |
| 388.1394 nm | 250 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 5P* | Measured | NIST | |
| 522.4661 nm | 250 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 7D* | Measured | NIST | |
| 524.2973 nm | 250 | W I | emission | 5d4.6s2 3G → * | Measured | NIST | |
| 424.4367 nm | 200 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 7D* | Measured | NIST | |
| 426.9384 nm | 200 | W I | emission | 5d5.(6S).6s 7S → * | Measured | NIST | |
| 430.2103 nm | 200 | W I | emission | 5d5.(6S).6s 7S → 5d4.6s.(6D).6p 7D* | Measured | NIST | |
| 488.6902 nm | 200 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 7F* | Measured | NIST | |
| 498.2586 nm | 200 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 7F* | Measured | NIST | |
| 380.9234 nm | 150 | W I | emission | 5d5.(6S).6s 7S → 5d4.6s.(6D).6p 5D* | Measured | NIST | |
| 384.749 nm | 150 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 5F* | Measured | NIST | |
| 505.4594 nm | 150 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 7F* | Measured | NIST | |
| 507.1736 nm | 150 | W I | emission | 5d4.6s.(6D).6p 7F* → 5d4.6s.(6D).7s 7D | Measured | NIST | |
| 523.352 nm | 150 | W I | emission | 5d4.6s2 3P2 → * | Measured | NIST | |
| 527.5538 nm | 150 | W I | emission | 5d5.(4G).6s 5G → * | Measured | NIST | |
| 549.2315 nm | 150 | W I | emission | 5d4.6s.(6D).6p 7D* → 5d4.6s.(6D).7s 7D | Measured | NIST | |
| 381.0796 nm | 120 | W I | emission | 5d4.6s2 3F2 → * | Measured | NIST | |
| 506.9123 nm | 120 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 7F* | Measured | NIST | |
| 525.5401 nm | 120 | W I | emission | 5d5.(4D).6s 5D → * | Measured | NIST | |
| 434.811303 nm | 109 | W II | emission | 5d4.(5D).6s 4D | Measured | NIST | |
| 381.0385 nm | 100 | W I | emission | 5d5.(4G).6s 5G → * | Measured | NIST | |
| 401.5216 nm | 100 | W I | emission | 5d5.(4G).6s 5G → * | Measured | NIST | |
| 404.56 nm | 100 | W I | emission | 5d5.(6S).6s 7S → 5d4.6s.(6D).6p 5F* | Measured | NIST | |
| 410.2701 nm | 100 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 5P* | Measured | NIST | |
| 424.1444 nm | 100 | W I | emission | 5d4.6s2 3D → * | Measured | NIST | |
| 427.4553 nm | 100 | W I | emission | 5d4.6s.(6D).6p 7F* → 5d4.6s.(6D).7s 7D | Measured | NIST | |
| 525.4544 nm | 100 | W I | emission | 5d4.6s2 3D → * | Measured | NIST | |
| 526.3195 nm | 100 | W I | emission | 5d5.(4D).6s 5D → * | Measured | NIST | |
| 526.9315 nm | 100 | W I | emission | 5d4.6s2 3F2 → * | Measured | NIST | |
| 543.5042 nm | 100 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 7F* | Measured | NIST | |
| 395.105951 nm | 91 | W II | emission | 5d4.(5D).6s 4D → 5d3.(4F).6s.(5F).6p 6G* | Measured | NIST | |
| 406.9948 nm | 80 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 5P* | Measured | NIST | |
| 413.7464 nm | 80 | W I | emission | 5d4.6s2 5D → 5d5.(6S).6p 7P* | Measured | NIST | |
| 421.9375 nm | 80 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 5D* | Measured | NIST | |
| 425.9363 nm | 80 | W I | emission | 5d4.6s.(6D).6p 7F* → 5d4.6s.(6D).7s 7D | Measured | NIST | |
| 468.0513 nm | 80 | W I | emission | 5d4.6s2 5D → 5d4.6s.(6D).6p 7D* | Measured | NIST | |
| 498.6924 nm | 80 | W I | emission | 5d4.6s2 3H → * | Measured | NIST | |
| 526.8545 nm | 80 | W I | emission | 5d4.6s2 3F2 → * | Measured | NIST | |
| 547.7798 nm | 80 | W I | emission | 5d4.6s2 3P2 → 5d4.6s.(6D).6p 5D* | Measured | NIST | |
| 667.838 nm | 80 | W I | emission | 5d5.(4G).6s 5G → * | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 3687.15 K |
| Boiling point | 5828.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (14)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.4343 |
| 2 | p | 4.4258 |
| 2 | s | 19.3302 |
| 3 | d | 13.5476 |
| 3 | p | 21.3824 |
| 3 | s | 22.13 |
| 4 | d | 36.8268 |
| 4 | f | 39.2892 |
| 4 | p | 34.4516 |
| 4 | s | 33.4412 |
Crystal Radii Detail (5)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 4 | VI | 80 | from r^3 vs V plots, from metallic oxides, | |
| 5 | VI | 76 | from r^3 vs V plots, | |
| 6 | IV | 56 | ||
| 6 | V | 65 | ||
| 6 | VI | 74 |
Isotope Decay Modes (54)
| Isotope | Mode | Intensity |
|---|---|---|
| 157 | B+ | 100% |
| 157 | A | 0% |
| 158 | A | 100% |
| 159 | A | 100% |
| 159 | B+ | — |
| 160 | A | 87% |
| 160 | B+ | — |
| 161 | A | 73% |
| 161 | B+ | 27% |
| 162 | B+ | — |
X‑ray Scattering Factors (541)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.92551 |
| 10.1617 | — | 2.00949 |
| 10.3261 | — | 2.09714 |
| 10.4931 | — | 2.18428 |
| 10.6628 | — | 2.26758 |
| 10.8353 | — | 2.35405 |
| 11.0105 | — | 2.44381 |
| 11.1886 | — | 2.537 |
| 11.3696 | — | 2.63375 |
| 11.5535 | — | 2.73418 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.25 milligrams per kilogram
References (1)
- [5] Tungsten https://education.jlab.org/itselemental/ele074.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1×10-4 milligrams per liter
References (1)
- [5] Tungsten https://education.jlab.org/itselemental/ele074.html
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Tungsten.
The element property data was retrieved from publications.