Rhodium (Rh)
transition-metalSolid
Standard Atomic Weight
102.9055 uElectron configuration
[Kr] 5s1 4d8Melting point
1963.85 °C (2237 K)Boiling point
3694.85 °C (3968 K)Density
1.240000e+4 kg/m³Oxidation states
−3, −1, +1, +2, +3, +4, +5, +6, +7Electronegativity (Pauling)
2.28Ionization energy (1st)
Discovery year
1803Atomic radius
135 pmDetails
Rhodium is a very rare platinum-group transition metal. It is chemically noble, hard, highly reflective, and most often encountered in nature alloyed with platinum, palladium, and other platinum-group elements. Its industrial importance is dominated by catalysis, especially control of nitrogen oxides in automotive exhaust. Rhodium also forms stable coordination compounds, commonly with Rh(I) and Rh(III), that are important in homogeneous catalysis and organometallic chemistry.
The metal is silvery white and at red heat slowly changes in air to the resquioxide. At higher temperatures it converts back to the element. Rhodium has a higher melting point and lower density than platinum. It is highly reflective, hard, and durable.
The name derives from the Greek rhodon for rose because of the rose color of dilute solutions of its salts. It was discovered by the English chemist and physicist William Hyde Wollaston in 1803 in a crude platinum ore.
Rhodium was discovered by William Hyde Wollaston, an English chemist, in 1803 shortly after his discovery of the element palladium. He obtained rhodium from a sample of platinum ore that was obtained from South America. After removing the platinum and palladium from the sample, he was left with a dark red powder. The powder turned out to be sodium rhodium chloride (Na3RhCl6·12H2O). Wollaston obtained rhodium from the powder by treating it with hydrogen gas (H2). Rhodium tends to occur along with deposits of platinum and is primarily obtained as a byproduct of mining and refining platinum. Rhodium is also obtained as a byproduct of the nickel mining operation in the Sudbury region of Ontario, Canada.
From the Greek word rhodon, rose. Wollaston discovered rhodium between 1803 and 1804 in crude platinum ore he presumably obtained from South America.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
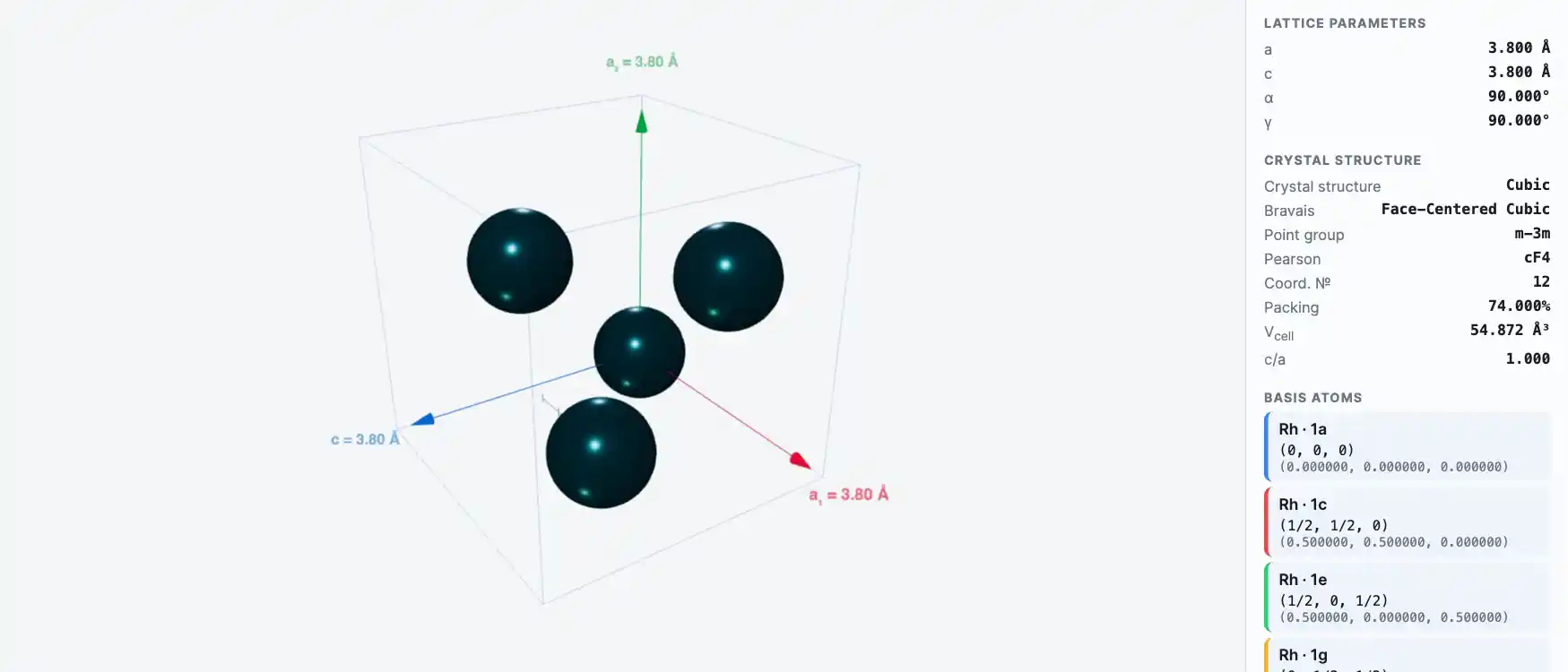
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Rh: 4d⁸ 5s¹[Kr] 4d⁸ 5s¹1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d⁸ 5s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 103 Stable | 102.905498 ± 0.0000026 | 100.0000% | Stable |
Phase / State
Reason: 1938.8 °C below melting point (1963.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 45 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Rh I | 0 | 468 | 111 | 443 |
| Rh II | +1 | 34 | 0 | 31 |
| Rh III | +2 | 73 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Rh I | 0 | 138 |
| Rh II | +1 | 126 |
| Rh III | +2 | 196 |
| Rh IV | +3 | 2 |
| Rh V | +4 | 2 |
| Rh VI | +5 | 2 |
| Rh VII | +6 | 2 |
| Rh VIII | +7 | 2 |
| Rh IX | +8 | 2 |
| Rh X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 66.5 pm |
| +4 | 6 | N/A | 60 pm |
| +5 | 6 | N/A | 55.00000000000001 pm |
Compounds
Isotopes (1)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 103 Stable | 102.905498 ± 0.0000026 | 100.0000% | Stable | stable |
Spectral Lines
