Tellurium (Te)
metalloidSolid
Standard Atomic Weight
127.6 uElectron configuration
[Kr] 5s2 4d10 5p4Melting point
449.51 °C (722.66 K)Boiling point
987.85 °C (1261 K)Density
6232 kg/m³Oxidation states
−2, −1, 0, +1, +2, +3, +4, +5, +6Electronegativity (Pauling)
2.1Ionization energy (1st)
Discovery year
1782Atomic radius
140 pmDetails
Tellurium is a brittle metalloid in group 16, below selenium and above polonium. It is chemically related to sulfur and selenium but is more metallic, less abundant, and more easily reduced. In nature it occurs mainly as telluride minerals and as a minor constituent of copper ores. Its technological importance comes from semiconducting and thermoelectric compounds, cadmium telluride photovoltaics, and small alloying additions that modify machinability and corrosion behavior.
Crystalline tellurium has a silvery-white appearance, and when pure it exhibits a metallic luster. It is brittle and easily pulverized. Amorphous tellurium is found by precipitating tellurium from a solution of telluric or tellurous acid. Whether this form is truly amorphous, or made of minute crystals, is open to question. Tellurium is a p-type semiconductor, and shows greater conductivity in certain directions, depending on alignment of the atoms.
Its conductivity increases slightly with exposure to light. It can be doped with silver, copper, gold, tin, or other elements. In air, tellurium burns with a greenish-blue flames, forming the dioxide. Molten tellurium corrodes iron, copper, and stainless steel.
The name derives from the Latin Tellus, who was the Roman goddess of the Earth. Tellurium was discovered by Franz Joseph Müller von Reichenstein in 1782 and overlooked for 15 years until it was isolated by the German chemist Martin-Heinrich Klaproth in 1798. The Hungarian chemist Paul Kitaibel independently discovered tellurium in 1789, prior to Klaproth's work but after von Reichenstein.
Tellurium was discovered by Franz Joseph Müller von Reichenstein, a Romanian mining official, in 1782. Reichenstein was the chief inspector of all mines, smelters and saltworks in Transylvania. He also had an interest in chemistry and extracted a new metal from an ore of gold, known as aurum album, which he believed was antimony. He shortly realized that the metal he had produced wasn't antimony at all, but a previously unknown element. Reichenstein's work was forgotten until 1798 when Martin Heinrich Klaproth, a German chemist, mentioned the substance in a paper. Klaproth named the new element tellurium but gave full credit for its discovery to Reichenstein. Tellurium is found free in nature, but is most often found in the ores sylvanite (AgAuTe4), calaverite (AuTe2) and krennerite (AuTe2). Today, most tellurium is obtained as a byproduct of mining and refining copper.
From the Latin word tellus, earth. Discovered by Muller von Reichenstein in 1782; named by Klaproth, who isolated it in 1798.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
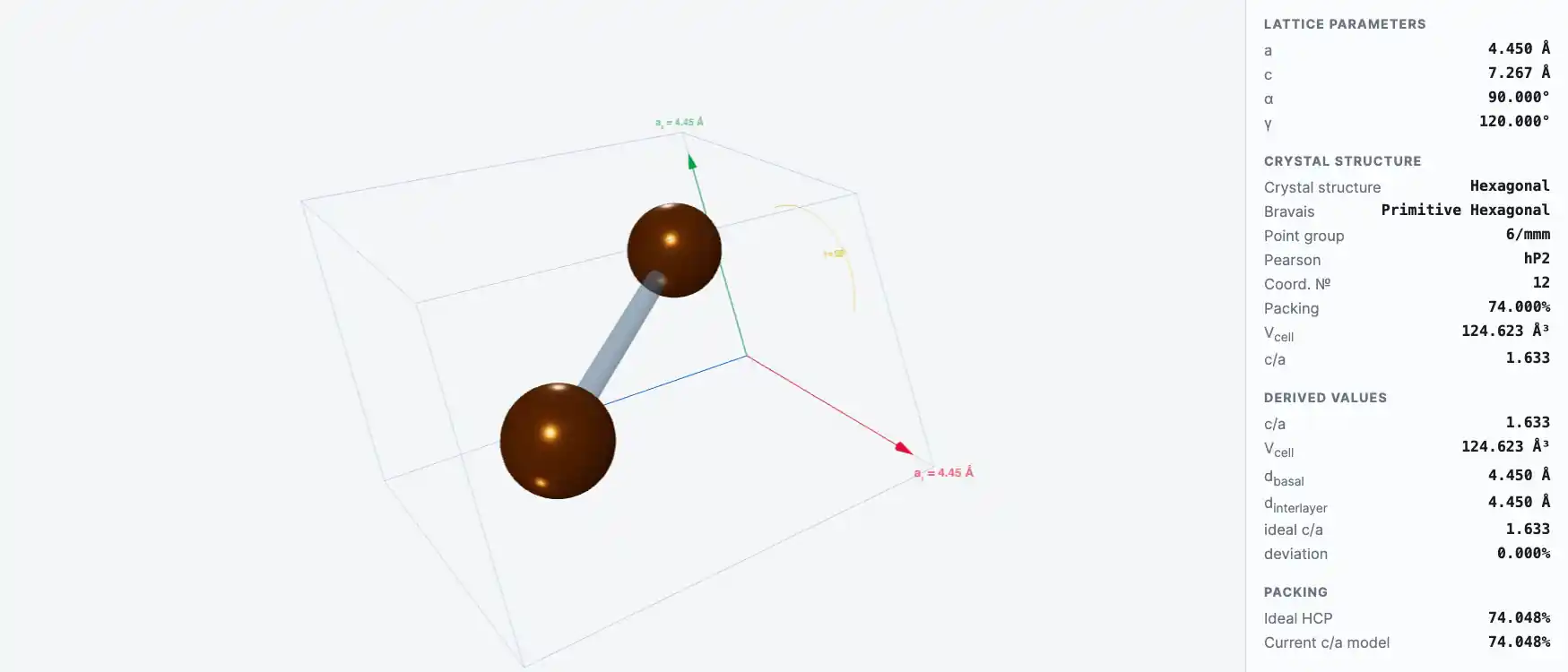
