Ytterbium (Yb)
lanthanideSolid
Standard Atomic Weight
173.054 uElectron configuration
[Xe] 6s2 4f14Melting point
818.85 °C (1092 K)Boiling point
1195.85 °C (1469 K)Density
6900 kg/m³Oxidation states
0, +1, +2, +3Electronegativity (Pauling)
N/AIonization energy (1st)
Discovery year
1878Atomic radius
175 pmDetails
Ytterbium is a soft, silvery lanthanide metal with atomic number 70. It is one of the heavier rare-earth elements and is chemically notable for the relative stability of the divalent Yb²⁺ state as well as the usual trivalent Yb³⁺ state. This accessible redox pair gives ytterbium a larger and more variable metallic radius than neighboring lanthanides and is important in its organometallic and solid-state chemistry. Natural ytterbium is a mixture of several stable isotopes.
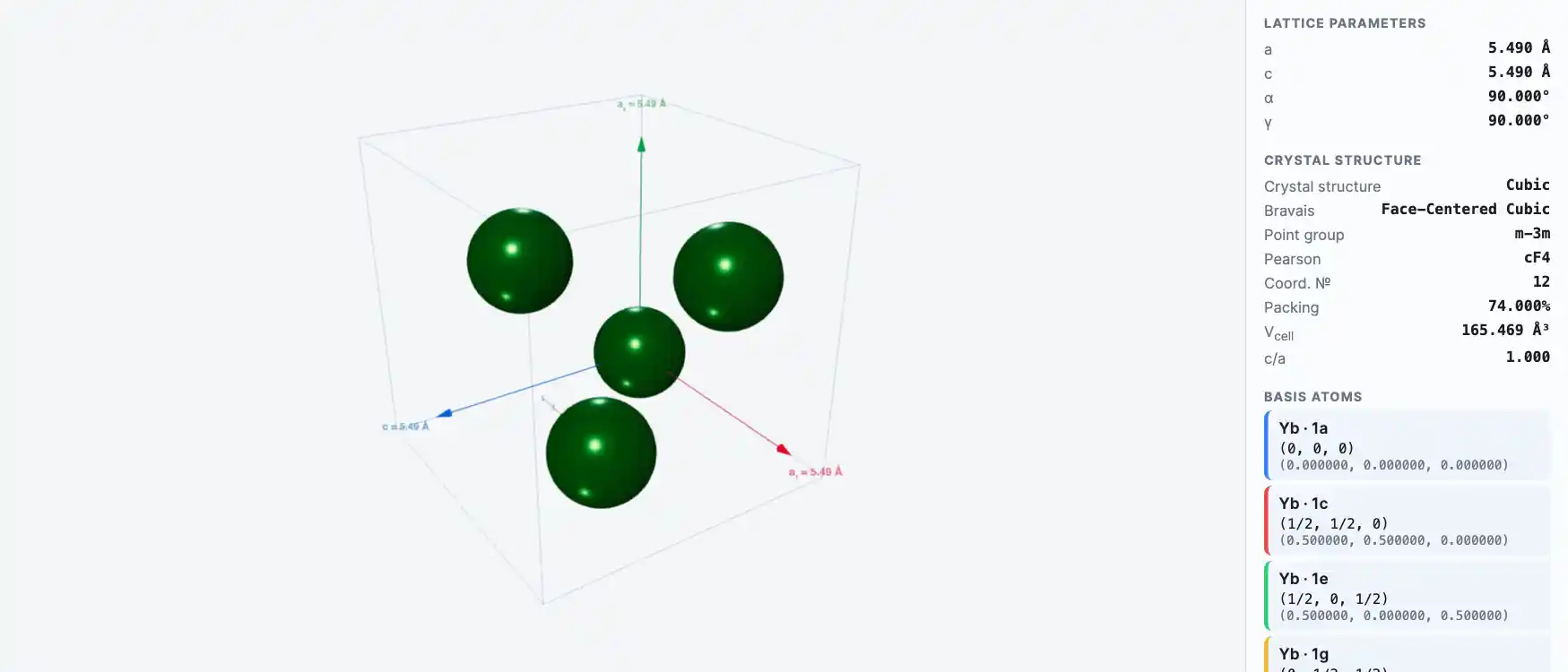
Ytterbium has a bright silvery luster, is soft, malleable, and quite ductile. Even though the element is fairly stable, it should be kept in closed containers to protect it from air and moisture. Ytterbium is readily attacked and dissolved by dilute and concentrated mineral acids and reacts slowly with water. Ytterbium has three allotropic forms with transformation points at -13°C and 795°C: The beta form is a room-temperature, face-centered, cubic modification, while the high-temperature gamma form is a body-centered cubic form. Another body-centered cubic phase has recently been found to be stable at high pressures at room temperatures. The beta form ordinarily has metallic-type conductivity, but becomes a semiconductor when the pressure is increased about 16,000 atm. The electrical resistance increases tenfold as the pressure is increased to 39,000 atm and drops to about 10% of its standard temperature-pressure resistivity at a pressure of 40,000 atm. Natural ytterbium is a mixture of seven stable isotopes. Seven other unstable isotopes are known.
