Boron (B)
metalloidSolid
Standard Atomic Weight
10.81 u [10.806, 10.821]Electron configuration
[He] 2s2 2p1Melting point
2074.85 °C (2348 K)Boiling point
3999.85 °C (4273 K)Density
2370 kg/m³Oxidation states
−5, −1, 0, +1, +2, +3Electronegativity (Pauling)
2.04Ionization energy (1st)
Discovery year
1808Atomic radius
85 pmDetails
Boron is a light metalloid in group 13, notable for electron-deficient bonding and a rich cluster chemistry. It occurs naturally only in compounds, mainly as borates in evaporite minerals and brines. Elemental boron is difficult to prepare in high purity and has several allotropes built from B₁₂ icosahedra. Technologically, boron is most important through borate minerals, borosilicate glass, detergents, ceramics, fertilizers, and neutron-absorbing materials.
An element of group 13 of the periodic table. There are two allotropes, amorphous boron is a brown power, but metallic boron is black. The metallic form is hard (9.3 on Mohs' scale) and a bad conductor in room temperatures. It is never found free in nature. Boron-10 is used in nuclear reactor control rods and shields. It was discovered in 1808 by Sir Humphry Davy and by J.L. Gay-Lussac and L.J. Thenard.
The name derives from the Arabic buraq for "white". Although its compounds were known for thousands of years, it was not isolated until 1808 by the French chemists Louis-Joseph Gay-Lussac and Louis-Jacques Thenard.
Boron was discovered by Joseph-Louis Gay-Lussac and Louis-Jaques Thénard, French chemists, and independently by Sir Humphry Davy, an English chemist, in 1808. They all isolated boron by combining boric acid (H3BO3) with potassium. Today, boron is obtained by heating borax (Na2B4O7·10H2O) with carbon, although other methods are used if high-purity boron is required.
From the Arabic word Buraq, Persian Burah. Boron compounds have been known for thousands of years, but the element was not discovered until 1808 by Sir Humphry Davy and by Gay-Lussac and Thenard.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
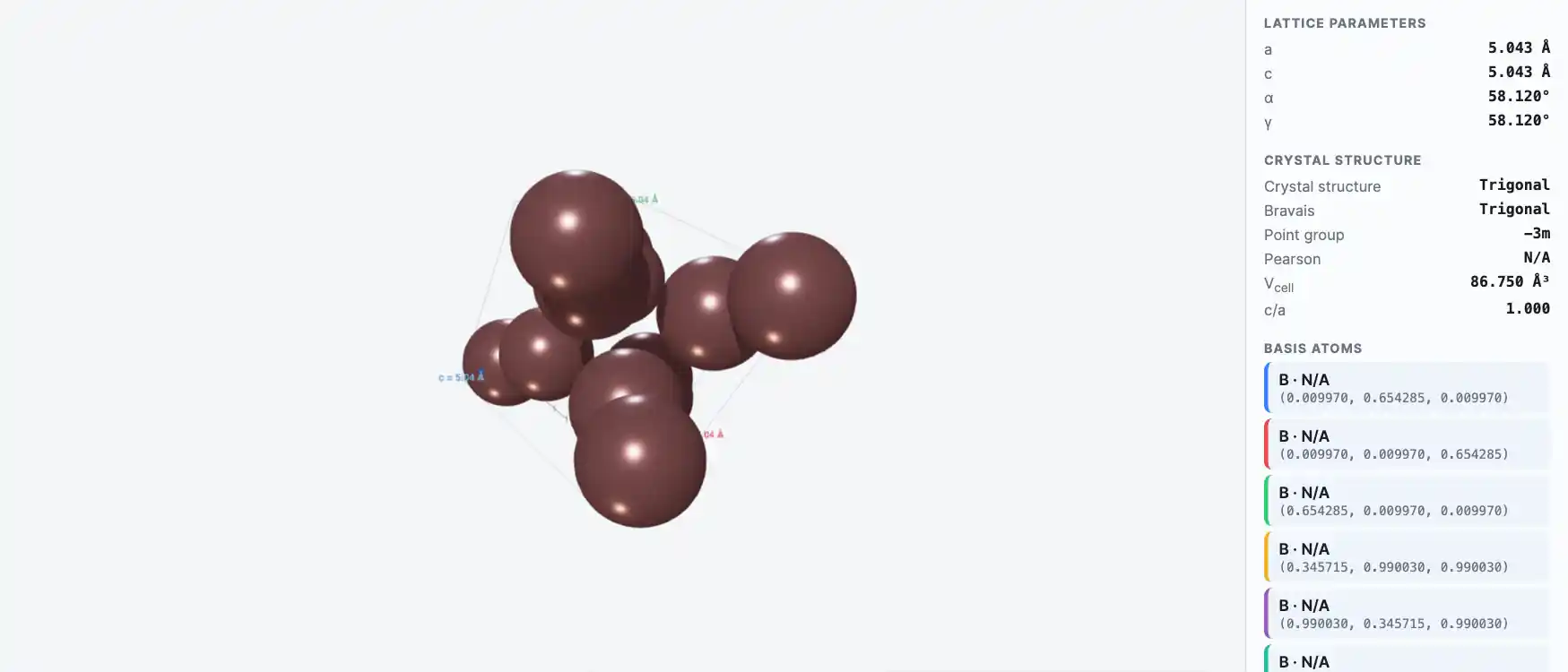
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
B: 2s² 2p¹[He] 2s² 2p¹1s² 2s² 2p¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 10 Stable | 10.01293695 ± 0.00000041 | 19.9000% | Stable |
| 11 Stable | 11.00930536 ± 0.00000045 | 80.1000% | Stable |
Phase / State
Reason: 2049.8 °C below melting point (2074.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| B I | 0 | 371 | 269 | 371 |
| 11B I Isotope | 0 | 53 | 0 | 53 |
| 10B I Isotope | 0 | 11 | 0 | 11 |
| B II | +1 | 592 | 435 | 592 |
| 10B II Isotope | +1 | 9 | 0 | 9 |
| 11B II Isotope | +1 | 9 | 0 | 9 |
| B III | +2 | 390 | 106 | 390 |
| B IV | +3 | 478 | 234 | 478 |
| B V | +4 | 258 | 240 | 258 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| B I | 0 | 125 |
| 11B I Isotope | 0 | 69 |
| 10B I Isotope | 0 | 29 |
| B II | +1 | 157 |
| 10B II Isotope | +1 | 10 |
| 11B II Isotope | +1 | 10 |
| B III | +2 | 150 |
| B IV | +3 | 174 |
| B V | +4 | 101 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 3 | N/A | 1 pm |
| +3 | 4 | N/A | 11 pm |
| +3 | 6 | N/A | 27 pm |
Compounds
