Barium (Ba)
alkaline-earth-metalSolid
Standard Atomic Weight
137.327 uElectron configuration
[Xe] 6s2Melting point
726.85 °C (1000 K)Boiling point
1896.85 °C (2170 K)Density
3620 kg/m³Oxidation states
+1, +2Electronegativity (Pauling)
0.89Ionization energy (1st)
Discovery year
1808Atomic radius
215 pmDetails
Barium is a soft alkaline earth metal, below strontium and above radium in group 2. It is highly reactive, forms Ba²⁺ compounds almost exclusively, and is not found free in nature. Its most important minerals are barite, barium sulfate, and witherite, barium carbonate. The element is technologically significant less as a metal than through dense, insoluble, or optically useful compounds.
Barium is a metallic element, soft, and when pure is silvery white; it belongs to the alkaline earth group, chemically resembling calcium. The metal oxidizes very easily and should be kept under petroleum or other suitable oxygen-free liquids to exclude air. It is decomposed by water or alcohol.
Barium was first isolated by Sir Humphry Davy, an English chemist, in 1808 through the electrolysis of molten baryta (BaO). Barium is never found free in nature since it reacts with oxygen in the air, forming barium oxide (BaO), and with water, forming barium hydroxide (Ba(OH)2) and hydrogen gas (H2). Barium is most commonly found as the mineral barite (BaSO4) and witherite (BaCO3) and is primarily produced through the electrolysis of barium chloride (BaCl2).
From the Greek word barys, heavy. Baryta was distinguished from lime by Scheele in 1774; the element was discovered by Sir Humphrey Davy in 1808.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
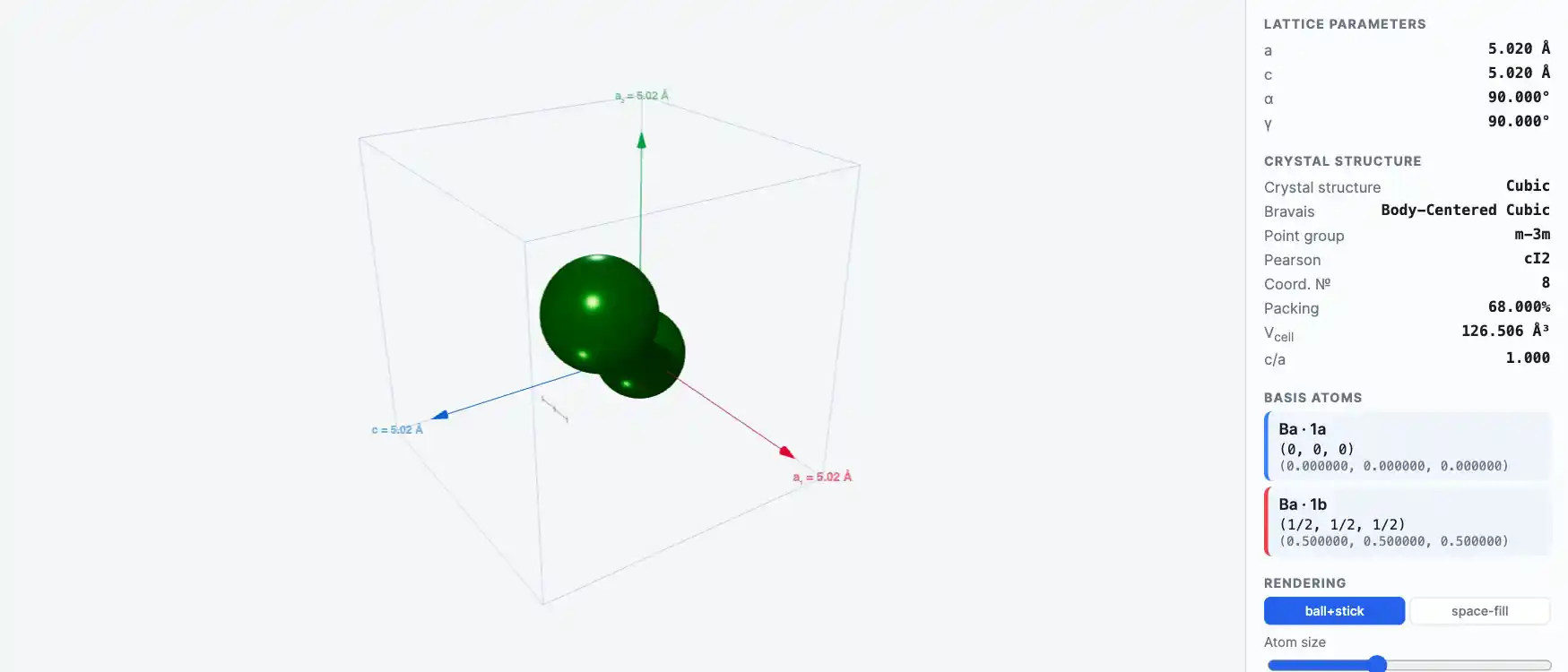
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Ba: 6s²[Xe] 6s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 6s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 134 Stable | 133.90450818 ± 0.0000003 | 2.4170% | Stable |
| 135 Stable | 134.90568838 ± 0.00000029 | 6.5920% | Stable |
| 136 Stable | 135.90457573 ± 0.00000029 | 7.8540% | Stable |
| 137 Stable | 136.90582714 ± 0.0000003 | 11.2320% | Stable |
| 138 Stable | 137.905247 ± 0.00000031 | 71.6980% | Stable |
Phase / State
Reason: 701.9 °C below melting point (726.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 56 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Ba I | 0 | 312 | 109 | 312 |
| Ba II | +1 | 112 | 94 | 112 |
| Ba III | +2 | 535 | 5 | 529 |
| Ba IV | +3 | 42 | 3 | 42 |
| Ba V | +4 | 135 | 0 | 126 |
| Ba VI | +5 | 138 | 0 | 127 |
| Ba VII | +6 | 70 | 0 | 70 |
| Ba VIII | +7 | 141 | 141 | 141 |
| Ba IX | +8 | 110 | 110 | 110 |
| Ba X | +9 | 31 | 31 | 31 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Ba I | 0 | 356 |
| Ba II | +1 | 162 |
| Ba III | +2 | 162 |
| Ba IV | +3 | 34 |
| Ba V | +4 | 52 |
| Ba VI | +5 | 50 |
| Ba VII | +6 | 32 |
| Ba VIII | +7 | 79 |
