Iodine (I)
halogenSolid
Standard Atomic Weight
126.90447 uElectron configuration
[Kr] 5s2 4d10 5p5Melting point
113.7 °C (386.85 K)Boiling point
184.4 °C (457.55 K)Density
4930 kg/m³Oxidation states
−1, +1, +2, +3, +4, +5, +6, +7Electronegativity (Pauling)
2.66Ionization energy (1st)
Discovery year
1811Atomic radius
140 pmDetails
Iodine is a heavy halogen and the least abundant stable halogen in Earth’s crust. The element occurs naturally as iodide and iodate rather than as free I₂. It is chemically less electronegative and less strongly oxidizing than bromine or chlorine, and it forms a wide range of covalent, ionic, and polyiodide species. Iodine is an essential trace element for vertebrates because thyroid hormones contain iodine atoms.
Iodine is a bluish-black, lustrous solid, volatizing at ordinary temperatures into a blue-violet gas with an irritating odor; it forms compounds with many elements, but is less active than the other halogens, which displace it from iodides. Iodine exhibits some metallic-like properties. It dissolves readily in chloroform, carbon tetrachloride, or carbon disulfide to form beautiful purple solutions. It is only slightly soluble in water.
The name derives from the Greek iodes for "violet" because of its violet vapours. Iodine was discovered in seaweed by the French chemist Bernard Courtois in 1811, and named by the French chemist Louis-Joseph Gay-Lussac, when he proved it was an element in 1814.
Iodine was discovered by the French chemist Barnard Courtois in 1811. Courtois was extracting sodium and potassium compounds from seaweed ash. Once these compounds were removed, he added sulfuric acid (H2SO4) to further process the ash. He accidentally added too much acid and a violet colored cloud erupted from the mass. The gas condensed on metal objects in the room, creating solid iodine. Today, iodine is chiefly obtained from deposits of sodium iodate (NaIO3) and sodium periodate (NaIO4) in Chile and Bolivia. Trace amounts of iodine are required by the human body. Iodine is part of thyroxin, a hormone produced by the thyroid gland that controls the body's rate of physical and mental development. A lack of iodine can also cause a goiter, a swelling of the thyroid gland. Iodine is added to salt (iodized salt) to prevent these diseases.
From the Greek word iodes, violet. Discovered by Courtois in 1811, Iodine, a halogen, occurs sparingly in the form of iodides in sea water from which it is assimilated by seaweeds, Chilean saltpeter, nitrate-bearing earth (known as caliche), brines from old sea deposits, and in brackish waters from oil and salt wells.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
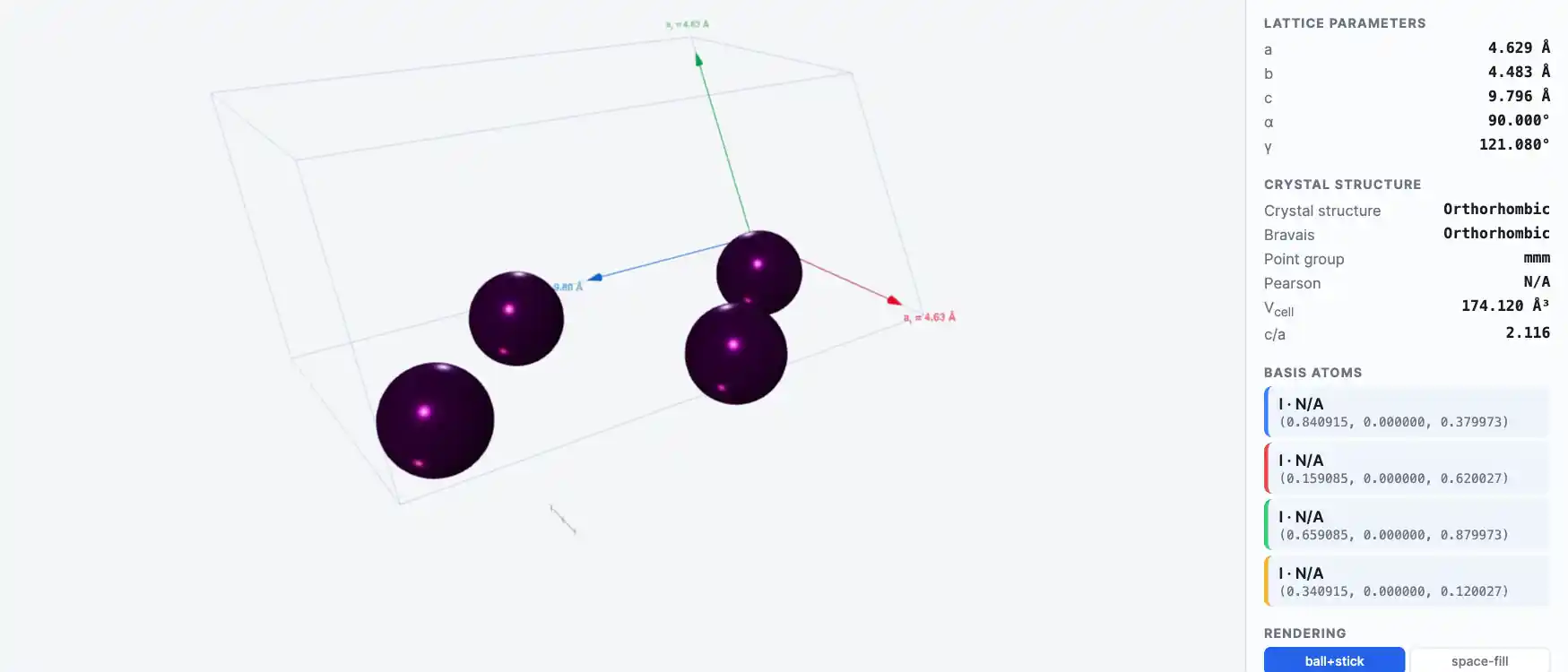