The name derives from the Swedish village of Ytterby where the mineral ytterbite (the source of ytterbium) was originally found. It was discovered by the Swiss chemist Jean-Charles Galissard de Marignac in 1878 in erbium nitrate from gadolinite (ytterbite renamed).
The mineral gadolinite ((Ce, La, Nd, Y)2FeBe2Si2O10), discovered in a quarry near the town of Ytterby, Sweden, has been the source of a great number of rare earth elements. In 1843, Carl Gustaf Mosander, a Swedish chemist, was able to separate gadolinite into three materials, which he named yttria, erbia and terbia. As might be expected considering the similarities between their names and properties, scientists soon confused erbia and terbia and, by 1877, had reversed their names. What Mosander called erbia is now called terbia and visa versa. In 1878 Jean Charles Galissard de Marignac, a Swiss chemist, discovered that erbia was itself consisted of two components. One component was named ytterbia by Marignac while the other component retained the name erbia. Marignac believed that ytterbia was a compound of a new element, which he named ytterbium. Other chemists produced and experimented with ytterbium in an attempt to determine some of it's properties. Unfortunately, different scientists obtained different results from the same experiments. While some scientists believed that these inconsistent results were caused by poor procedures or faulty equipment, Georges Urbain, a French chemist, believed that ytterbium wasn't an element at all, but a mixture of two elements. In 1907, Urbain was able to separate ytterbium into two elements. Urbain named one of the elements neoytterbium (new ytterbium) and the other element lutecium. Chemists eventually changed the name neoytterbium back to ytterbium and changed the spelling of lutecium to lutetium. Due to his original belief of the composition of ytterbia, Marignac is credited with the discovery of ytterbium. Today, ytterbium is primarily obtained through an ion exchange process from monazite sand ((Ce, La, Th, Nd, Y)PO4), a material rich in rare earth elements.
Named after Ytterby, a village in Sweden. Marignac in 1878 discovered a new component, which he called ytterbia, in the earth then known as erbia. In 1907, Urbain separated ytterbia into two components, which he called neoytterbia and lutecia. The elements in these earths are now known as ytterbium and lutetium, respectively. These elements are identical with aldebaranium and cassiopeium, discovered independently and at about the same time by von Welsbach.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Yb: 4f¹⁴ 6s²[Xe] 4f¹⁴ 6s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹⁴ 6s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 170 Stable | 169.9347664 ± 0.0000022 | 2.9820% | Stable |
| 171 Stable | 170.9363302 ± 0.0000022 | 14.0900% | Stable |
| 172 Stable | 171.9363859 ± 0.0000022 | 21.6800% | Stable |
| 173 Stable | 172.9382151 ± 0.0000022 | 16.1030% | Stable |
| 174 Stable | 173.9388664 ± 0.0000022 | 32.0260% | Stable |
Phase / State
Reason: 793.9 °C below melting point (818.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 70 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Yb I | 0 | 99 | 5 | 10 |
| Yb II | +1 | 327 | 10 | 10 |
| Yb III | +2 | 272 | 0 | 0 |
| Yb IV | +3 | 92 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Yb I | 0 | 250 |
| Yb II | +1 | 349 |
| Yb III | +2 | 55 |