| Ba IX | +8 | 55 |
| Ba X | +9 | 30 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 6 | N/A | 135 pm |
| +2 | 7 | N/A | 138 pm |
| +2 | 8 | N/A | 142 pm |
| +2 | 9 | N/A | 147 pm |
| +2 | 10 | N/A | 152 pm |
| +2 | 11 | N/A | 157 pm |
| +2 | 12 | N/A | 161 pm |
Compounds
Isotopes (5)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 134 Stable | 133.90450818 ± 0.0000003 | 2.4170% ± 0.0180% | Stable | stable | |
| 135 Stable | 134.90568838 ± 0.00000029 | 6.5920% ± 0.0120% | Stable | stable | |
| 136 Stable | 135.90457573 ± 0.00000029 | 7.8540% ± 0.0240% | Stable | stable | |
| 137 Stable | 136.90582714 ± 0.0000003 | 11.2320% ± 0.0240% | Stable | stable | |
| 138 Stable | 137.905247 ± 0.00000031 | 71.6980% ± 0.0420% | Stable | stable |
Spectral Lines
Showing 50 of 92 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 392.686 nm | 25 | Ba III | emission | 5p5.(2P*<3/2>).6s 2[3/2]* → 5p5.(2P*<3/2>).6p 2[1/2] | Measured | NIST | |
| 399.306 nm | 25 | Ba III | emission | 5p5.(2P*<3/2>).6s 2[3/2]* → 5p5.(2P*<3/2>).6p 2[5/2] | Measured | NIST | |
| 381.3128 nm | 16 | Ba III | emission | 5p5.(2P*<1/2>).6s 2[1/2]* → 5p5.(2P*<1/2>).4f 2[5/2] | Measured | NIST | |
| 469.7428 nm | 15 | Ba III | emission | 5p5.(2P*<1/2>).5d 2[3/2]* → 5p5.(2P*<1/2>).6p 2[1/2] | Measured | NIST | |
| 448.1646 nm | 14 | Ba III | emission | 5p5.(2P*<1/2>).5d 2[3/2]* → 5p5.(2P*<3/2>).6p 2[5/2] | Measured | NIST | |
| 610.1987 nm | 13 | Ba III | emission | 5p5.(2P*<1/2>).5d 2[3/2]* → 5p5.(2P*<1/2>).4f 2[5/2] | Measured | NIST | |
| 389.6957 nm | 12 | Ba III | emission | 5p5.(2P*<3/2>).5d 2[5/2]* → 5p5.(2P*<3/2>).4f 2[3/2] | Measured | NIST | |
| 392.723 nm | 10 | Ba III | emission | 5p5.(2P*<3/2>).5d 2[5/2]* → 5p5.(2P*<3/2>).4f 2[9/2] | Measured | NIST | |
| 432.793 nm | 10 | Ba III | emission | 5p5.(2P*<3/2>).6s 2[3/2]* → 5p5.(2P*<3/2>).6p 2[1/2] | Measured | NIST | |
| 482.0642 nm | 10 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[3/2] → 5p5.(2P*<3/2>).5g 2[5/2]* | Measured | NIST | |
| 485.0833 nm | 10 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[3/2] → 5p5.(2P*<3/2>).5g 2[5/2]* | Measured | NIST | |
| 496.4038 nm | 10 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[5/2] → 5p5.(2P*<3/2>).5g 2[7/2]* | Measured | NIST | |
| 504.9533 nm | 10 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[5/2] → 5p5.(2P*<3/2>).5g 2[7/2]* | Measured | NIST | |
| 509.7537 nm | 10 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[7/2] → 5p5.(2P*<3/2>).5g 2[9/2]* | Measured | NIST | |
| 513.4529 nm | 10 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[7/2] → 5p5.(2P*<3/2>).5g 2[9/2]* | Measured | NIST | |
| 599.7996 nm | 10 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[5/2] → 5p5.(2P*<3/2>).7d 2[7/2]* | Measured | NIST | |
| 637.7094 nm | 10 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[3/2] → 5p5.(2P*<3/2>).7d 2[5/2]* | Measured | NIST | |