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
I: 4d¹⁰ 5s² 5p⁵[Kr] 4d¹⁰ 5s² 5p⁵1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁵Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 127 Stable | 126.9044719 ± 0.0000039 | 100.0000% | Stable |
Phase / State
Reason: 88.7 °C below melting point (113.7 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 53 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| I I | 0 | 1432 | 417 | 1432 |
| I II | +1 | 126 | 0 | 122 |
| I III | +2 | 76 | 0 | 0 |
| I IV | +3 | 47 | 0 | 0 |
| I V | +4 | 4 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| I I | 0 | 229 |
| I II | +1 | 315 |
| I III | +2 | 116 |
| I IV | +3 | 61 |
| I V | +4 | 54 |
| I VI | +5 | 40 |
| I VII | +6 | 25 |
| I VIII | +7 | 36 |
| I IX | +8 | 2 |
| I X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| -1 | 6 | N/A | 220.00000000000003 pm |
| +5 | 3 | N/A | 44 pm |
| +5 | 6 | N/A | 95 pm |
| +7 | 4 | N/A | 42 pm |
| +7 | 6 | N/A | 53 pm |
Compounds
Isotopes (1)
Thirty isotopes are recognized. Only one stable isotope, 127I is found in nature. The artificial radioisotope 131I, with a half-life of 8 days, has been used in treating the thyroid gland. The most common compounds are the iodides of sodium and potassium (KI) and the iodates (KIO3). Lack of iodine is the cause of goiter.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 127 Stable | 126.9044719 ± 0.0000039 | 100.0000% | Stable | stable |
Spectral Lines
Showing 50 of 474 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 511.92792 nm | 120000 | I I | emission | 5s2.5p4.(3P<2>).6s 2[2] → 5s2.5p4.(3P<2>).7p 2[1]* | Measured | NIST | |
| 740.20433 nm | 98000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[3]* → 5s2.5p4.(3P<2>).7d 2[4] | Measured | NIST | |
| 661.96418 nm | 88000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[3]* → 5s2.5p4.(3P<2>).8d 2[4] | Measured | NIST | |
| 746.89862 nm | 87000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[3]* → 5s2.5p4.(3P<2>).7d 2[4] | Measured | NIST | |
| 723.78303 nm | 68000 | I I | emission | 5s2.5p4.(3P<2>).5d 2[3] → 5s2.5p4.(3P<2>).5f 2[4]* | Measured | NIST | |
| 714.20318 nm | 53000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).7d 2[3] | Measured | NIST | |
| 658.3733 nm | 48000 | I I | emission | 5s2.5p4.(3P<2>).5d 2[3] → 5s2.5p4.(3P<2>).6f 2[4]* | Measured | NIST | |
| 633.78649 nm | 44000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).8d 2[3] | Measured | NIST | |
| 619.1891 nm | 36000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[3]* → 5s2.5p4.(3P<2>).9d 2[4] | Measured | NIST | |
| 712.20331 nm | 33000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).7d 2[3] | Measured | NIST | |
