Protactinium (Pa)
actinideSolid
Standard Atomic Weight
231.03588 uElectron configuration
[Rn] 7s2 5f2 6d1Melting point
1571.85 °C (1845 K)Boiling point
N/ADensity
1.537000e+4 kg/m³Oxidation states
+2, +3, +4, +5Electronegativity (Pauling)
1.5Ionization energy (1st)
Discovery year
1913Atomic radius
180 pmDetails
Protactinium is a dense, silvery actinide metal with atomic number 91. It lies between thorium and uranium and is chemically notable for the stability of the +5 oxidation state, although +4 compounds are also known. All isotopes are radioactive. Natural protactinium occurs only in trace amounts, chiefly as ²³¹Pa in the ²³⁵U decay series and as short-lived products in other decay chains, so it has little technological role outside nuclear and geochemical research.
Protactinium metal is a dense, silvery-gray material with a bright metallic luster which it retains for some time in air but it does readily react with oxygen, water vapor and inorganic acids to form various compounds. In solid compounds protactinium is most stable in the oxidation state +5, but it also exists in the +4, +3 and +2 oxidation states. In solution the +5 state rapidly hydrolyzes by combining with hydroxide ions to form soluble or insoluble hydroxy-oxide solids which have a tendency to stick to the surfaces of vessels in which it is contained. A number of protactinium compounds are known, some of which are colored. The element is superconductive below 1.4K.
The name derives from the Greek protos (first) for preceding the element actinium, because its most common isotope (231Pa) decays to 227Ac by loss of an alpha particle.
In 1913 the German chemists K. Fajans and O. H. Gohring identified the first isotope of protactinium, 234Pa, and proposed the name brevium because of that isotope's short half-life of 6.7 h. 231Pa, with a longer half-life of 3.25(1)×104 a, was identified in 1918 by the German chemist O. Hahn and the Austrian physicist L. Meitner; and, independently in Britain, by F. Soddy and J. A. Cranston.
Protactinium was first identified by Kasimir Fajans and O.H. Göhring in 1913 while studying uranium's decay chain. The particular isotope they found, protactinium-234m, has a half-life of about 1.17 minutes. They named the element brevium, meaning brief, and then continued with their studies. Protactinium's existence was confirmed in 1918 when another isotope, protactinium-231, was independently discovered and studied by two groups of scientists, Otto Hahn and Lise