| 638.3756 nm | 10 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[5/2] → 5p5.(2P*<3/2>).7d 2[7/2]* | Measured | NIST | |
| 438.5824 nm | 9 | Ba III | emission | 5p5.(2P*<1/2>).5d 2[5/2]* → 5p5.(2P*<3/2>).6p 2[5/2] | Measured | NIST | |
| 464.6207 nm | 9 | Ba III | emission | 5p5.(2P*<3/2>).5d 2[5/2]* → 5p5.(2P*<3/2>).4f 2[3/2] | Measured | NIST | |
| 542.699 nm | 9 | Ba III | emission | 5p5.(2P*<1/2>).5d 2[5/2]* → 5p5.(2P*<3/2>).6p 2[5/2] | Measured | NIST | |
| 491.7171 nm | 8 | Ba III | emission | 5p5.(2P*<1/2>).5f 2[7/2] → 5p5.(2P*<1/2>).5g 2[9/2]* | Measured | NIST | |
| 494.5436 nm | 8 | Ba III | emission | 5p5.(2P*<1/2>).5f 2[5/2] → 5p5.(2P*<1/2>).5g 2[7/2]* | Measured | NIST | |
| 495.2914 nm | 8 | Ba III | emission | 5p5.(2P*<1/2>).5f 2[5/2] → 5p5.(2P*<1/2>).5g 2[7/2]* | Measured | NIST | |
| 496.3235 nm | 8 | Ba III | emission | 5p5.(2P*<1/2>).5f 2[7/2] → 5p5.(2P*<1/2>).5g 2[9/2]* | Measured | NIST | |
| 503.3498 nm | 8 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[5/2] → 5p5.(2P*<3/2>).5g 2[5/2]* | Measured | NIST | |
| 503.7341 nm | 8 | Ba III | emission | 5p5.(2P*<1/2>).5d 2[3/2]* → 5p5.(2P*<1/2>).6p 2[1/2] | Measured | NIST | |
| 590.0288 nm | 8 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[9/2] → 5p5.(2P*<3/2>).7d 2[7/2]* | Measured | NIST | |
| 601.6412 nm | 8 | Ba III | emission | 5p5.(2P*<1/2>).5d 2[3/2]* → 5p5.(2P*<1/2>).6p 2[3/2] | Measured | NIST | |
| 603.6589 nm | 8 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[5/2] → 5p5.(2P*<3/2>).7d 2[7/2]* | Measured | NIST | |
| 607.7807 nm | 8 | Ba III | emission | 5p5.(2P*<3/2>).6s 2[3/2]* → 5p5.(2P*<3/2>).4f 2[3/2] | Measured | NIST | |
| 627.0084 nm | 8 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[3/2] → 5p5.(2P*<3/2>).7d 2[5/2]* | Measured | NIST | |
| 652.6166 nm | 8 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[5/2] → 5p5.(2P*<3/2>).8s 2[3/2]* | Measured | NIST | |
| 709.5497 nm | 8 | Ba III | emission | 5p5.(2P*<3/2>).6s 2[3/2]* → 5p5.(2P*<3/2>).4f 2[3/2] | Measured | NIST | |
| 519.6426 nm | 7 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[5/2] → 5p5.(2P*<3/2>).5g 2[7/2]* | Measured | NIST | |
| 552.8138 nm | 7 | Ba III | emission | 5p5.(2P*<1/2>).5d 2[3/2]* → 5p5.(2P*<3/2>).6p 2[1/2] | Measured | NIST | |
| 574.0413 nm | 7 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[3/2] → 5p5.(2P*<3/2>).7d 2[7/2]* | Measured | NIST | |
| 579.8254 nm | 7 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[5/2] → 5p5.(2P*<3/2>).7d 2[5/2]* | Measured | NIST | |
| 585.9192 nm | 7 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[5/2] → 5p5.(2P*<3/2>).7d 2[5/2]* | Measured | NIST | |
| 588.1879 nm | 7 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[9/2] → 5p5.(2P*<3/2>).7d 2[7/2]* | Measured | NIST | |