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Te: 4d¹⁰ 5s² 5p⁴[Kr] 4d¹⁰ 5s² 5p⁴1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁴Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 122 Stable | 121.9030435 ± 0.0000016 | 2.5500% | Stable |
| 124 Stable | 123.9028171 ± 0.0000016 | 4.7400% | Stable |
| 125 Stable | 124.9044299 ± 0.0000016 | 7.0700% | Stable |
| 126 Stable | 125.9033109 ± 0.0000016 | 18.8400% | Stable |
Phase / State
Reason: 424.5 °C below melting point (449.51 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 52 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Te I | 0 | 133 | 6 | 112 |
| Te II | +1 | 345 | 0 | 310 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Te I | 0 | 120 |
| Te II | +1 | 129 |
| Te III | +2 | 55 |
| Te IV | +3 | 16 |
| Te V | +4 | 45 |
| Te VI | +5 | 9 |
| Te VII | +6 | 60 |
| Te VIII | +7 | 2 |
| Te IX | +8 | 2 |
| Te X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| -2 | 6 | N/A | 221 pm |
| +4 | 3 | N/A | 52 pm |
| +4 | 4 | N/A | 66 pm |
| +4 | 6 | N/A | 97 pm |
| +6 | 4 | N/A | 43 pm |
| +6 | 6 | N/A | 56.00000000000001 pm |
Compounds
Isotopes (4)
Thirty isotopes of tellurium are known, with atomic masses ranging from 108 to 137. Natural tellurium consists of eight isotopes.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 122 Stable | 121.9030435 ± 0.0000016 | 2.5500% ± 0.1200% | Stable | stable | |
| 124 Stable | 123.9028171 ± 0.0000016 | 4.7400% ± 0.1400% | Stable | stable | |
| 125 Stable | 124.9044299 ± 0.0000016 | 7.0700% ± 0.1500% | Stable | stable | |
| 126 Stable | 125.9033109 ± 0.0000016 | 18.8400% ± 0.2500% | Stable | stable |
Spectral Lines
Showing 50 of 74 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 486.623 nm | 2300 | Te II | emission | 5s2.5p2.(3P).6p 4D* → 5s2.5p2.(3P).6d 4F | Measured | NIST | |
| 557.636 nm | 2100 | Te II | emission | 5s2.5p2.(1D).6s 2D → 5s2.5p2.(1D).6p 2F* | Measured | NIST | |
| 570.812 nm | 1900 | Te II | emission | 5s2.5p2.(3P).6s 4P → 5s2.5p2.(3P).6p 4D* | Measured | NIST | |
| 483.13 nm | 1600 | Te II | emission | 5s2.5p2.(3P).6p 4D* → 5s2.5p2.(3P).6d 4P | Measured | NIST | |
| 564.926 nm | 1500 | Te II | emission | 5s2.5p2.(3P).6s 4P → 5s2.5p2.(3P).6p 4D* | Measured | NIST | |
| 575.586 nm | 1500 | Te II | emission | 5s2.5p2.(3P).6s 4P → 5s2.5p2.(3P).6p 4D* | Measured | NIST | |
| 544.984 nm | 1400 | Te II | emission | 5s2.5p2.(3P).6s 4P → 5s2.5p2.(3P).6p 4P* | Measured | NIST | |
| 468.691 nm | 1310 | Te II | emission | 5s2.5p2.(3P).6p 4P* → 5s2.5p2.(3P).6d 4D | Measured | NIST | |
| 476.605 nm | 1300 | Te II | emission | 5s2.5p2.(3P).6p 2D* → 5s2.5p2.(3P).6d 2F | Measured | NIST | |
| 490.442 nm | 1300 | Te II | emission | 5s2.5p2.(3P).6p 2D* → 5s2.5p2.(3P).6d 2F | Measured | NIST | |
| 566.622 nm | 1200 | Te II | emission | 5s2.5p2.(3P).6s 2P → 5s2.5p2.(3P).6p 2D* | Measured | NIST | |
| 597.468 nm | 1200 | Te II | emission | 5s2.5p2.(3P).6s 4P → 5s2.5p2.(3P).6p 4P* | Measured | NIST | |