Isotopes (2)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 10 Stable | 10.01293695 ± 0.00000041 | 19.9000% ± 0.7000% | Stable | stable | |
| 11 Stable | 11.00930536 ± 0.00000045 | 80.1000% ± 0.7000% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 391.482 nm | N/A | B II | emission | 1s2.2s.2p 1P* → 1s2.2p2 3P | Measured | NIST | |
| 391.687 nm | N/A | B II | emission | 1s2.2s.2p 1P* → 1s2.2p2 3P | Measured | NIST | |
| 391.817 nm | N/A | B II | emission | 1s2.2s.2p 1P* → 1s2.2p2 3P | Measured | NIST | |
| 394.447 nm | N/A | B II | emission | 1s2.2p.3d 3F* → 1s2.2p.4f 3F | Measured | NIST | |
| 394.587 nm | N/A | B II | emission | 1s2.2p.3d 3F* → 1s2.2p.4f 3F | Measured | NIST | |
| 394.82 nm | N/A | B II | emission | 1s2.2p.3d 3F* → 1s2.2p.4f 3F | Measured | NIST | |
| 395.038 nm | 18 | B II | emission | 1s2.2p.3d 1D* → 1s2.2p.4f 1F | Measured | NIST | |
| 395.1698 nm | N/A | B II | emission | 1s2.2p2 1D → 1s2.2p2 1S | Measured | NIST | |
| 399.024 nm | 70 | B II | emission | 1s2.2s.4p 1P* → 1s2.2s.8d 1D | Measured | NIST | |
| 400.017 nm | 136 | B III | emission | 1s.2s.(3S).4d 4D → 1s.2s.(3S).5f 4F* | Measured | NIST | |
| 412.1928 nm | N/A | B II | emission | 1s2.2s.3d 3D → 1s2.2s.4f 3F* | Measured | NIST | |
| 412.1928 nm | N/A | B II | emission | 1s2.2s.3d 3D → 1s2.2s.4f 3F* | Measured | NIST | |
| 412.1928 nm | N/A | B II | emission | 1s2.2s.3d 3D → 1s2.2s.4f 3F* | Measured | NIST | |
| 412.1928 nm | N/A | B II | emission | 1s2.2s.3d 3D → 1s2.2s.4f 3F* | Measured | NIST | |
| 412.1928 nm | N/A | B II | emission | 1s2.2s.3d 3D → 1s2.2s.4f 3F* | Measured | NIST | |
| 412.1928 nm | N/A | B II | emission | 1s2.2s.3d 3D → 1s2.2s.4f 3F* | Measured | NIST | |
| 414.697 nm | N/A | B III | emission | 1s2.5d 2D → 1s2.8f 2F* | Measured | NIST | |
| 414.708 nm | N/A | B III | emission | 1s2.5d 2D → 1s2.8f 2F* | Measured | NIST | |
| 414.708 nm | N/A | B III | emission | 1s2.5d 2D → 1s2.8f 2F* | Measured | NIST | |
| 415.284 nm | N/A | B II | emission | 1s2.2s.4p 3P* → 1s2.2p.3p 3P | Measured | NIST | |
| 415.284 nm | N/A | B II | emission | 1s2.2s.4p 3P* → 1s2.2p.3p 3P | Measured | NIST | |
| 415.471 nm | N/A | B II | emission | 1s2.2s.4p 3P* → 1s2.2p.3p 3P | Measured | NIST | |
| 415.471 nm | N/A | B II | emission | 1s2.2s.4p 3P* → 1s2.2p.3p 3P | Measured | NIST | |
| 415.471 nm | N/A | B II | emission | 1s2.2s.4p 3P* → 1s2.2p.3p 3P | Measured | NIST | |
| 415.584 nm | N/A | B II | emission | 1s2.2s.4p 3P* → 1s2.2p.3p 3P | Measured | NIST | |
| 417.896 nm | N/A | B I | emission | 2s2.3p 2P* → 2s.2p2 2P | Measured | NIST | |
| 417.927 nm | N/A | B I | emission | 2s2.3p 2P* → 2s.2p2 2P | Measured | NIST | |
| 418.099 nm | N/A | B I | emission | 2s2.3p 2P* → 2s.2p2 2P | Measured | NIST | |
| 418.13 nm | N/A | B I | emission | 2s2.3p 2P* → 2s.2p2 2P | Measured | NIST | |
| 419.4792 nm | 180 | B II | emission | 1s2.2s.3p 1P* → 1s2.2s.4s 1S | Measured | NIST | |
| 419.773 nm | 30 | B IV | emission | 1s.5s 3S → 1s.6p 3P* | Measured | NIST | |
| 424.3 nm | 300 | B III | emission | 1s2.4p 2P* → 1s2.5d 2D | Measured | NIST | |
| 424.359 nm | N/A | B III | emission | 1s2.4p 2P* → 1s2.5d 2D | Measured | NIST | |
| 424.37 nm | N/A | B III | emission | 1s2.4p 2P* → 1s2.5d 2D | Measured | NIST | |
