Ruthenium (Ru)
transition-metalSolid
Standard Atomic Weight
101.07 uElectron configuration
[Kr] 5s1 4d7Melting point
2333.85 °C (2607 K)Boiling point
4149.85 °C (4423 K)Density
1.210000e+4 kg/m³Oxidation states
−4, −2, +1, +2, +3, +4, +5, +6, +7, +8Electronegativity (Pauling)
2.2Ionization energy (1st)
Discovery year
1828Atomic radius
130 pmDetails
Ruthenium is a hard, platinum-group transition metal with atomic number 44. It is rare in the crust and is recovered chiefly with platinum and nickel-copper sulfide ores. Chemically it is notable for a wide range of oxidation states, especially +2, +3, +4, +6, and +8, and for forming many coordination and organometallic compounds. Metallic ruthenium improves hardness and corrosion resistance in some platinum and palladium alloys, while its oxides and complexes are important in catalysis and electrochemistry.
Ruthenium is a hard, white metal and has four crystal modifications. It does not tarnish at room temperatures, but oxidizes explosively. It is attacked by halogens, hydroxides, etc. Ruthenium can be plated by electrodeposition or by thermal decomposition methods. The metal is one of the most effective hardeners for platinum and palladium, and is alloyed with these metals to make electrical contacts for severe wear resistance. A ruthenium-molybdenum alloy is said to be superconductive at 10.6 K. The corrosion resistance of titanium is improved a hundredfold by addition of 0.1% ruthenium. It is a versatile catalyst. Hydrogen sulfide can be split catalytically by light using an aqueous suspension of CdS particles loaded with ruthenium dioxide. It is thought this may have application to removal of H2S from oil refining and other industrial processes. Compounds in at least eight oxidation states have been found, but of these, the +2, +3, and +4 states are the most common. Ruthenium tetroxide, like osmium tetroxide, is highly toxic. In addition, it may explode. Ruthenium compounds show a marked resemblance to those of cadmium.
The name derives from the Latin ruthenia for the old name of Russia. It was discovered in a crude platinum ore by the Russian chemist Gottfried Wilhelm Osann in 1828. Osann thought that he had found three new metals in the sample, pluranium, ruthenium, and polinium. In 1844, Russian chemist Karl Karlovich Klaus was able to show that Osann's mistake was due to the impurity of the sample, and Klaus was able to isolate the ruthenium metal.
Ruthenium was discovered by Karl Karlovich Klaus, a Russian chemist, in 1844 while analyzing the residue of a sample of platinum ore obtained from the Ural mountains. Apparently, Jedrzej Sniadecki, a Polish chemist, had produced ruthenium in 1807 but he withdrew his claim of discovery after other scientists failed to replicate his results. Ruthenium tends to occur along with deposits of platinum and is primarily obtained as a byproduct of mining and refining platinum. Ruthenium is also obtained as a byproduct of the nickel mining operation in the Sudbury region of Ontario, Canada.