| 514.55362 nm | 26000 | I I | emission | 5s2.5p4.(3P<1>).6s 2[1] → 5s2.5p4.(3P<1>).7p 2[1]* | Measured | NIST | |
| 741.0472 nm | 25000 | I I | emission | 5s2.5p4.(3P<2>).5d 2[4] → 5s2.5p4.(3P<2>).6f 2[5]* | Measured | NIST | |
| 656.64687 nm | 23000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[3]* → 5s2.5p4.(3P<2>).8d 2[4] | Measured | NIST | |
| 633.94468 nm | 22000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).8d 2[3] | Measured | NIST | |
| 722.72727 nm | 22000 | I I | emission | 5s2.5p4.(3P<2>).5d 2[3] → 5s2.5p4.(3P<2>).5f 2[3]* | Measured | NIST | |
| 716.47586 nm | 21000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).7d 2[3] | Measured | NIST | |
| 698.6488 nm | 20000 | I I | emission | 5s2.5p4.(3P<2>).5d 2[4] → 5s2.5p4.(3P<2>).7f 2[5]* | Measured | NIST | |
| 621.3101 nm | 19000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[3]* → 5s2.5p4.(3P<2>).9d 2[4] | Measured | NIST | |
| 608.24072 nm | 18000 | I I | emission | 5s2.5p4.(3P<2>).6s 2[2] → 5s2.5p4.(3P<1>).6p 2[2]* | Measured | NIST | |
| 624.4475 nm | 17000 | I I | emission | 5s2.5p4.(3P<2>).5d 2[3] → 5s2.5p4.(3P<2>).7f 2[4]* | Measured | NIST | |
| 631.31292 nm | 17000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).8d 2[0] | Measured | NIST | |
| 589.39929 nm | 16000 | I I | emission | 5s2.5p4.(3P<2>).6s 2[2] → 5s2.5p4.(3P<1>).6p 2[1]* | Measured | NIST | |
| 666.20777 nm | 15000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[3]* → 5s2.5p4.(3P<2>).8d 2[3] | Measured | NIST | |
| 712.0036 nm | 15000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).7d 2[1] | Measured | NIST | |
| 666.10964 nm | 14000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).9s 2[2] | Measured | NIST | |
| 741.64587 nm | 14000 | I I | emission | 5s2.5p4.(3P<2>).5d 2[1] → 5s2.5p4.(3P<2>).5f 2[2]* | Measured | NIST | |
| 595.6854 nm | 13000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).9d 2[3] | Measured | NIST | |
| 598.4862 nm | 13000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).9d 2[2] | Measured | NIST | |
| 637.16776 nm | 12000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).8d 2[3] | Measured | NIST | |
| 486.23094 nm | 11000 | I I | emission | 5s2.5p4.(3P<2>).6s 2[2] → 5s2.5p4.(3P<2>).7p 2[3]* | Measured | NIST | |
| 491.69357 nm | 11000 | I I | emission | 5s2.5p4.(3P<2>).6s 2[2] → 5s2.5p4.(3P<2>).7p 2[2]* | Measured | NIST | |
| 520.41202 nm | 11000 | I I | emission | 5s2.5p4.(3P<2>).6s 2[2] → 5s2.5p4.(3P<2>).7p 2[1]* | Measured | NIST | |
| 629.39502 nm | 11000 | I I | emission | 5s2.5p4.(3P<2>).6s 2[2] → 5s2.5p4.(3P<0>).6p 2[1]* | Measured | NIST | |
| 633.0376 nm | 11000 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).8d 2[2] | Measured | NIST | |
| 523.45653 nm | 10000 | I I | emission | 5s2.5p4.(3P<2>).6s 2[2] → 5s2.5p4.(3P<2>).7p 2[3]* | Measured | NIST | |