| 427.274 nm | 50 | B II | emission | 1s2.2s.4s 3S → 1s2.2s.6p 3P* | Measured | NIST | |
| 429.571 nm | 50 | B II | emission | 1s2.2s.4p 1P* → 1s2.2s.7d 1D | Measured | NIST | |
| 436.147 nm | 60 | B III | emission | 1s.2p.(3P*).4f 2F → 1s.2p.(3P*).5g 2G* | Measured | NIST | |
| 436.61 nm | 100 | B III | emission | 1s.2p.(3P*).4f 4F → 1s.2p.(3P*).5g 4G* | Measured | NIST | |
| 443.11 nm | N/A | B II | emission | 1s2.2p.3d 3D* → 1s2.2p.4f 3F | Measured | NIST | |
| 443.185 nm | N/A | B II | emission | 1s2.2p.3d 3D* → 1s2.2p.4f 3F | Measured | NIST | |
| 443.291 nm | N/A | B II | emission | 1s2.2p.3d 3D* → 1s2.2p.4f 3F | Measured | NIST | |
| 445.943 nm | N/A | B IV | emission | 1s.5p 3P* → 1s.6d 3D | Measured | NIST | |
| 445.943 nm | N/A | B IV | emission | 1s.5p 3P* → 1s.6d 3D | Measured | NIST | |
| 445.943 nm | N/A | B IV | emission | 1s.5p 3P* → 1s.6d 3D | Measured | NIST | |
| 447.112 nm | N/A | B III | emission | 1s2.5s 2S → 1s2.7p 2P* | Measured | NIST | |
| 447.112 nm | N/A | B III | emission | 1s2.5s 2S → 1s2.7p 2P* | Measured | NIST | |
| 447.2029 nm | N/A | B II | emission | 1s2.2s.3p 3P* → 1s2.2s.4s 3S | Measured | NIST | |
| 447.2151 nm | N/A | B II | emission | 1s2.2s.3p 3P* → 1s2.2s.4s 3S | Measured | NIST | |
| 447.2862 nm | 470 | B II | emission | 1s2.2s.3p 3P* → 1s2.2s.4s 3S | Measured | NIST | |
| 448.692 nm | N/A | B III | emission | 1s2.4d 2D → 1s2.5f 2F* | Measured | NIST | |
| 448.71 nm | N/A | B III | emission | 1s2.4d 2D → 1s2.5f 2F* | Measured | NIST | |
| 449.09 nm | 20 | B IV | emission | 1s.2s 1S → 1s.2p 1P* | Measured | NIST | |
| 449.773 nm | 1700 | B III | emission | 1s2.4f 2F* → 1s2.5g 2G | Measured | NIST | |
| 449.853 nm | N/A | B III | emission | 1s2.4f 2F* → 1s2.5f 2F* | Measured | NIST | |
| 449.859 nm | N/A | B III | emission | 1s2.4f 2F* → 1s2.5f 2F* | Measured | NIST | |
| 450.481 nm | N/A | B III | emission | 1s2.4f 2F* → 1s2.5d 2D | Measured | NIST | |
| 450.482 nm | N/A | B III | emission | 1s2.4f 2F* → 1s2.5d 2D | Measured | NIST | |
| 451.9912773 nm | N/A | B V | emission | 7i 2I → 9k 2K* | Measured | NIST | |
| 451.9946377 nm | N/A | B V | emission | 7i 2I → 9k 2K* | Measured | NIST | |
| 453.229 nm | N/A | B II | emission | 1s2.2s.4f 1F* → 1s2.2p.3p 1D | Measured | NIST | |
| 459.72 nm | N/A | B III | emission | 1s.2s.(3S).4d 4D → 1s.2s.(3S).5p 4P* | Measured | NIST | |
| 459.73 nm | N/A | B III | emission | 1s.2p.(3P*).4p 4P → 1s.2p.(3P*).5s 4P* | Measured | NIST | |
| 461.114 nm | N/A | B II | emission | 1s2.2s.4p 3P* → 1s2.2p.3p 3S | Measured | NIST | |
| 461.114 nm | N/A | B II | emission | 1s2.2s.4p 3P* → 1s2.2p.3p 3S | Measured | NIST | |
| 461.114 nm | N/A | B II | emission | 1s2.2s.4p 3P* → 1s2.2p.3p 3S | Measured | NIST | |
| 461.32 nm | N/A | B IV | emission | 1s.5d 3D → 1s.6p 1P* | Measured | NIST | |
| 463.217 nm | N/A | B III | emission | 1s2.4d 2D → 1s2.5p 2P* | Measured | NIST | |
| 463.243 nm | N/A | B III | emission | 1s2.4d 2D → 1s2.5p 2P* | Measured | NIST | |
| 463.263 nm | N/A | B III | emission | 1s2.4d 2D → 1s2.5p 2P* | Measured | NIST | |
| 464.69 nm | N/A | B IV | emission | 1s.5d 3D → 1s.6f 1F* | Measured | NIST | |
| 464.69 nm | N/A | B IV | emission | 1s.5d 3D → 1s.6f 1F* | Measured | NIST | |