From the Latin word Ruthenia, Russia. In 1827, Berzelius and Osann examined the residues left after dissolving crude platinum from the Ural mountains in aqua regia. While Berzelius found no unusual metals, Osann thought he found three new metals, one of which he named ruthenium. In 1844 Klaus, generally recognized as the discoverer, showed that Osann's ruthenium oxide was very impure and that it contained a new metal. Klaus obtained 6 g of ruthenium from the portion of crude platinum that is insoluble in aqua regia.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
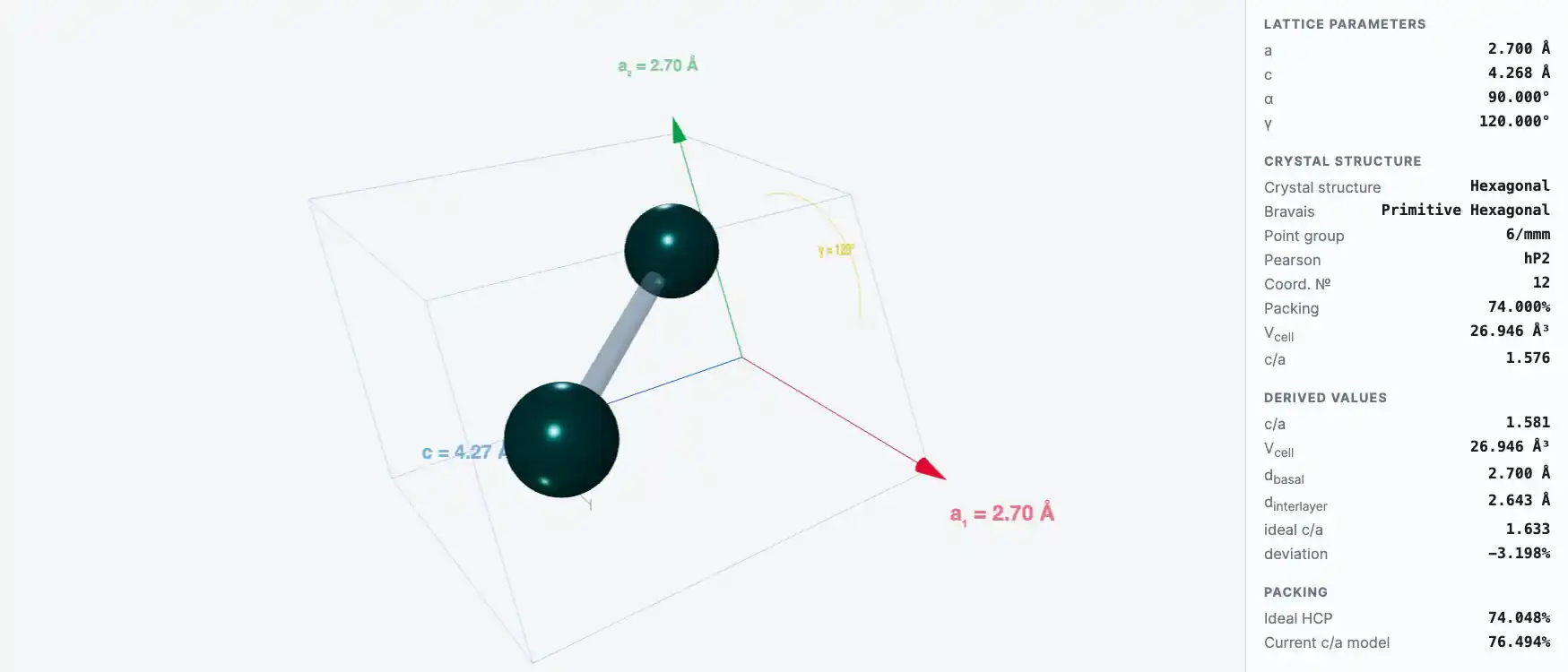
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Ru: 4d⁷ 5s¹[Kr] 4d⁷ 5s¹1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d⁷ 5s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 98 Stable | 97.9052868 ± 0.0000069 | 1.8700% | Stable |
| 99 Stable | 98.9059341 ± 0.0000011 | 12.7600% | Stable |
| 100 Stable | 99.9042143 ± 0.0000011 | 12.6000% | Stable |
| 101 Stable | 100.9055769 ± 0.0000012 | 17.0600% | Stable |
| 102 Stable | 101.9043441 ± 0.0000012 | 31.5500% | Stable |
| 104 Stable | 103.9054275 ± 0.0000028 | 18.6200% | Stable |
Phase / State
Reason: 2308.8 °C below melting point (2333.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 44 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Ru I | 0 | 541 | 11 | 519 |
| Ru II | +1 | 59 | 8 | 59 |
| Ru III | +2 | 93 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Ru I | 0 | 329 |
| Ru II | +1 | 235 |
| Ru III | +2 | 26 |
| Ru IV | +3 | 2 |
| Ru V | +4 | 2 |
| Ru VI | +5 | 2 |
| Ru VII | +6 | 2 |
| Ru VIII | +7 | 2 |
| Ru IX | +8 | 2 |
| Ru X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 68 pm |
| +4 | 6 | N/A | 62 pm |
| +5 | 6 | N/A | 56.49999999999999 pm |
| +7 | 4 | N/A | 38 pm |
| +8 | 4 | N/A | 36 pm |
Compounds
Isotopes (6)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 98 Stable | 97.9052868 ± 0.0000069 | 1.8700% ± 0.0300% | Stable | stable | |
| 99 Stable | 98.9059341 ± 0.0000011 | 12.7600% ± 0.1400% | Stable | stable | |
| 100 Stable | 99.9042143 ± 0.0000011 | 12.6000% ± 0.0700% | Stable | stable | |
| 101 Stable | 100.9055769 ± 0.0000012 | 17.0600% ± 0.0200% | Stable | stable | |
| 102 Stable | 101.9043441 ± 0.0000012 | 31.5500% ± 0.1400% | Stable | stable | |
| 104 Stable | 103.9054275 ± 0.0000028 | 18.6200% ± 0.2700% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 2606.15 K |