| 533.82 nm | 10000 | I II | emission | 5s2.5p3.(2D*).6s 3D* → 5s2.5p3.(2D*).6p 3F | Measured | NIST | |
| 562.569 nm | 10000 | I II | emission | 5s2.5p3.(4S*).6s 3S* → 5s2.5p3.(4S*).6p 3P | Measured | NIST | |
| 707.78407 nm | 9700 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).7d 2[1] | Measured | NIST | |
| 598.4207 nm | 8900 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).9d 2[3] | Measured | NIST | |
| 742.00062 nm | 8300 | I I | emission | 5s2.5p4.(3P<2>).5d 2[4] → 5s2.5p4.(3P<2>).6f 2[4]* | Measured | NIST | |
| 596.8258 nm | 7900 | I I | emission | 5s2.5p4.(3P<2>).6p 2[3]* → 5s2.5p4.(3P<2>).10d 2[4] | Measured | NIST | |
| 698.97761 nm | 7800 | I I | emission | 5s2.5p4.(3P<2>).6p 2[3]* → 5s2.5p4.(3P<2>).9s 2[2] | Measured | NIST | |
| 658.05101 nm | 7600 | I I | emission | 5s2.5p4.(3P<2>).5d 2[3] → 5s2.5p4.(3P<2>).6f 2[3]* | Measured | NIST | |
| 673.20067 nm | 7600 | I I | emission | 5s2.5p4.(3P<2>).5d 2[1] → 5s2.5p4.(3P<2>).6f 2[3]* | Measured | NIST | |
| 595.4372 nm | 6700 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).9d 2[3] | Measured | NIST | |
| 741.1195 nm | 6700 | I I | emission | 5s2.5p4.(3P<2>).5d 2[1] → 5s2.5p4.(3P<2>).5f 2[3]* | Measured | NIST | |
| 656.08006 nm | 6600 | I I | emission | 5s2.5p4.(3P<2>).6p 2[3]* → 5s2.5p4.(3P<2>).8d 2[2] | Measured | NIST | |
| 723.49797 nm | 6600 | I I | emission | 5s2.5p4.(3P<2>).5d 2[3] → 5s2.5p4.(3P<2>).5f 2[4]* | Measured | NIST | |
| 633.35136 nm | 6300 | I I | emission | 5s2.5p4.(3P<2>).6p 2[2]* → 5s2.5p4.(3P<2>).8d 2[1] | Measured | NIST | |
| 723.17992 nm | 6200 | I I | emission | 5s2.5p4.(3P<2>).5d 2[3] → 5s2.5p4.(3P<2>).5f 2[3]* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 386.85 K |
| Boiling point | 457.55 K |
| Critical point (temperature) | 819.15 K |
| Triple point (temperature) | 386.75 K |
| Triple point (pressure) | 12.11 kPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (11)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.0609 |
| 2 | p | 4.1526 |
| 2 | s | 13.933 |
| 3 | d | 14.0993 |
| 3 | p | 18.1586 |
| 3 | s | 18.2126 |
| 4 | d | 32.066 |
| 4 | p | 28.9704 |
| 4 | s | 27.7028 |
| 5 | p | 41.3885 |
Crystal Radii Detail (5)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| -1 | VI | 206 | Ahrens (1952) ionic radius, | |
| 5 | IIIPY | 58 | ||
| 5 | VI | 109 | ||
| 7 | IV | 56 | ||
| 7 | VI | 67 |
Isotope Decay Modes (82)
| Isotope | Mode | Intensity |
|---|---|---|
| 106 | A | — |
| 107 | A | — |
| 108 | A | 99.5% |
| 108 | p | 0.5% |
| 108 | B+ | — |
| 108 | B+p | — |
| 109 | p | 100% |
| 109 | A | 0% |
| 110 | B+ | 83% |
| 110 | A | 17% |
X‑ray Scattering Factors (508)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 7.8167 |