| 464.701 nm | N/A | B IV | emission | 1s.5d 3D → 1s.6f 3F* | Measured | NIST | |
| 464.701 nm | N/A | B IV | emission | 1s.5d 3D → 1s.6f 3F* | Measured | NIST | |
| 464.701 nm | N/A | B IV | emission | 1s.5d 3D → 1s.6f 3F* | Measured | NIST | |
| 464.701 nm | N/A | B IV | emission | 1s.5d 3D → 1s.6f 3F* | Measured | NIST | |
| 464.701 nm | N/A | B IV | emission | 1s.5d 3D → 1s.6f 3F* | Measured | NIST | |
| 464.701 nm | N/A | B IV | emission | 1s.5d 3D → 1s.6f 3F* | Measured | NIST | |
| 465.58 nm | N/A | B IV | emission | 1s.5d 1D → 1s.6f 1F* | Measured | NIST | |
| 465.786 nm | N/A | B IV | emission | 1s.5f 3F* → 1s.6g 3G | Measured | NIST | |
| 465.786 nm | N/A | B IV | emission | 1s.5f 3F* → 1s.6g 3G | Measured | NIST | |
| 465.786 nm | N/A | B IV | emission | 1s.5f 3F* → 1s.6g 3G | Measured | NIST | |
| 465.8 nm | N/A | B IV | emission | 1s.5f 1F* → 1s.6g 3G | Measured | NIST | |
| 465.815 nm | N/A | B IV | emission | 1s.5g 3G → 1s.6h 3H* | Measured | NIST | |
| 465.815 nm | N/A | B IV | emission | 1s.5g 3G → 1s.6h 3H* | Measured | NIST | |
| 465.815 nm | N/A | B IV | emission | 1s.5g 3G → 1s.6h 3H* | Measured | NIST | |
| 465.815 nm | N/A | B IV | emission | 1s.5g 1G → 1s.6h 3H* | Measured | NIST | |
| 465.92 nm | N/A | B IV | emission | 1s.5g 1G → 1s.6f 1F* | Measured | NIST | |
| 465.92 nm | N/A | B IV | emission | 1s.5g 3G → 1s.6f 1F* | Measured | NIST | |
| 465.92 nm | N/A | B IV | emission | 1s.5g 3G → 1s.6f 1F* | Measured | NIST | |
| 465.927 nm | N/A | B IV | emission | 1s.5g 3G → 1s.6f 3F* | Measured | NIST | |
| 465.927 nm | N/A | B IV | emission | 1s.5g 3G → 1s.6f 3F* | Measured | NIST | |
| 465.927 nm | N/A | B IV | emission | 1s.5g 3G → 1s.6f 3F* | Measured | NIST | |
| 465.927 nm | N/A | B IV | emission | 1s.5g 1G → 1s.6f 3F* | Measured | NIST | |
| 468.31 nm | N/A | B IV | emission | 1s.5p 1P* → 1s.6p 1P* | Measured | NIST | |
| 468.481 nm | N/A | B IV | emission | 1s.6g 3G → 1s.8h 3H* | Measured | NIST | |
| 468.489 nm | N/A | B IV | emission | 1s.6f 3F* → 1s.8g 3G | Measured | NIST | |
| 468.489 nm | N/A | B IV | emission | 1s.6f 3F* → 1s.8g 3G | Measured | NIST | |
| 468.489 nm | N/A | B IV | emission | 1s.6f 3F* → 1s.8g 3G | Measured | NIST | |
| 468.5 nm | N/A | B IV | emission | 1s.6h 3H* → 1s.8i 3I | Measured | NIST | |
| 471.612 nm | 15 | B II | emission | 1s2.2p.3d 1D* → 1s2.2p.4p 1P | Measured | NIST | |
| 471.99 nm | N/A | B IV | emission | 1s.5p 1P* → 1s.6d 1D | Measured | NIST | |
| 477.384 nm | N/A | B IV | emission | 1s.5d 3D → 1s.6p 3P* | Measured | NIST | |
| 477.384 nm | N/A | B IV | emission | 1s.5d 3D → 1s.6p 3P* | Measured | NIST | |
| 477.384 nm | N/A | B IV | emission | 1s.5d 3D → 1s.6p 3P* | Measured | NIST | |
| 478.42 nm | N/A | B II | emission | 1s2.2s.3d 3D → 1s2.2s.4p 3P* | Measured | NIST | |
| 478.42 nm | N/A | B II | emission | 1s2.2s.3d 3D → 1s2.2s.4p 3P* | Measured | NIST | |
| 478.4203 nm | N/A | B II | emission | 1s2.2s.3d 3D → 1s2.2s.4p 3P* | Measured | NIST | |
| 478.4203 nm | N/A | B II | emission | 1s2.2s.3d 3D → 1s2.2s.4p 3P* | Measured | NIST | |
| 478.4203 nm | N/A | B II | emission | 1s2.2s.3d 3D → 1s2.2s.4p 3P* | Measured | NIST | |
