Neon (Ne)
noble-gasGas
Standard Atomic Weight
20.1797 uElectron configuration
[He] 2s2 2p6Melting point
-248.59 °C (24.56 K)Boiling point
-246.05 °C (27.1 K)Density
0.8999 kg/m³Oxidation states
0Electronegativity (Pauling)
N/AIonization energy (1st)
Discovery year
1898Atomic radius
160 pmDetails
Neon is a noble gas and the second lightest member of group 18. It is monatomic, colorless, and chemically very inert under ordinary conditions because its outer electron shell is closed. In the atmosphere it is present only as a minor constituent, but it is readily recognized by the intense reddish-orange light emitted in low-pressure electrical discharges. Its technological importance rests mainly on this optical behavior and on its cryogenic properties.
Colourless gaseous element of group 18 on the periodic table (noble gases). Neon occurs in the atmosphere, and comprises 0.0018% of the volume of the atmosphere. It has a distinct reddish glow when used in discharge tubes and neon based lamps. It forms almost no chemical compounds. Neon was discovered in 1898 by Sir William Ramsey and M.W. Travers.
The name derives from the Greek neos for "new". It was discovered from its bright orange spectral lines by the Scottish chemist William Ramsay and the English chemist Morris William Travers in 1898 from a liquefied air sample.
Neon was discovered by Sir William Ramsay, a Scottish chemist, and Morris M. Travers, an English chemist, shortly after their discovery of the element krypton in 1898. Like krypton, neon was discovered through the study of liquefied air. Although neon is the fourth most abundant element in the universe, only 0.0018% of the earth's atmosphere is neon.
From the Greek word neos, new. Discovered by Ramsay and Travers in 1898. Neon is a rare gaseous element present in the atmosphere to the extent of 1 part in 65,000 of air. It is obtained by liquefaction of air and separated from the other gases by fractional distillation.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
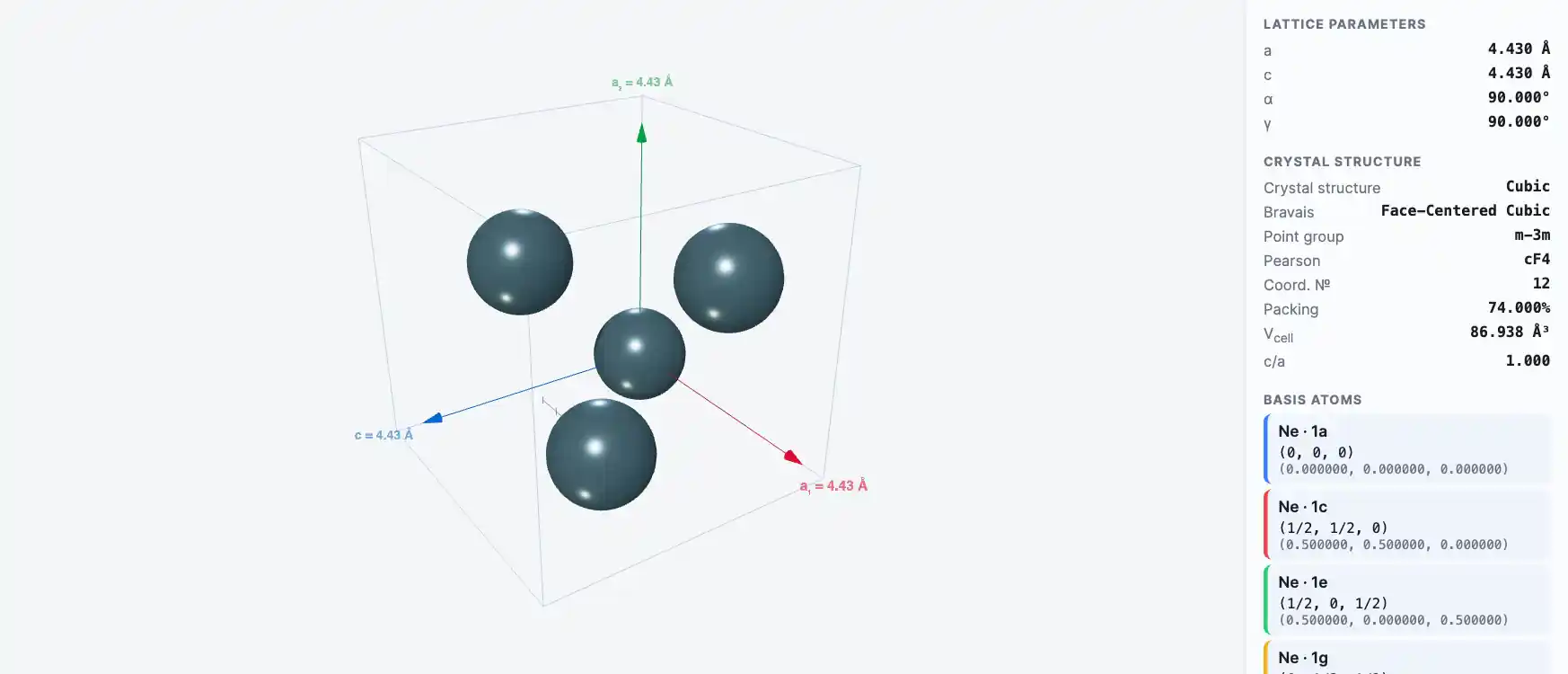
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Ne: 2s² 2p⁶[He] 2s² 2p⁶1s² 2s² 2p⁶Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 20 Stable | 19.9924401762 ± 0.0000000017 | 90.4800% | Stable |
| 21 Stable | 20.993846685 ± 0.000000041 | 0.2700% | Stable |
| 22 Stable | 21.991385114 ± 0.000000018 | 9.2500% | Stable |
Phase / State
Reason: 271.1 °C above boiling point (-246.05 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Density
At standard conditions
Estimated via ideal gas law at current T
Advanced
Atomic Spectra
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Ne I | 0 | 1597 | 533 | 1597 |
| Ne II | +1 | 1914 | 233 | 1912 |
| Ne III | +2 | 910 | 637 | 910 |
| Ne IV | +3 | 643 | 72 | 643 |
| Ne V | +4 | 374 | 139 | 374 |
| Ne VI | +5 | 515 | 449 | 515 |
| Ne VII | +6 | 661 | 442 | 661 |