Meitner of Germany and Frederick Soddy and John Cranston of Great Britain. Protactinium was first isolated by Aristid V. Grosse in 1934. Protactinium is a rare, poisonous and expensive element that is present in uranium ores in very small amounts. In 1961, the Great Britain Atomic Energy Authority was able to produce 125 grams of 99.9% pure protactinium, although they had to process about 55,000 kilograms of ore and spend about $500,000 to get it.
Protactinium's most stable isotope, protactinium-231, has a half-life of about 32,760 years. It decays into actinium-227 through alpha decay.
The name "protactinium" comes from adding the Greek protos meaning first, before the word "actinium." In 1871, Dmitri Mendeleevpredicted the existence of an element between thorium and uranium. In 1900, William Crookes isolated protactinium from uraniu. It was an intensely radioactive material, however, he could not characterize it as a new chemical element and thus named it uranium-X. In 1913 the first isotope of element 91, 234Pa, was discovered by K. Fajans and O.H. Gohring. It was a very short-lived member of the naturally occurring 238U decay series and as such they named it "brevium." In 1917/18, two groups of scientists, Otto Hahn and Lise Meitner of Germany and Frederick Soddy and John Cranston of Great Britain, independently discovered another isotope of protactinium, 231Pa having much longer half-life of about 32,000 years. The name was changed to proto-actinium as being more consistent with the longer-lived characteristics of the most abundant isotope. In 1927, Grosse prepared 2 mg of a white powder, which was shown to be Pa2O5. In 1934 he isolated the element from 0.1 g of pure Pa2O5 by two methods, one of which was by converting the oxide to an iodide and "cracking" it in a high vacuum by an electrically heated filament by the reaction: 2PaI5 > 2Pa + 5I2. In 1949, the name protoactinium was shortened by the IUPAC who officially named it protactinium and confirmed Hahn and Meitner as co-discoverers. The new name meant "parent of actinium" and reflected the fact that actinium is a decay product of the radioactive decay of protactinium.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
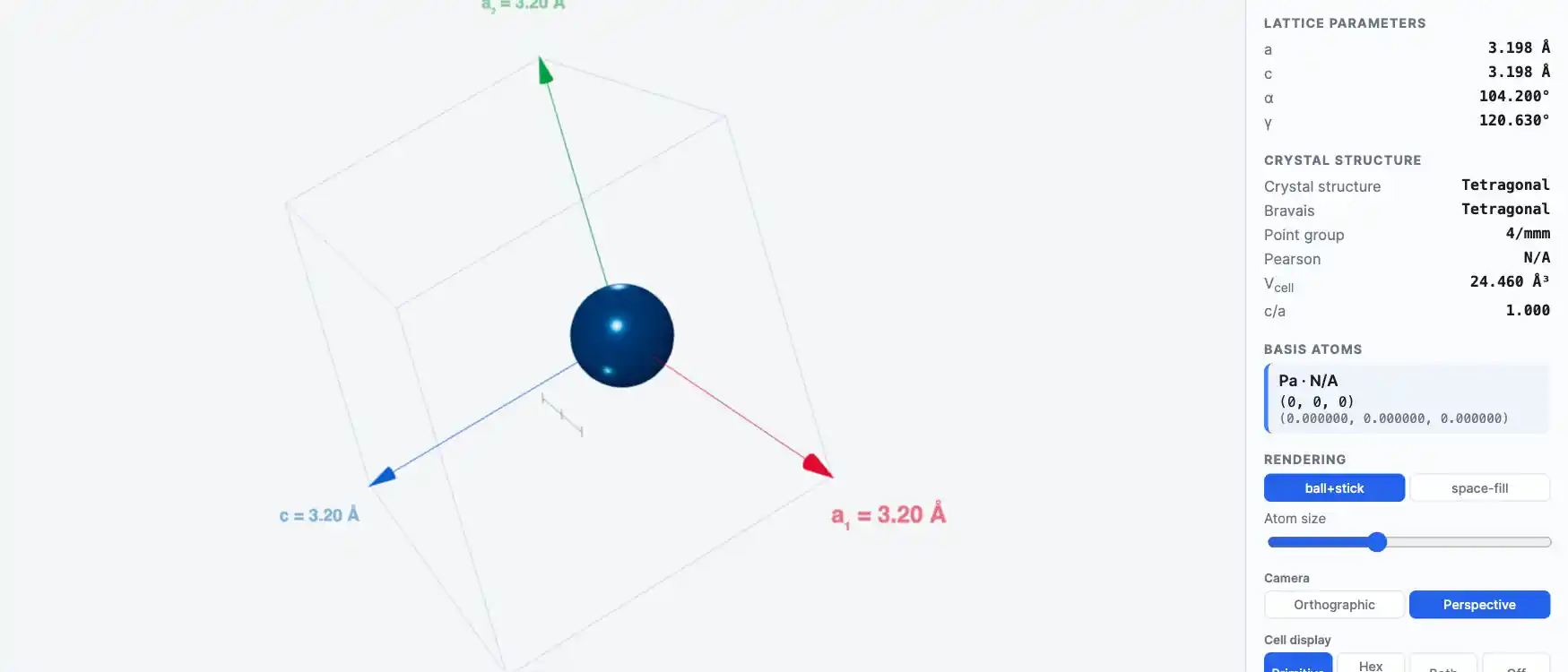
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Pa: 5f² 6d¹ 7s²[Rn] 5f² 6d¹ 7s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 5d¹⁰ 6s² 6p⁶ 5f² 6d¹ 7s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
No stable isotopes.