| 10.1617 | — | 7.56781 |
| 10.3261 | — | 7.32685 |
| 10.4931 | — | 7.08081 |
| 10.6628 | — | 6.8332 |
| 10.8353 | — | 6.78435 |
| 11.0106 | — | 6.80888 |
| 11.1886 | — | 7.27334 |
| 11.3696 | — | 7.86775 |
| 11.5535 | — | 8.52786 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
4.5×10-1 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
6×10-2 milligrams per liter
References (1)
Sources
Sources of this element.
Ultrapure iodine can be obtained from the reaction of potassium iodide with copper sulfate. Several other methods of isolating the element are known.
References (1)
- [6] Iodine https://periodic.lanl.gov/53.shtml
Isotopes in Forensic Science and Anthropology
Information on the use of this element's isotopes in forensic science and anthropology.
131I (with a half-life of about 8 days) and 129I are both fission products; 129I is a long-lived fission product with a half-life of 1.7×107 years that can be helpful in the detection of the movement of radiation after a radioactive event, such as occurred at the Japanese reactors at Fukushima. In nuclear reactors and weapons tests, uranium and plutonium undergo fission processes in which one of the fission products is the long-lived isotope 129I. This isotope has been used as a groundwater tracer to determine evidence of nuclear fission, and it can also be tracked in rainwater as evidence of a fission event in the air (weapons explosion; Fig. IUPAC.53.1) [390] [390] D. Elmore, H. E. Gove, R. Ferraro, L. R. Kilius, H. W. Lee, K. H. Chang, R. P. Beukens, A. E. Litherland, C. J. Russo, K. H. Purser, M. T. Murrell, R. C. Finkel. Nature286, 138 (1980).[390] D. Elmore, H. E. Gove, R. Ferraro, L. R. Kilius, H. W. Lee, K. H. Chang, R. P. Beukens, A. E. Litherland, C. J. Russo, K. H. Purser, M. T. Murrell, R. C. Finkel. Nature286, 138 (1980)., [391] [391] G. Snyder, U. Fehn. Nucl. Instrum. Methods Phys. Res. B223, 579 (2004).[391] G. Snyder, U. Fehn. Nucl. Instrum. Methods Phys. Res. B223, 579 (2004)., [392] [392] G. Snyder, A. Aldahan, G. Possnert. Geochem. Geophys.11, Q04010 (2010).[392] G. Snyder, A. Aldahan, G. Possnert. Geochem. Geophys.11, Q04010 (2010)..
References (4)
- [390] D. Elmore, H. E. Gove, R. Ferraro, L. R. Kilius, H. W. Lee, K. H. Chang, R. P. Beukens, A. E. Litherland, C. J. Russo, K. H. Purser, M. T. Murrell, R. C. Finkel. Nature286, 138 (1980).
- [391] G. Snyder, U. Fehn. Nucl. Instrum. Methods Phys. Res. B223, 579 (2004).
- [392] G. Snyder, A. Aldahan, G. Possnert. Geochem. Geophys.11, Q04010 (2010).
- [4] IUPAC Periodic Table of the Elements and Isotopes (IPTEI) https://doi.org/10.1515/pac-2015-0703
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Iodine.
The element property data was retrieved from publications.