| 548.795 nm | 1100 | Te II | emission | 5s2.5p2.(3P).5d 2P → 5s2.5p2.(3P).6p 4D* | Measured | NIST | |
| 484.29 nm | 1000 | Te II | emission | 5s2.5p2.(3P).5d 2D → 5s2.5p2.(3P).4f 4D* | Measured | NIST | |
| 486.513 nm | 1000 | Te II | emission | 5s2.5p2.(3P).6p 4D* → 5s2.5p2.(3P).6d 4D | Measured | NIST | |
| 482.712 nm | 900 | Te II | emission | 5s2.5p2.(3P).6p 4P* → 5s2.5p2.(3P).6d 4D | Measured | NIST | |
| 447.865 nm | 830 | Te II | emission | 5s2.5p2.(3P).6p 4P* → 5s2.5p2.(3P).6d 4D | Measured | NIST | |
| 500.081 nm | 810 | Te II | emission | 5s2.5p2.(3P).6s 4P → 5s2.5p2.(3P).6p 2D* | Measured | NIST | |
| 477.155 nm | 800 | Te II | emission | 5s2.5p2.(3P).6p 4D* → 5s2.5p2.(3P).6d 4F | Measured | NIST | |
| 593.615 nm | 730 | Te II | emission | 5s2.5p2.(3P).6s 4P → 5s2.5p2.(3P).6p 4S* | Measured | NIST | |
| 464.111 nm | 680 | Te II | emission | 5s2.5p2.(3P).6p 4D* → 5s2.5p2.(3P).6d 4D | Measured | NIST | |
| 470.654 nm | 670 | Te II | emission | 5s2.5p2.(1D).6s 2D → 5s2.5p2.(1D).6p 2P* | Measured | NIST | |
| 436.402 nm | 650 | Te II | emission | 5s2.5p2.(3P).5d 4P → 5s2.5p2.(3P).4f 4D* | Measured | NIST | |
| 636.713 nm | 570 | Te II | emission | 5s.(2S).5p4.(1D) 2D → 5s2.5p2.(3P).6p 4D* | Measured | NIST | |
| 469.638 nm | 560 | Te II | emission | 5s2.5p2.(3P).5d 4P → 5s2.5p2.(3P).4f 2D* | Measured | NIST | |
| 416.977 nm | 540 | Te II | emission | 5s2.5p2.(3P).5d 2D → 5s2.5p2.(3P).4f 2F* | Measured | NIST | |
| 463.062 nm | 540 | Te II | emission | 5s2.5p2.(3P).6p 4S* → 5s2.5p2.(3P).7s 2P | Measured | NIST | |
| 478.488 nm | 510 | Te II | emission | 5s2.5p2.(1D).6s 2D → 5s2.5p2.(1D).6p 2P* | Measured | NIST | |
| 455.778 nm | 480 | Te II | emission | 5s2.5p2.(3P).6p 4S* → 5s2.5p2.(3P).6d 4D | Measured | NIST | |
| 683.7663 nm | 430 | Te I | emission | 5p3.(4S*).6p 5P → 5p3.(4S*).8d 5D* | Measured | NIST | |
| 404.716 nm | 400 | Te II | emission | 5s2.5p2.(3P).5d 4P → 5s2.5p2.(3P).4f 2D* | Measured | NIST | |
| 428.583 nm | 370 | Te II | emission | 5s2.5p2.(3P).6p 2S* → 5s2.5p2.(3P).6d 4D | Measured | NIST | |
| 394.798 nm | 340 | Te II | emission | 5s2.5p2.(3P).5d 4P → 5s2.5p2.(3P).4f 4F* | Measured | NIST | |
| 422.572 nm | 340 | Te II | emission | 5s2.5p2.(3P).5d 4P → 5s2.5p2.(3P).4f 4D* | Measured | NIST | |
| 396.921 nm | 320 | Te II | emission | 5s.(2S).5p4.(1D) 2D → 5s2.5p2.(1D).6p 2D* | Measured | NIST | |
| 410.105 nm | 320 | Te II | emission | 5s2.5p2.(1D).5d 2S → 5s2.5p2.(3P).4f 2D* | Measured | NIST | |
| 412.732 nm | 320 | Te II | emission | 5s2.5p2.(1D).5d 2S → 5s2.5p2.(3P).4f 4D* | Measured | NIST | |
| 496.187 nm | 320 | Te II | emission | 5s2.5p2.(3P).6p 4P* → 5s2.5p2.(3P).6d 4D | Measured | NIST | |
| 400.653 nm | 310 | Te II | emission | 5s.(2S).5p4.(3P) 4P → 5s2.5p2.(3P).6p 4D* | Measured | NIST | |
| 438.51 nm | 310 | Te II | emission | 5s2.5p2.(3P).5d 4P → 5s2.5p2.(3P).4f 2D* | Measured | NIST | |
| 417.929 nm | 300 | Te II | emission | 5s2.5p2.(3P).5d 4P → 5s2.5p2.(1S).6p 2P* | Measured | NIST | |