| Boiling point | 4420.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (10)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.9077 |
| 2 | p | 4.0492 |
| 2 | s | 11.6202 |
| 3 | d | 14.6411 |
| 3 | p | 16.7789 |
| 3 | s | 16.3988 |
| 4 | d | 31.1872 |
| 4 | p | 27.5652 |
| 4 | s | 26.344 |
| 5 | s | 37.5155 |
Crystal Radii Detail (5)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 82 | ||
| 4 | VI | 76 | from r^3 vs V plots, from metallic oxides, | |
| 5 | VI | 70.5 | estimated, from r^3 vs V plots, | |
| 7 | IV | 52 | ||
| 8 | IV | 50 |
Isotope Decay Modes (62)
| Isotope | Mode | Intensity |
|---|---|---|
| 85 | B+ | — |
| 85 | B+p | — |
| 85 | p | — |
| 86 | B+ | — |
| 86 | B+p | — |
| 87 | B+ | — |
| 87 | B+p | — |
| 88 | B+ | 100% |
| 88 | B+p | 3.6% |
| 89 | B+ | 100% |
X‑ray Scattering Factors (615)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.51919 |
| 10.1617 | — | 1.51438 |
| 10.3261 | — | 1.51486 |
| 10.4931 | — | 1.54335 |
| 10.6628 | — | 1.57238 |
| 10.8353 | — | 1.60195 |
| 11.0105 | — | 1.63207 |
| 11.1886 | — | 1.66277 |
| 11.3696 | — | 1.7032 |
| 11.5535 | — | 1.79614 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1×10-3 milligrams per kilogram
References (1)
- [5] Ruthenium https://education.jlab.org/itselemental/ele044.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
7×10-7 milligrams per liter
References (1)
- [5] Ruthenium https://education.jlab.org/itselemental/ele044.html
Sources
Sources of this element.
A member of the platinum group, ruthenium occurs native with other members of the group in ores found in the Ural mountains and in North and South America. It is also found along with other platinum metals in small but commercial quantities in pentlandite in the Sudbury, Ontario nickel-mining region, and in the pyroxinite deposits of South Africa.
References (1)
- [6] Ruthenium https://periodic.lanl.gov/44.shtml
Production
Production of this element (from raw materials or other compounds containing the element).
The metal is isolated commercially by a complex chemical process, the final stage of which is the hydrogen reduction of ammonium ruthenium chloride, which yields a powder. The powder is consolidated by powder metallurgy techniques or by argon-arc welding.
References (1)
- [6] Ruthenium https://periodic.lanl.gov/44.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Ruthenium.
The element property data was retrieved from publications.