Showing 50 of 186 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 385.6513 nm | 5900 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 2G* | Measured | NIST | |
| 437.4809 nm | 4200 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 4G* | Measured | NIST | |
| 382.226 nm | 3800 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 2F* | Measured | NIST | |
| 395.8856 nm | 3800 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 2G* | Measured | NIST | |
| 421.1133 nm | 3300 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 4F* | Measured | NIST | |
| 382.8478 nm | 2300 | Rh I | emission | 4d8.(3P).5s a 4P → 4d8.(3P).5p z 4P* | Measured | NIST | |
| 413.5275 nm | 2100 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 4F* | Measured | NIST | |
| 383.3884 nm | 2000 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 2D* | Measured | NIST | |
| 393.4224 nm | 2000 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 4G* | Measured | NIST | |
| 412.8886 nm | 1500 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 2F* | Measured | NIST | |
| 380.6759 nm | 1300 | Rh I | emission | 4d8.(3F).5s a 4F → 4d8.(3F).5p z 4D* | Measured | NIST | |
| 381.8186 nm | 1300 | Rh I | emission | 4d8.(3P).5s a 4P → 4d8.(3P).5p z 4P* | Measured | NIST | |
| 412.1683 nm | 1100 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 2D* | Measured | NIST | |
| 428.8702 nm | 820 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 4G* | Measured | NIST | |
| 380.592 nm | 760 | Rh I | emission | 4d8.(1D).5s b 2D → 8* | Measured | NIST | |
| 381.6474 nm | 760 | Rh I | emission | 4d8.(1D).5s b 2D → 4d8.(1D).5p y 2F* | Measured | NIST | |
| 394.271 nm | 590 | Rh I | emission | 4d8.(3P).5s a 4P → 4d8.(3P).5p z 4P* | Measured | NIST | |
| 408.278 nm | 560 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 4F* | Measured | NIST | |
| 387.0018 nm | 490 | Rh I | emission | 4d8.(1G).5s a 2G → 12* | Measured | NIST | |
| 381.5021 nm | 470 | Rh I | emission | 4d8.(1G).5s a 2G → 13* | Measured | NIST | |
| 387.7346 nm | 380 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 4F* | Measured | NIST | |
| 397.5313 nm | 380 | Rh I | emission | 4d8.(1D).5s b 2D → 4* | Measured | NIST | |
| 399.6149 nm | 380 | Rh I | emission | 4d8.(3P).5s a 2P → 4d8.(3P).5p y 4D* | Measured | NIST | |
| 419.6496 nm | 330 | Rh I | emission | 4d8.(3P).5s a 4P → 4d8.(3F).5p z 2G* | Measured | NIST | |
| 392.2195 nm | 240 | Rh I | emission | 4d9 a 2D → 4d8.(3F).5p z 4D* | Measured | NIST | |
| 398.4393 nm | 240 | Rh I | emission | 4d8.(3P).5s a 4P → 4d8.(3P).5p z 4P* | Measured | NIST | |
| 399.5602 nm | 240 | Rh I | emission | 4d8.(3P).5s a 4P → 4d8.(3P).5p z 4P* | Measured | NIST | |
| 415.4343 nm | 240 | Rh I | emission | 4d7.5s2 b 4F → 4d8.(3P).5p y 4D* | Measured | NIST | |
| 559.9419 nm | 160 | Rh I | emission | 4d8.(3P).5s a 4P → 4d8.(3F).5p z 4D* | Measured | NIST | |
| 467.5022 nm | 150 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 4D* | Measured | NIST | |
| 409.7508 nm | 140 | Rh I | emission | 4d8.(3P).5s a 4P → 4d8.(3P).5p z 4P* | Measured | NIST | |
| 456.8993 nm | 130 | Rh I | emission | 4d8.(3P).5s a 4P → 4d8.(3F).5p z 4G* | Measured | NIST | |
| 535.4428 nm | 130 | Rh I | emission | 4d8.(3F).5p z 2G* → 16 | Measured | NIST | |
| 598.3575 nm | 130 | Rh I | emission | 4d7.5s2 b 4F → 4d8.(3F).5p z 4F* | Measured | NIST | |
| 391.3508 nm | 120 | Rh I | emission | 4d8.(3F).5s a 4F → 4d8.(3F).5p z 4D* | Measured | NIST | |