| 478.4203 nm | N/A | B II | emission | 1s2.2s.3d 3D → 1s2.2s.4p 3P* | Measured | NIST | |
| 481.276 nm | N/A | B IV | emission | 1s.5p 3P* → 1s.6s 3S | Measured | NIST | |
| 481.276 nm | N/A | B IV | emission | 1s.5p 3P* → 1s.6s 3S | Measured | NIST | |
| 481.276 nm | N/A | B IV | emission | 1s.5p 3P* → 1s.6s 3S | Measured | NIST | |
| 491.746 nm | 500 | B III | emission | 1s2.4p 2P* → 1s2.5s 2S | Measured | NIST | |
| 491.84 nm | 500 | B III | emission | 1s2.4p 2P* → 1s2.5s 2S | Measured | NIST | |
| 494.0365 nm | 440 | B II | emission | 1s2.2s.3d 1D → 1s2.2s.4f 1F* | Measured | NIST | |
| 494.4788284 nm | N/A | B V | emission | 6h 2H* → 7i 2I | Measured | NIST | |
| 494.4864305 nm | N/A | B V | emission | 6h 2H* → 7i 2I | Measured | NIST | |
| 498.848 nm | N/A | B III | emission | 1s2.5p 2P* → 1s2.7d 2D | Measured | NIST | |
| 498.901 nm | N/A | B III | emission | 1s2.5p 2P* → 1s2.7d 2D | Measured | NIST | |
| 512.579 nm | N/A | B II | emission | 1s2.2s.4d 3D → 1s2.2s.7f 3F* | Measured | NIST | |
| 512.579 nm | N/A | B II | emission | 1s2.2s.4d 3D → 1s2.2s.7f 3F* | Measured | NIST | |
| 512.579 nm | N/A | B II | emission | 1s2.2s.4d 3D → 1s2.2s.7f 3F* | Measured | NIST | |
| 515.776 nm | N/A | B III | emission | 1s2.5d 2D → 1s2.7f 2F* | Measured | NIST | |
| 515.793 nm | N/A | B III | emission | 1s2.5d 2D → 1s2.7f 2F* | Measured | NIST | |
| 516.57 nm | N/A | B III | emission | 1s2.5f 2F* → 1s2.7g 2G | Measured | NIST | |
| 516.579 nm | N/A | B III | emission | 1s2.5f 2F* → 1s2.7g 2G | Measured | NIST | |
| 522.65 nm | N/A | B III | emission | 1s2.5d 2D → 1s2.7p 2P* | Measured | NIST | |
| 522.65 nm | N/A | B III | emission | 1s2.5d 2D → 1s2.7p 2P* | Measured | NIST | |
| 522.667 nm | N/A | B III | emission | 1s2.5d 2D → 1s2.7p 2P* | Measured | NIST | |
| 526.311 nm | N/A | B II | emission | 1s2.2s.4f 3F* → 1s2.2s.7g 3G | Measured | NIST | |
| 526.311 nm | N/A | B II | emission | 1s2.2s.4f 3F* → 1s2.2s.7g 3G | Measured | NIST | |
| 526.311 nm | N/A | B II | emission | 1s2.2s.4f 3F* → 1s2.2s.7g 3G | Measured | NIST | |
| 529.28 nm | N/A | B III | emission | 1s2.5p 2P* → 1s2.7s 2S | Measured | NIST | |
| 529.34 nm | N/A | B III | emission | 1s2.5p 2P* → 1s2.7s 2S | Measured | NIST | |
| 534.765 nm | 15 | B II | emission | 1s2.2s.4s 1S → 1s2.2p.3s 1P* | Measured | NIST | |
| 539.322 nm | 30 | B II | emission | 1s2.2s.4p 1P* → 1s2.2p.3p 1P | Measured | NIST | |
| 550.4527 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.11f 2F* | Measured | NIST | |
| 550.4622 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.11f 2F* | Measured | NIST | |
| 556.3146 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.10f 2F* | Measured | NIST | |
| 556.3244 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.10f 2F* | Measured | NIST | |
| 563.30717 nm | N/A | B I | emission | 2s2.3s 2S → 2s2.4p 2P* | Measured | NIST | |
| 563.32732 nm | N/A | B I | emission | 2s2.3s 2S → 2s2.4p 2P* | Measured | NIST | |
| 564.4278 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.9f 2F* | Measured | NIST | |
| 564.4379 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.9f 2F* | Measured | NIST | |
| 576.1901 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.8f 2F* | Measured | NIST | |
