Lanthanum (La)
lanthanideSolid
Standard Atomic Weight
138.90547 uElectron configuration
[Xe] 6s2 5d1Melting point
917.85 °C (1191 K)Boiling point
3463.85 °C (3737 K)Density
6150 kg/m³Oxidation states
0, +1, +2, +3Electronegativity (Pauling)
1.1Ionization energy (1st)
Discovery year
1839Atomic radius
195 pmDetails
Lanthanum is the first element of the lanthanide series by common convention, although its 4f shell is empty in the neutral atom. It is a soft, reactive rare-earth metal that occurs with other light rare earths in minerals such as monazite and bastnäsite. Its chemistry is dominated by the large La³⁺ ion, which gives mostly colorless, strongly ionic compounds. Lanthanum is important in optical glass, catalysts, battery alloys, and high-temperature ceramic materials.
Lanthanum is silvery white, malleable, ductile, and soft enough to be cut with a knife. It is one of the most reactive of the rare-earth metals. It oxidizes rapidly when exposed to air. Cold water attacks lanthanum slowly, while hot water attacks it much more rapidly.
The metal reacts directly with elemental carbon, nitrogen, boron, selenium, silicon, phosphorus, sulfur, and with halogens.
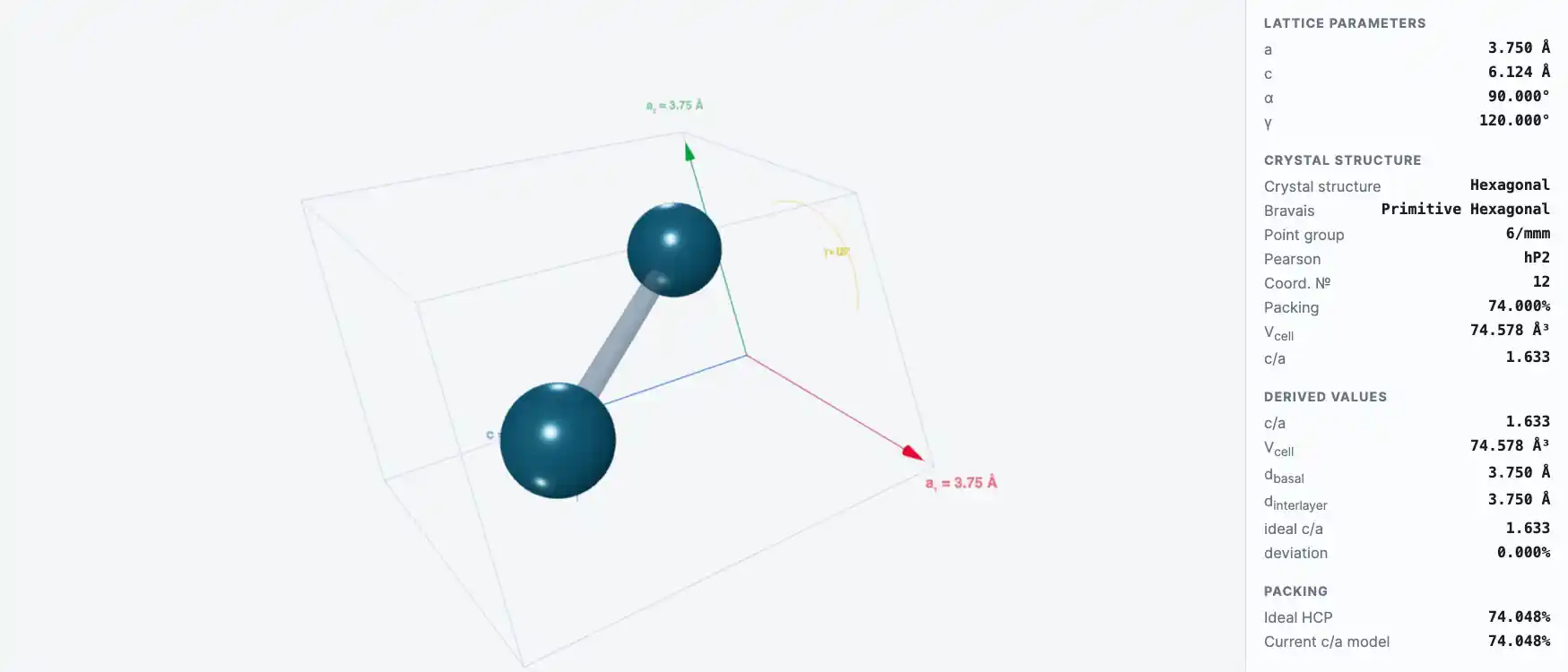
At 310°C, lanthanum changes from a hexagonal to a face-centered cubic structure, and at 865°C it again transforms into a body-centered cubic structure.