| 427.343 nm | 300 | Te II | emission | 5s2.5p2.(3P).6p 2S* → 5s2.5p2.(3P).7s 2P | Measured | NIST | |
| 679.109 nm | 300 | Te I | emission | 5p3.(4S*).6p 5P → 5p3.(4S*).8d 3D* | Measured | NIST | |
| 669.0154 nm | 290 | Te I | emission | 5p3.(4S*).6p 5P → 5p3.(4S*).8d 5D* | Measured | NIST | |
| 453.708 nm | 260 | Te II | emission | 5s2.5p2.(3P).5d 4P → 5s2.5p2.(3P).4f 4F* | Measured | NIST | |
| 397.592 nm | 250 | Te II | emission | 5s.(2S).5p4.(1D) 2D → 5s2.5p2.(1D).6p 2F* | Measured | NIST | |
| 416.356 nm | 250 | Te II | emission | 5s2.5p2.(3P).5d 4P → 5s2.5p2.(3P).4f 4D* | Measured | NIST | |
| 398.176 nm | 240 | Te II | emission | 5s2.5p2.(3P).6s 2P → 5s2.5p2.(1D).6p 2P* | Measured | NIST | |
| 425.114 nm | 240 | Te II | emission | 5s2.5p2.(3P).5d 2D → 5s2.5p2.(3P).4f 2F* | Measured | NIST | |
| 404.888 nm | 230 | Te II | emission | 5s.(2S).5p4.(3P) 4P → 5s2.5p2.(3P).6p 4S* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Phase Transitions & Allotropes
| Melting point | 722.66 K |
| Boiling point | 1261.15 K |
| Critical point (temperature) | 2329.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (11)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.0432 |
| 2 | p | 4.14 |
| 2 | s | 13.6688 |
| 3 | d | 14.1607 |
| 3 | p | 17.9911 |
| 3 | s | 18.0019 |
| 4 | d | 32.04 |
| 4 | p | 28.878 |
| 4 | s | 27.5916 |
| 5 | p | 41.1915 |
Crystal Radii Detail (6)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| -2 | VI | 207 | Pauling's (1960) crystal radius, | |
| 4 | III | 66 | ||
| 4 | IV | 80 | ||
| 4 | VI | 111 | ||
| 6 | IV | 57 | calculated, | |
| 6 | VI | 70 |
Isotope Decay Modes (67)
| Isotope | Mode | Intensity |
|---|---|---|
| 104 | A | 100% |
| 105 | A | 100% |
| 106 | A | 100% |
| 107 | A | 70% |
| 107 | B+ | — |
| 107 | B+p | — |
| 108 | B+ | 51% |
| 108 | A | 49% |
| 108 | B+p | 2.4% |
| 108 | B+A | 0.1% |
X‑ray Scattering Factors (508)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 9.70237 |
| 10.1617 | — | 9.72653 |
| 10.3261 | — | 9.75076 |
| 10.4931 | — | 9.77506 |
| 10.6628 | — | 9.7994 |
| 10.8353 | — | 9.77638 |
| 11.0106 | — | 9.72308 |
| 11.1886 | — | 9.67008 |
| 11.3696 | — | 9.61736 |
| 11.5535 | — | 9.54395 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1×10-3 milligrams per kilogram
References (1)
- [5] Tellurium https://education.jlab.org/itselemental/ele052.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
Not Applicable
References (1)
- [5] Tellurium https://education.jlab.org/itselemental/ele052.html
Sources
Sources of this element.
Tellurium is occasionally found native, but is more often found as the telluride of gold (calaverite), and combined with other metals. It is recovered commercially from anode muds produced during the electrolytic refining of blister copper. The U.S., Canada, Peru, and Japan are the largest Free World producers of the element.
References (1)
- [6] Tellurium https://periodic.lanl.gov/52.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Tellurium.
The element property data was retrieved from publications.