| Ne VIII | +7 | 745 | 540 | 745 |
| Ne IX | +8 | 229 | 228 | 229 |
| Ne X | +9 | 137 | 137 | 137 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Ne I | 0 | 375 |
| Ne II | +1 | 385 |
| Ne III | +2 | 283 |
| Ne IV | +3 | 215 |
| Ne V | +4 | 161 |
| Ne VI | +5 | 135 |
| Ne VII | +6 | 208 |
| Ne VIII | +7 | 178 |
| Ne IX | +8 | 110 |
| Ne X | +9 | 149 |
Compounds
Isotopes (3)
Natural neon is a mixture of three isotopes. Six other unstable isotopes are known.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 20 Stable | 19.9924401762 ± 0.0000000017 | 90.4800% ± 0.0300% | Stable | stable | |
| 21 Stable | 20.993846685 ± 0.000000041 | 0.2700% ± 0.0100% | Stable | stable | |
| 22 Stable | 21.991385114 ± 0.000000018 | 9.2500% ± 0.0300% | Stable | stable |
Spectral Lines
Showing 50 of 1087 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 692.94673 nm | 100000 | Ne I | emission | 2s2.2p5.(2P*<1/2>).3s 2[1/2]* → 2s2.2p5.(2P*<3/2>).3p 2[3/2] | Measured | NIST | |
| 703.24131 nm | 85000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3s 2[3/2]* → 2s2.2p5.(2P*<3/2>).3p 2[1/2] | Measured | NIST | |
| 717.39381 nm | 77000 | Ne I | emission | 2s2.2p5.(2P*<1/2>).3s 2[1/2]* → 2s2.2p5.(2P*<3/2>).3p 2[5/2] | Measured | NIST | |
| 724.51666 nm | 77000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3s 2[3/2]* → 2s2.2p5.(2P*<3/2>).3p 2[1/2] | Measured | NIST | |
| 743.88984 nm | 60000 | Ne I | emission | 2s2.2p5.(2P*<1/2>).3s 2[1/2]* → 2s2.2p5.(2P*<3/2>).3p 2[1/2] | Measured | NIST | |
| 702.40504 nm | 34000 | Ne I | emission | 2s2.2p5.(2P*<1/2>).3s 2[1/2]* → 2s2.2p5.(2P*<3/2>).3p 2[3/2] | Measured | NIST | |
| 748.88712 nm | 32000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[1/2] → 2s2.2p5.(2P*<3/2>).3d 2[3/2]* | Measured | NIST | |
| 540.05618 nm | 20000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3s 2[3/2]* → 2s2.2p5.(2P*<1/2>).3p 2[1/2] | Measured | NIST | |
| 585.24879 nm | 20000 | Ne I | emission | 2s2.2p5.(2P*<1/2>).3s 2[1/2]* → 2s2.2p5.(2P*<1/2>).3p 2[1/2] | Measured | NIST | |
| 640.22472 nm | 20000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3s 2[3/2]* → 2s2.2p5.(2P*<3/2>).3p 2[5/2] | Measured | NIST | |
| 470.43948 nm | 15000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[1/2] → 2s2.2p5.(2P*<3/2>).5d 2[3/2]* | Measured | NIST | |
| 471.20625 nm | 15000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[5/2] → 2s2.2p5.(2P*<3/2>).6d 2[5/2]* | Measured | NIST | |
| 471.53441 nm | 15000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[5/2] → 2s2.2p5.(2P*<3/2>).6d 2[7/2]* | Measured | NIST | |
| 650.65281 nm | 15000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3s 2[3/2]* → 2s2.2p5.(2P*<3/2>).3p 2[5/2] | Measured | NIST | |
| 470.88584 nm | 12000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[1/2] → 2s2.2p5.(2P*<3/2>).5d 2[1/2]* | Measured | NIST | |