| 402.3139 nm | 120 | Rh I | emission | 4d8.(1D).5s b 2D → 4d8.(1D).5p y 2P* | Measured | NIST | |
| 411.9679 nm | 120 | Rh I | emission | 4d8.(1G).5s a 2G → 4d8.(1D).5p y 2F* | Measured | NIST | |
| 381.2462 nm | 95 | Rh I | emission | 4d8.(1D).5s b 2D → 4d8.(3P).5p z 2S* | Measured | NIST | |
| 395.8233 nm | 95 | Rh I | emission | 4d8.(3P).5s a 2P → 4d8.(1D).5p y 2P* | Measured | NIST | |
| 437.9911 nm | 95 | Rh I | emission | 4d8.(3P).5s a 4P → 4d8.(3F).5p z 2D* | Measured | NIST | |
| 519.313 nm | 95 | Rh I | emission | 4d8.(3F).5p z 4G* → 2 | Measured | NIST | |
| 539.0433 nm | 95 | Rh I | emission | 4d8.(3P).5s a 4P → 4d8.(3F).5p z 4D* | Measured | NIST | |
| 387.239 nm | 70 | Rh I | emission | 4d9 a 2D → 4d8.(3F).5p z 4F* | Measured | NIST | |
| 388.8331 nm | 70 | Rh I | emission | 4d8.(1D).5s b 2D → 4d8.(1D).5p y 2P* | Measured | NIST | |
| 407.758 nm | 70 | Rh I | emission | 4d8.(1D).5s b 2D → 4d8.(3P).5p y 4D* | Measured | NIST | |
| 411.6329 nm | 70 | Rh I | emission | 4d8.(1D).5s b 2D → 4* | Measured | NIST | |
| 420.6613 nm | 70 | Rh I | emission | 4d9 a 2D → 4d8.(3F).5p z 4D* | Measured | NIST | |
| 429.6763 nm | 70 | Rh I | emission | 4d8.(1D).5s b 2D → 4d8.(3P).5p y 4D* | Measured | NIST | |
| 474.5116 nm | 70 | Rh I | emission | 4d8.(3F).5s a 2F → 4d8.(3F).5p z 4D* | Measured | NIST | |
| 509.064 nm | 70 | Rh I | emission | 4d8.(3P).5s a 4P → 4d8.(3F).5p z 4D* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 2236.15 K |
| Boiling point | 3968.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (10)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.9244 |
| 2 | p | 4.0596 |
| 2 | s | 11.8454 |
| 3 | d | 14.595 |
| 3 | p | 16.8456 |
| 3 | s | 16.5615 |
| 4 | d | 31.5576 |
| 4 | p | 27.8604 |
| 4 | s | 26.4184 |
| 5 | s | 38.3605 |
Crystal Radii Detail (3)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 80.5 | from r^3 vs V plots, | |
| 4 | VI | 74 | from r^3 vs V plots, from metallic oxides, | |
| 5 | VI | 69 |
Isotope Decay Modes (72)
| Isotope | Mode | Intensity |
|---|---|---|
| 88 | B+ | — |
| 89 | B+ | — |
| 89 | B+p | — |
| 89 | p | — |
| 90 | B+ | 100% |
| 90 | B+p | 0.7% |
| 91 | B+ | 100% |
| 91 | B+p | 1.3% |
| 92 | B+ | 100% |
| 92 | B+p | 2% |
X‑ray Scattering Factors (508)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.17537 |
| 10.1617 | — | 1.24044 |
| 10.3261 | — | 1.30912 |
| 10.4931 | — | 1.3816 |
| 10.6628 | — | 1.4581 |
| 10.8353 | — | 1.53883 |
| 11.0106 | — | 1.62403 |
| 11.1886 | — | 1.71394 |
| 11.3696 | — | 1.80884 |
| 11.5535 | — | 1.90899 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1×10-3 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
Not Applicable
References (1)
Sources
Sources of this element.
Rhodium occurs natively with other platinum metals in river sands of the Urals and in North and South America. It is also found with other platinum metals in the copper-nickel sulfide area of the Sudbury, Ontario region. Although the quantity occurring there is very small, the large tonnages of nickel processed make the recovery commercially feasible. The annual world production of rhodium is only 7 or 8 tons.
References (1)
- [6] Rhodium https://periodic.lanl.gov/45.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Rhodium.
The element property data was retrieved from publications.