| 598.3721 nm | 7 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[1/2] → 5p5.(2P*<3/2>).7d 2[1/2]* | Measured | NIST | |
| 658.3333 nm | 7 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[3/2] → 5p5.(2P*<3/2>).7d 2[3/2]* | Measured | NIST | |
| 565.8601 nm | 6 | Ba III | emission | 5p5.(2P*<1/2>).5d 2[5/2]* → 5p5.(2P*<3/2>).4f 2[7/2] | Measured | NIST | |
| 569.7415 nm | 6 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[3/2] → 5p5.(2P*<3/2>).7d 2[3/2]* | Measured | NIST | |
| 571.6614 nm | 6 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[1/2] → 5p5.(2P*<3/2>).7d 2[3/2]* | Measured | NIST | |
| 572.6169 nm | 6 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[3/2] → 5p5.(2P*<3/2>).7d 2[3/2]* | Measured | NIST | |
| 581.3545 nm | 6 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[3/2] → 5p5.(2P*<3/2>).7d 2[1/2]* | Measured | NIST | |
| 607.6665 nm | 6 | Ba III | emission | 5p5.(2P*<3/2>).5f 2[7/2] → 5p5.(2P*<3/2>).7d 2[5/2]* | Measured | NIST | |
| 613.1372 nm | 6 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[5/2] → 5p5.(2P*<3/2>).7d 2[1/2]* | Measured | NIST | |
| 640.614 nm | 6 | Ba III | emission | 5p5.(2P*<3/2>).7p 2[1/2] → 5p5.(2P*<3/2>).7d 2[3/2]* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1000.15 K |
| Boiling point | 2118.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (12)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.1139 |
| 2 | p | 4.1904 |
| 2 | s | 14.7532 |
| 3 | d | 13.9757 |
| 3 | p | 18.6836 |
| 3 | s | 18.8444 |
| 4 | d | 32.216 |
| 4 | p | 29.1968 |
| 4 | s | 28.08 |
| 5 | p | 41.1995 |
Crystal Radii Detail (7)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | VI | 149 | ||
| 2 | VII | 152 | calculated, | |
| 2 | VIII | 156 | ||
| 2 | IX | 161 | ||
| 2 | X | 166 | ||
| 2 | XI | 171 | ||
| 2 | XII | 175 | calculated, |
Isotope Decay Modes (55)
| Isotope | Mode | Intensity |
|---|---|---|
| 113 | p | — |
| 113 | A | — |
| 114 | B+ | 100% |
| 114 | B+p | 20% |
| 114 | A | 0.9% |
| 114 | 12C | 0% |
| 115 | B+ | 100% |
| 115 | B+p | 15% |
| 116 | B+ | 100% |
| 116 | B+p | 3% |
X‑ray Scattering Factors (508)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.10258 |
| 10.1617 | — | 0.10761 |
| 10.3261 | — | 0.11287 |
| 10.4931 | — | 0.1184 |
| 10.6628 | — | 0.1242 |
| 10.8353 | — | 0.13028 |
| 11.0106 | — | 0.13666 |
| 11.1886 | — | 0.14335 |
| 11.3696 | — | 0.15037 |
| 11.5535 | — | 0.15773 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
4.25×102 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.3×10-2 milligrams per liter
References (1)
Sources
Sources of this element.
It is found only in combination with other elements, chiefly with sulfate and carbonate and is prepared by electrolysis of the chloride.
References (1)
- [6] Barium https://periodic.lanl.gov/56.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Barium.
The element property data was retrieved from publications.