| Yb IV | +3 | 121 |
| Yb V | +4 | 2 |
| Yb VI | +5 | 2 |
| Yb VII | +6 | 2 |
| Yb VIII | +7 | 2 |
| Yb IX | +8 | 2 |
| Yb X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 6 | N/A | 102 pm |
| +2 | 7 | N/A | 108 pm |
| +2 | 8 | N/A | 113.99999999999999 pm |
| +3 | 6 | N/A | 86.8 pm |
| +3 | 7 | N/A | 92.5 pm |
| +3 | 8 | N/A | 98.5 pm |
| +3 | 9 | N/A | 104.2 pm |
Compounds
Isotopes (5)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 170 Stable | 169.9347664 ± 0.0000022 | 2.9820% ± 0.0390% | Stable | stable | |
| 171 Stable | 170.9363302 ± 0.0000022 | 14.0900% ± 0.1400% | Stable | stable | |
| 172 Stable | 171.9363859 ± 0.0000022 | 21.6800% ± 0.1300% | Stable | stable | |
| 173 Stable | 172.9382151 ± 0.0000022 | 16.1030% ± 0.0630% | Stable | stable | |
| 174 Stable | 173.9388664 ± 0.0000022 | 32.0260% ± 0.0800% | Stable | stable |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1097.15 K |
| Boiling point | 1469.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (13)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 1.3611 |
| 2 | p | 4.3716 |
| 2 | s | 18.306 |
| 3 | d | 13.6033 |
| 3 | p | 20.6635 |
| 3 | s | 21.2398 |
| 4 | d | 36.4104 |
| 4 | f | 40.568 |
| 4 | p | 33.598 |
| 4 | s | 32.4824 |
Crystal Radii Detail (7)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | VI | 116 | ||
| 2 | VII | 122 | estimated, | |
| 2 | VIII | 128 | ||
| 3 | VI | 100.8 | from r^3 vs V plots, | |
| 3 | VII | 106.5 | estimated, | |
| 3 | VIII | 112.5 | from r^3 vs V plots, | |
| 3 | IX | 118.2 | from r^3 vs V plots, |
Isotope Decay Modes (45)
| Isotope | Mode | Intensity |
|---|---|---|
| 148 | B+ | — |
| 148 | B+p | — |
| 149 | B+ | 100% |
| 149 | B+p | 100% |
| 150 | B+ | — |
| 151 | B+ | 100% |
| 151 | B+p | — |
| 152 | B+ | 100% |
| 153 | B+ | — |
| 153 | A | — |
X‑ray Scattering Factors (514)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.21734 |
| 10.1617 | — | 0.21864 |
| 10.3261 | — | 0.21994 |
| 10.4931 | — | 0.22125 |
| 10.6628 | — | 0.22256 |
| 10.8353 | — | 0.22389 |
| 11.0106 | — | 0.22522 |
| 11.1886 | — | 0.22656 |
| 11.3696 | — | 0.22886 |
| 11.5535 | — | 0.23378 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
3.2 milligrams per kilogram
References (1)
- [5] Ytterbium https://education.jlab.org/itselemental/ele070.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
8.2×10-7 milligrams per liter
References (1)
- [5] Ytterbium https://education.jlab.org/itselemental/ele070.html
Sources
Sources of this element.
Ytterbium occurs along with other rare earths in a number of rare minerals. It is commercially recovered principally from monazite sand, which contains about 0.03%. Ion-exchange and solvent extraction techniques developed in recent years have greatly simplified the separation of the rare earths from one another.
References (1)
- [6] Ytterbium https://periodic.lanl.gov/70.shtml
Production
Production of this element (from raw materials or other compounds containing the element).
The element was first prepared by Klemm and Bonner in 1937 by reducing ytterbium trichloride with potassium. Their metal was mixed, however, with KCl. Daane, Dennison, and Spedding prepared a much purer from in 1953 from which the chemical and physical properties of the element could be determined.
References (1)
- [6] Ytterbium https://periodic.lanl.gov/70.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Ytterbium.
The element property data was retrieved from publications.