| 576.1901 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.8f 2F* | Measured | NIST | |
| 576.2006 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.8f 2F* | Measured | NIST | |
| 578.747 nm | N/A | B II | emission | 1s2.2s.4s 3S → 1s2.2s.5p 3P* | Measured | NIST | |
| 578.747 nm | N/A | B II | emission | 1s2.2s.4s 3S → 1s2.2s.5p 3P* | Measured | NIST | |
| 578.747 nm | N/A | B II | emission | 1s2.2s.4s 3S → 1s2.2s.5p 3P* | Measured | NIST | |
| 581.833 nm | 60 | B I | emission | 2s.2p2 2P → 2s.2p.(3P*).3d 2D* | Measured | NIST | |
| 582.116 nm | 100 | B I | emission | 2s.2p2 2P → 2s.2p.(3P*).3d 2D* | Measured | NIST | |
| 582.228 nm | 10 | B I | emission | 2s.2p2 2P → 2s.2p.(3P*).3d 2D* | Measured | NIST | |
| 594.2619 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.7f 2F* | Measured | NIST | |
| 594.2619 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.7f 2F* | Measured | NIST | |
| 594.2731 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.7f 2F* | Measured | NIST | |
| 601.35 nm | N/A | B II | emission | 1s2.2s.4p 3P* → 1s2.2s.6s 3S | Measured | NIST | |
| 601.35 nm | N/A | B II | emission | 1s2.2s.4p 3P* → 1s2.2s.6s 3S | Measured | NIST | |
| 601.35 nm | N/A | B II | emission | 1s2.2s.4p 3P* → 1s2.2s.6s 3S | Measured | NIST | |
| 602.772 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.8d 2D | Measured | NIST | |
| 602.837 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.8d 2D | Measured | NIST | |
| 602.837 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.8d 2D | Measured | NIST | |
| 608.039 nm | 85 | B II | emission | 1s2.2p2 1S → 1s2.2s.3p 1P* | Measured | NIST | |
| 612.224 nm | N/A | B II | emission | 1s2.2p2 1S → 1s2.2s.3p 3P* | Measured | NIST | |
| 612.508 nm | 93 | B III | emission | 1s.2s.(3S).3s 4S → 1s.2s.(3S).3p 4P* | Measured | NIST | |
| 612.752 nm | N/A | B III | emission | 1s.2s.(3S).3s 4S → 1s.2s.(3S).3p 4P* | Measured | NIST | |
| 612.797 nm | N/A | B III | emission | 1s.2s.(3S).3s 4S → 1s.2s.(3S).3p 4P* | Measured | NIST | |
| 614.891 nm | N/A | B II | emission | 1s2.2s.4d 3D → 1s2.2s.6f 3F* | Measured | NIST | |
| 614.891 nm | N/A | B II | emission | 1s2.2s.4d 3D → 1s2.2s.6f 3F* | Measured | NIST | |
| 614.891 nm | N/A | B II | emission | 1s2.2s.4d 3D → 1s2.2s.6f 3F* | Measured | NIST | |
| 614.891 nm | N/A | B II | emission | 1s2.2s.4d 3D → 1s2.2s.6f 3F* | Measured | NIST | |
| 614.891 nm | N/A | B II | emission | 1s2.2s.4d 3D → 1s2.2s.6f 3F* | Measured | NIST | |
| 614.891 nm | N/A | B II | emission | 1s2.2s.4d 3D → 1s2.2s.6f 3F* | Measured | NIST | |
| 617.867 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.8s 2S | Measured | NIST | |
| 617.936 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.8s 2S | Measured | NIST | |
| 618.638 nm | N/A | B II | emission | 1s2.2s.4f 3F* → 1s2.2p.3p 3D | Measured | NIST | |
| 618.638 nm | N/A | B II | emission | 1s2.2s.4f 3F* → 1s2.2p.3p 3D | Measured | NIST | |
| 618.638 nm | N/A | B II | emission | 1s2.2s.4f 3F* → 1s2.2p.3p 3D | Measured | NIST | |
| 619.359 nm | N/A | B II | emission | 1s2.2s.4f 3F* → 1s2.2p.3p 3D | Measured | NIST | |
| 619.359 nm | N/A | B II | emission | 1s2.2s.4f 3F* → 1s2.2p.3p 3D | Measured | NIST | |