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 224 Radioactive | 224.0256176 ± 0.0000082 | N/A | 844 ms |
| 218 Radioactive | 218.020059 ± 0.00002 | N/A | 108 us |
| 216 Radioactive | 216.019109 ± 0.000057 | N/A | 105 ms |
| 219 Radioactive | 219.019904 ± 0.000055 | N/A | 56 ns |
| 227 Radioactive | 227.0288054 ± 0.000008 | N/A | 38.3 minutes |
Phase / State
Reason: 1546.8 °C below melting point (1571.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 91 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Pa I | 0 | 55 | 0 | 0 |
| Pa II | +1 | 33 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Pa I | 0 | 2 |
| Pa II | +1 | 2 |
| Pa III | +2 | 2 |
| Pa IV | +3 | 2 |
| Pa V | +4 | 2 |
| Pa VI | +5 | 2 |
| Pa VII | +6 | 2 |
| Pa VIII | +7 | 2 |
| Pa IX | +8 | 2 |
| Pa X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 104 pm |
| +3 | 9 | N/A | 119.9 pm |
| +4 | 6 | N/A | 90 pm |
| +4 | 8 | N/A | 101 pm |
| +5 | 6 | N/A | 78 pm |
| +5 | 8 | N/A | 91 pm |
| +5 | 9 | N/A | 95 pm |
Compounds
Isotopes (5)
Twenty-nine radioisotopes of protactinium have been discovered. Nearly all naturally occurring protactinium is 231Pa with a half-life of 32,700 years. It is an alpha emitter and is formed by the decay of uranium-235, whereas the beta radiating protactinium-234 with a half-life of 6.74 hours is produced as a result of uranium-238 decay. Nearly all uranium-238 (99.8%) decays first to the 234mPa isomer and then to 234Pa. Smaller trace amounts of the short-lived nuclear isomer protactinium-234m occur in the decay chain of uranium-238. Protactinium-233 results from the decay of thorium-233 as part of the chain of events used to produce uranium-233 by neutron irradiation of thorium-232.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 224 Radioactive | 224.0256176 ± 0.0000082 | N/A | 844 ms | α ≈100%β+ ? | |
| 218 Radioactive | 218.020059 ± 0.00002 | N/A | 108 us | α =100% | |
| 216 Radioactive | 216.019109 ± 0.000057 | N/A | 105 ms | α ≈100%β+ ? | |
| 219 Radioactive | 219.019904 ± 0.000055 | N/A | 56 ns | α =100%β+ ? | |
| 227 Radioactive | 227.0288054 ± 0.000008 | N/A | 38.3 minutes | α =85±0.2%ε =15±0.2% |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Phase Transitions & Allotropes
| Melting point | 1845.15 K |
Oxidation State Categories
Advanced Reference Data
Crystal Radii Detail (7)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 118 | estimated, | |
| 4 | VI | 104 | from r^3 vs V plots, | |
| 4 | VIII | 115 | ||
| 5 | VI | 92 | ||
| 5 | VIII | 105 | ||
| 5 | IX | 109 | ||
| 3 | IX | — | 133.9 |
Isotope Decay Modes (51)
| Isotope | Mode | Intensity |
|---|---|---|
| 211 | A | 100% |
| 211 | B+ | — |
| 211 | p | — |
| 212 | A | 100% |
| 213 | A | 100% |
| 214 | A | 100% |
| 215 | A | 100% |
| 216 | A | 100% |
| 216 | B+ | — |
| 217 | A | 100% |
X‑ray Scattering Factors (516)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.75788 |
| 10.1617 | — | 1.76101 |
| 10.3261 | — | 1.76414 |
| 10.4931 | — | 1.76728 |
| 10.6628 | — | 1.73466 |
| 10.8353 | — | 1.69295 |
| 11.0106 | — | 1.65224 |
| 11.1886 | — | 1.61457 |
| 11.3696 | — | 1.58512 |
| 11.5535 | — | 1.5562 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.4×10-6 milligrams per kilogram
References (1)
- [5] Protactinium https://education.jlab.org/itselemental/ele091.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
5×10-11 milligrams per liter
References (1)
- [5] Protactinium https://education.jlab.org/itselemental/ele091.html
Sources
Sources of this element.
Protactinium is one of the rarest and most expensive naturally occurring elements. The average concentrations of protactinium in the Earth's crust is typically on the order of a few parts per trillion, but may reach up to a few parts per million in some uraninite ore deposits. The element occurs in pitchblende to the extent of about 1 part 231Pa to 10 million parts of ore. Ores from Zaire have about 3 ppm. In 1959 and 1961, it was announced that the Great Britain Atomic Energy Authority extracted by a 12-stage process 125 g of 99.9% protactinium, the world's only stock of the metal for many years following. The extraction was made from 60 tons of waste material at a cost of about $500,000.
References (1)
- [6] Protactinium https://periodic.lanl.gov/91.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Protactinium.
The element property data was retrieved from publications.