| 453.77551 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[1/2] → 2s2.2p5.(2P*<1/2>).5d 2[3/2]* | Measured | NIST | |
| 471.00638 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[1/2] → 2s2.2p5.(2P*<3/2>).5d 2[1/2]* | Measured | NIST | |
| 478.89249 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[5/2] → 2s2.2p5.(2P*<3/2>).7s 2[3/2]* | Measured | NIST | |
| 482.73382 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[1/2] → 2s2.2p5.(2P*<3/2>).6s 2[3/2]* | Measured | NIST | |
| 488.49181 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[5/2] → 2s2.2p5.(2P*<1/2>).5d 2[5/2]* | Measured | NIST | |
| 495.70324 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[3/2] → 2s2.2p5.(2P*<1/2>).5d 2[5/2]* | Measured | NIST | |
| 534.10932 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[1/2] → 2s2.2p5.(2P*<3/2>).4d 2[1/2]* | Measured | NIST | |
| 588.18952 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3s 2[3/2]* → 2s2.2p5.(2P*<1/2>).3p 2[1/2] | Measured | NIST | |
| 602.99969 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3s 2[3/2]* → 2s2.2p5.(2P*<1/2>).3p 2[1/2] | Measured | NIST | |
| 607.43377 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3s 2[3/2]* → 2s2.2p5.(2P*<3/2>).3p 2[1/2] | Measured | NIST | |
| 614.30626 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3s 2[3/2]* → 2s2.2p5.(2P*<3/2>).3p 2[3/2] | Measured | NIST | |
| 616.35939 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<1/2>).3s 2[1/2]* → 2s2.2p5.(2P*<1/2>).3p 2[1/2] | Measured | NIST | |
| 621.72812 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3s 2[3/2]* → 2s2.2p5.(2P*<3/2>).3p 2[3/2] | Measured | NIST | |
| 626.6495 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<1/2>).3s 2[1/2]* → 2s2.2p5.(2P*<1/2>).3p 2[3/2] | Measured | NIST | |
| 633.44278 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3s 2[3/2]* → 2s2.2p5.(2P*<3/2>).3p 2[5/2] | Measured | NIST | |
| 638.29917 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3s 2[3/2]* → 2s2.2p5.(2P*<3/2>).3p 2[3/2] | Measured | NIST | |
| 659.89529 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<1/2>).3s 2[1/2]* → 2s2.2p5.(2P*<1/2>).3p 2[1/2] | Measured | NIST | |
| 705.91074 nm | 10000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[1/2] → 2s2.2p5.(2P*<1/2>).3d 2[3/2]* | Measured | NIST | |
| 576.44189 nm | 7000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[5/2] → 2s2.2p5.(2P*<3/2>).4d 2[7/2]* | Measured | NIST | |
| 533.07771 nm | 6000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[1/2] → 2s2.2p5.(2P*<3/2>).4d 2[3/2]* | Measured | NIST | |
| 534.3282 nm | 6000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[1/2] → 2s2.2p5.(2P*<3/2>).4d 2[1/2]* | Measured | NIST | |
| 597.5534 nm | 6000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3s 2[3/2]* → 2s2.2p5.(2P*<1/2>).3p 2[3/2] | Measured | NIST | |
| 475.27311 nm | 5000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[5/2] → 2s2.2p5.(2P*<3/2>).6d 2[7/2]* | Measured | NIST | |