| 619.735 nm | N/A | B II | emission | 1s2.2s.4f 3F* → 1s2.2p.3p 3D | Measured | NIST | |
| 622.745 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.7d 2D | Measured | NIST | |
| 622.815 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.7d 2D | Measured | NIST | |
| 622.815 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.7d 2D | Measured | NIST | |
| 624.4557 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.6f 2F* | Measured | NIST | |
| 624.4557 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.6f 2F* | Measured | NIST | |
| 624.4681 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.6f 2F* | Measured | NIST | |
| 628.551 nm | 30 | B II | emission | 1s2.2s.3d 1D → 1s2.2s.4p 1P* | Measured | NIST | |
| 634.927 nm | N/A | B II | emission | 1s2.2s.4f 3F* → 1s2.2s.6g 3G | Measured | NIST | |
| 634.927 nm | N/A | B II | emission | 1s2.2s.4f 3F* → 1s2.2s.6g 3G | Measured | NIST | |
| 634.927 nm | N/A | B II | emission | 1s2.2s.4f 3F* → 1s2.2s.6g 3G | Measured | NIST | |
| 635.676 nm | 1 | B II | emission | 1s2.2s.4f 1F* → 1s2.2s.6g 1G | Measured | NIST | |
| 643.151 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.7s 2S | Measured | NIST | |
| 643.225 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.7s 2S | Measured | NIST | |
| 652.056 nm | N/A | B II | emission | 1s2.2s.4s 3S → 1s2.2p.3s 3P* | Measured | NIST | |
| 652.959 nm | N/A | B II | emission | 1s2.2s.4s 3S → 1s2.2p.3s 3P* | Measured | NIST | |
| 653.371 nm | N/A | B II | emission | 1s2.2s.4s 3S → 1s2.2p.3s 3P* | Measured | NIST | |
| 656.269 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.6d 2D | Measured | NIST | |
| 656.345 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.6d 2D | Measured | NIST | |
| 656.345 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.6d 2D | Measured | NIST | |
| 657.112 nm | 0.5 | B II | emission | 1s2.2s.5p 1P* → 1s2.2p.3p 1D | Measured | NIST | |
| 671.765 nm | 0.5 | B II | emission | 1s2.2s.4d 1D → 1s2.2s.6f 1F* | Measured | NIST | |
| 677.866 nm | N/A | B I | emission | 2s2.4p 2P* → 2s.2p2 2P | Measured | NIST | |
| 677.895 nm | N/A | B I | emission | 2s2.4p 2P* → 2s.2p2 2P | Measured | NIST | |
| 678.401 nm | N/A | B I | emission | 2s2.4p 2P* → 2s.2p2 2P | Measured | NIST | |
| 678.431 nm | N/A | B I | emission | 2s2.4p 2P* → 2s.2p2 2P | Measured | NIST | |
| 678.614 nm | 0.5 | B II | emission | 1s2.2s.4p 1P* → 1s2.2s.5d 1D | Measured | NIST | |
| 681.95167 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.5f 2F* | Measured | NIST | |
| 681.95167 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.5f 2F* | Measured | NIST | |
| 681.96637 nm | N/A | B I | emission | 2s.2p2 2D → 2s2.5f 2F* | Measured | NIST | |
| 697.688 nm | N/A | B II | emission | 1s2.2s.3s 3S → 1s2.2s.3p 1P* | Measured | NIST | |
| 703.027 nm | 4 | B II | emission | 1s2.2s.3s 3S → 1s2.2s.3p 3P* | Measured | NIST | |
| 703.203 nm | 3 | B II | emission | 1s2.2s.3s 3S → 1s2.2s.3p 3P* | Measured | NIST | |
| 703.233 nm | 2 | B II | emission | 1s2.2s.3s 3S → 1s2.2s.3p 3P* | Measured | NIST | |
| 715.955 nm | N/A | B II | emission | 1s2.2p.3s 3P* → 1s2.2p.3p 3P | Measured | NIST | |