The name derives from the Greek lanthanein for "to be hidden" or "to escape notice" because it hid in cerium ore and was difficult to separate from that rare earth mineral. Lanthanum was discovered by the Swedish surgeon and chemist Carl-Gustav Mosander in 1839. In 1842, Mosander separated his lanthanium sample into two oxides; for one of these he retained the name lanthanum and for the other he gave the name didymium (or twin).
Lanthanum was discovered by Carl Gustaf Mosander, a Swedish chemist, in 1839. Mosander was searching for impurities he believed existed within samples of cerium. He treated cerium nitrate (Ce(NO3)3) with dilute nitric acid (HNO3) and found a new substance he named lanthana (La2O3). Roughly 0.0018% of the earth's crust is composed of lanthanum. Today, lanthanum is primarily obtained through an ion exchange process from monazite sand ((Ce, La, Th, Nd, Y)PO4), a material rich in rare earth elements that can contain as much as 25% lanthanum.
From the Greek word lanthanein, to escape notice. Mosander in 1839 extracted lanthana from impure cerium nitrate and recognized the new element.
Lanthanum was isolated in relatively pure form in 1923. Iron exchange and solvent extraction techniques have led to much easier isolation of the so-called "rare-earth" elements.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
La: 5d¹ 6s²[Xe] 5d¹ 6s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 5d¹ 6s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 139 Stable | 138.9063563 ± 0.0000024 | 99.9112% | Stable |
Phase / State
Reason: 892.9 °C below melting point (917.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 57 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| La I | 0 | 393 | 315 | 393 |
| La II | +1 | 273 | 84 | 273 |
| La III | +2 | 122 | 0 | 0 |
| La IV | +3 | 87 | 0 | 0 |
| La V | +4 | 42 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| La I | 0 | 343 |
| La II | +1 | 119 |
| La III | +2 | 42 |
| La IV | +3 | 52 |
| La V | +4 | 37 |
| La VI | +5 | 2 |
| La VII | +6 | 2 |
| La VIII | +7 | 2 |
| La IX | +8 | 2 |
| La X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 103.2 pm |
| +3 | 7 | N/A | 110.00000000000001 pm |
| +3 | 8 | N/A | 115.99999999999999 pm |
| +3 | 9 | N/A | 121.6 pm |
| +3 | 10 | N/A | 127 pm |
| +3 | 12 | N/A | 136 pm |
Compounds
Isotopes (1)
Natural lanthanum is a mixture of two stable isotopes, 138La and 139La. Twenty three other radioactive isotopes are recognized.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 139 Stable | 138.9063563 ± 0.0000024 | 99.9112% ± 0.0007% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1193.15 K |
| Boiling point | 3737.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (13)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.1317 |
| 2 | p | 4.2044 |
| 2 | s | 15.0466 |
| 3 | d | 13.9398 |
| 3 | p | 18.8604 |
| 3 | s | 19.0569 |
| 4 | d | 32.2748 |
| 4 | f | 55.64 |
| 4 | p | 29.2936 |
| 4 | s | 28.2036 |
Crystal Radii Detail (6)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 117.2 | from r^3 vs V plots, | |
| 3 | VII | 124 | ||
| 3 | VIII | 130 | from r^3 vs V plots, | |
| 3 | IX | 135.6 | from r^3 vs V plots, | |
| 3 | X | 141 | ||
| 3 | XII | 150 | calculated, |
Isotope Decay Modes (64)
| Isotope | Mode | Intensity |
|---|---|---|
| 116 | B+ | — |
| 116 | B+p | — |
| 116 | p | — |
| 117 | p | 100% |
| 117 | B+ | — |
| 117 | B+p | — |
| 118 | B+ | — |
| 118 | B+p | — |
| 119 | B+ | — |
| 120 | B+ | 100% |
X‑ray Scattering Factors (711)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 3.31251 |
| 10.1152 | — | 3.28769 |
| 10.2317 | — | 3.26306 |
| 10.3496 | — | 3.23861 |
| 10.4688 | — | 3.20975 |
| 10.5894 | — | 3.15961 |
| 10.7114 | — | 3.11024 |
| 10.8348 | — | 3.06165 |
| 10.9596 | — | 3.01382 |
| 11.0859 | — | 2.96673 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
3.9×101 milligrams per kilogram
References (1)
- [5] Lanthanum https://education.jlab.org/itselemental/ele057.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
3.4×10-6 milligrams per liter
References (1)
- [5] Lanthanum https://education.jlab.org/itselemental/ele057.html
Sources
Sources of this element.
Lanthanum is found in rare-earth minerals such as cerite, monazite, allanite, and bastnasite. Monazite and bastnasite are principal ores in which lanthanum occurs in percentages up to 25 percent and 38 percent respectively. Misch metal, used in making lighter flints, contains about 25 percent lanthanum.
The availability of lanthanum and other rare earths has improved greatly in recent years. The metal can be produced by reducing the anhydrous fluoride with calcium.
References (1)
- [6] Lanthanum https://periodic.lanl.gov/57.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Lanthanum.
The element property data was retrieved from publications.