| 479.02171 nm | 5000 | Ne I | emission | 2s2.2p5.(2P*<1/2>).3p 2[3/2] → 2s2.2p5.(2P*<1/2>).6d 2[5/2]* | Measured | NIST | |
| 483.73128 nm | 5000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[1/2] → 2s2.2p5.(2P*<3/2>).6s 2[3/2]* | Measured | NIST | |
| 489.20896 nm | 5000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[3/2] → 2s2.2p5.(2P*<3/2>).7s 2[3/2]* | Measured | NIST | |
| 500.51582 nm | 5000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[3/2] → 2s2.2p5.(2P*<1/2>).5d 2[5/2]* | Measured | NIST | |
| 503.77504 nm | 5000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[5/2] → 2s2.2p5.(2P*<3/2>).5d 2[7/2]* | Measured | NIST | |
| 514.49371 nm | 5000 | Ne I | emission | 2s2.2p5.(2P*<1/2>).3p 2[3/2] → 2s2.2p5.(2P*<1/2>).5d 2[5/2]* | Measured | NIST | |
| 514.50308 nm | 5000 | Ne I | emission | 2s2.2p5.(2P*<1/2>).3p 2[3/2] → 2s2.2p5.(2P*<1/2>).5d 2[5/2]* | Measured | NIST | |
| 556.27668 nm | 5000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[5/2] → 2s2.2p5.(2P*<1/2>).4d 2[5/2]* | Measured | NIST | |
| 565.66578 nm | 5000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[3/2] → 2s2.2p5.(2P*<1/2>).4d 2[5/2]* | Measured | NIST | |
| 571.92256 nm | 5000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[3/2] → 2s2.2p5.(2P*<1/2>).4d 2[5/2]* | Measured | NIST | |
| 574.82979 nm | 5000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[5/2] → 2s2.2p5.(2P*<3/2>).4d 2[5/2]* | Measured | NIST | |
| 580.44496 nm | 5000 | Ne I | emission | 2s2.2p5.(2P*<3/2>).3p 2[5/2] → 2s2.2p5.(2P*<3/2>).4d 2[5/2]* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Noble Gas Properties
Phase Transitions & Allotropes
| Melting point | 24.56 K |
| Boiling point | 27.1 K |
| Critical point (temperature) | 44.49 K |
| Critical point (pressure) | 2.68 MPa |
| Triple point (temperature) | 24.56 K |
| Triple point (pressure) | 43.37 kPa |
Advanced Reference Data
Screening Constants (3)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.3579 |
| 2 | p | 4.2416 |
| 2 | s | 4.2416 |
Isotope Decay Modes (35)
| Isotope | Mode | Intensity |
|---|---|---|
| 15 | 2p | 100% |
| 16 | 2p | 100% |
| 17 | B+ | 100% |
| 17 | B+p | 94.4% |
| 17 | B+A | 3.5% |
| 17 | B+pA | 0% |
| 18 | B+ | 100% |
| 19 | B+ | 100% |
| 23 | B- | 100% |
| 24 | B- | 100% |
X‑ray Scattering Factors (503)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0 |
| 10.1617 | — | 0 |
| 10.3261 | — | 0 |
| 10.4931 | — | 0 |
| 10.6628 | — | 0 |
| 10.8353 | — | 0 |
| 11.0106 | — | 0 |
| 11.1886 | — | 0 |
| 11.3696 | — | 0 |
| 11.5535 | — | 0 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
5×10-3 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.2×10-4 milligrams per liter
References (1)
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Neon.
The element property data was retrieved from publications.