| 716.016 nm | N/A | B II | emission | 1s2.2p.3s 3P* → 1s2.2p.3p 3P | Measured | NIST | |
| 716.511 nm | N/A | B II | emission | 1s2.2p.3s 3P* → 1s2.2p.3p 3P | Measured | NIST | |
| 716.846 nm | N/A | B II | emission | 1s2.2p.3s 3P* → 1s2.2p.3p 3P | Measured | NIST | |
| 717.045 nm | N/A | B II | emission | 1s2.2p.3s 3P* → 1s2.2p.3p 3P | Measured | NIST | |
| 717.602 nm | N/A | B II | emission | 1s2.2p.3s 3P* → 1s2.2p.3p 3P | Measured | NIST | |
| 720.593 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.5d 2D | Measured | NIST | |
| 720.685 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.5d 2D | Measured | NIST | |
| 720.685 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.5d 2D | Measured | NIST | |
| 720.766 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.6s 2S | Measured | NIST | |
| 720.859 nm | N/A | B I | emission | 2s2.3p 2P* → 2s2.6s 2S | Measured | NIST | |
| 722.85 nm | N/A | B II | emission | 1s2.2s.4s 1S → 1s2.2s.5p 1P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 2350.15 K |
| Boiling point | 4273.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (3)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.3205 |
| 2 | p | 2.5786 |
| 2 | s | 2.4238 |
Crystal Radii Detail (3)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | III | 15 | ||
| 3 | IV | 25 | ||
| 3 | VI | 41 | calculated, |
Isotope Decay Modes (30)
| Isotope | Mode | Intensity |
|---|---|---|
| 6 | 2p | — |
| 7 | p | 100% |
| 8 | B+ | 100% |
| 8 | B+A | 100% |
| 9 | p | 100% |
| 12 | B- | 100% |
| 12 | B-A | 0.6% |
| 13 | B- | 100% |
| 13 | B-n | 0.3% |
| 14 | B- | 100% |
X‑ray Scattering Factors (502)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.48933 |
| 10.1617 | — | 1.48084 |
| 10.3261 | — | 1.4724 |
| 10.4931 | — | 1.46401 |
| 10.6628 | — | 1.45567 |
| 10.8353 | — | 1.44738 |
| 11.0106 | — | 1.43913 |
| 11.1886 | — | 1.43093 |
| 11.3696 | — | 1.42278 |
| 11.5535 | — | 1.41467 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.0×101 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
4.44 milligrams per liter
References (1)
Sources
Sources of this element.
The element is not found free in nature, but occurs as orthoboric acid usually found in certain volcanic spring waters and as borates in boron and colemantie.
Important sources of boron are ore rasorite (kernite) and tincal (borax ore). Both of these ores are found in the Mojave Desert. Tincal is the most important source of boron from the Mojave. Extensive borax deposits are also found in Turkey.
Boron exists naturally as 19.78% 10B isotope and 80.22% 11B isotope. High-purity crystalline boron may be prepared by the vapor phase reduction of boron trichloride or tribromide with hydrogen on electrically heated filaments. The impure or amorphous, boron, a brownish-black powder, can be obtained by heating the trioxide with magnesium powder.
Boron of 99.9999% purity has been produced and is available commercially. Elemental boron has an energy band gap of 1.50 to 1.56 eV, which is higher than that of either silicon or germanium.
References (1)
- [6] Boron https://periodic.lanl.gov/5.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Boron.
The element property data was retrieved from publications.