Zinc (Zn)
transition-metalSolid
Standard Atomic Weight
65.38 uElectron configuration
[Ar] 4s2 3d10Melting point
419.53 °C (692.68 K)Boiling point
906.85 °C (1180 K)Density
7134 kg/m³Oxidation states
−2, 0, +1, +2Electronegativity (Pauling)
1.65Ionization energy (1st)
Discovery year
1746Atomic radius
135 pmDetails
Zinc is a moderately reactive, bluish-white transition metal with a filled 3d shell and chemistry dominated by the +2 oxidation state. It is an essential trace element for living organisms and an important industrial metal, especially for corrosion protection of steel. In minerals it occurs chiefly as sulfide and carbonate ores, and in technology it is valued for sacrificial galvanic behavior, alloy formation, and stable, often colorless Zn²⁺ compounds.
Zinc is a bluish-white, lustrous metal. It is brittle at ordinary temperatures but malleable at 100 to 150°C. It is a fair conductor of electricity, and burns in air at high red heat with evolution of white clouds of the oxide.
It exhibits superplasticity. Neither zinc nor zirconium is ferromagnetic; but ZrZn2 exhibits ferromagnetism at temperatures below 35°K. It has unusual electrical, thermal, optical, and solid-state properties that have not been fully investigated.
The name derives from the German zink of unknown origin. It was first used in prehistoric times, where its compounds were used for healing wounds and sore eyes and for making brass. Zinc was recognized as a metal as early as 1374.
Although zinc compounds have been used for at least 2,500 years in the production of brass, zinc wasn't recognized as a distinct element until much later. Metallic zinc was first produced in India sometime in the 1400s by heating the mineral calamine (ZnCO3) with wool. Zinc was rediscovered by Andreas Sigismund Marggraf in 1746 by heating calamine with charcoal. Today, most zinc is produced through the electrolysis of aqueous zinc sulfate (ZnSO4).
From the German word Zink, of obscure origin. Centuries before zinc was recognized as a distinct element, zinc ores were used for making brass. An alloy containing 87 percent zinc has been found in prehistoric ruins in Transylvania.
Metallic zinc was produced in the 13th century A.D. India by reducing calamine with organic substances such as wool. The metal was rediscovered in Europe by Marggraf in 1746. He demonstrated that zinc could be obtained by reducing calamine with charcoal.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
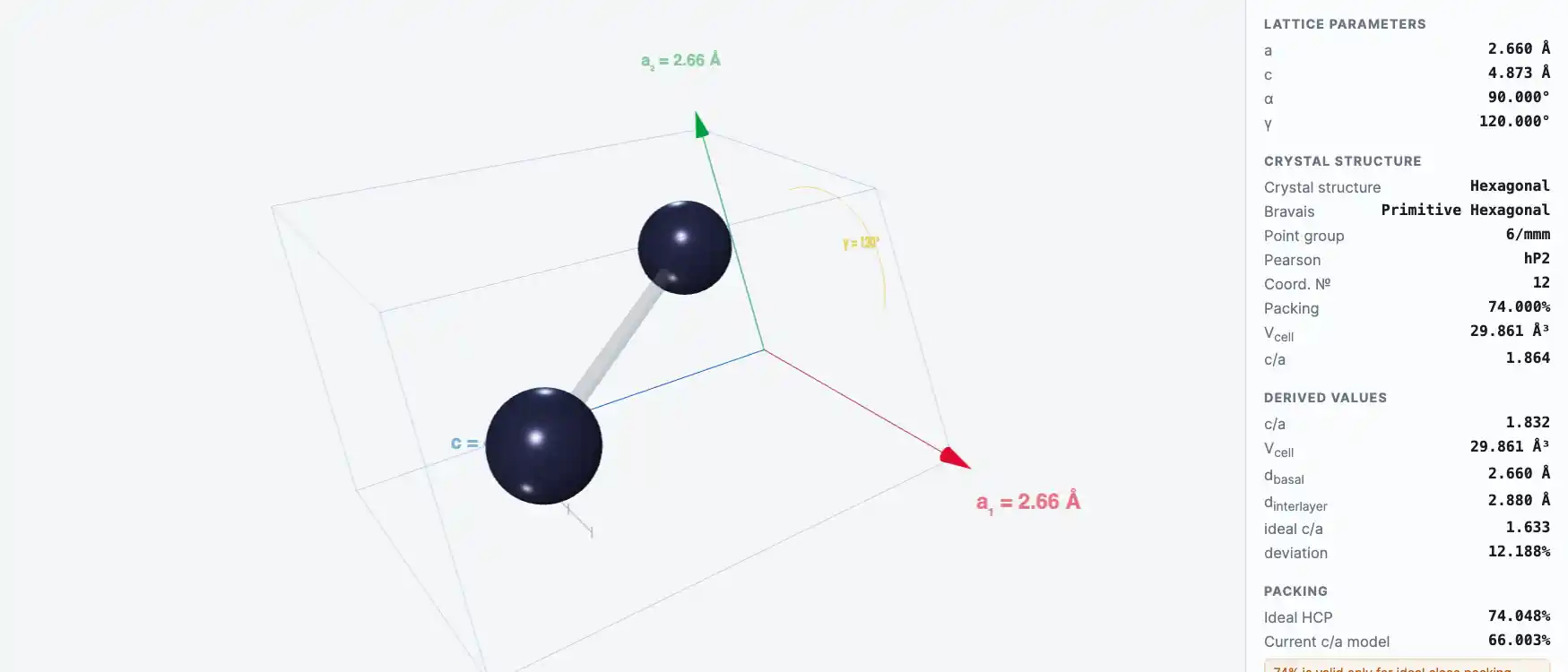
Crystal Structure
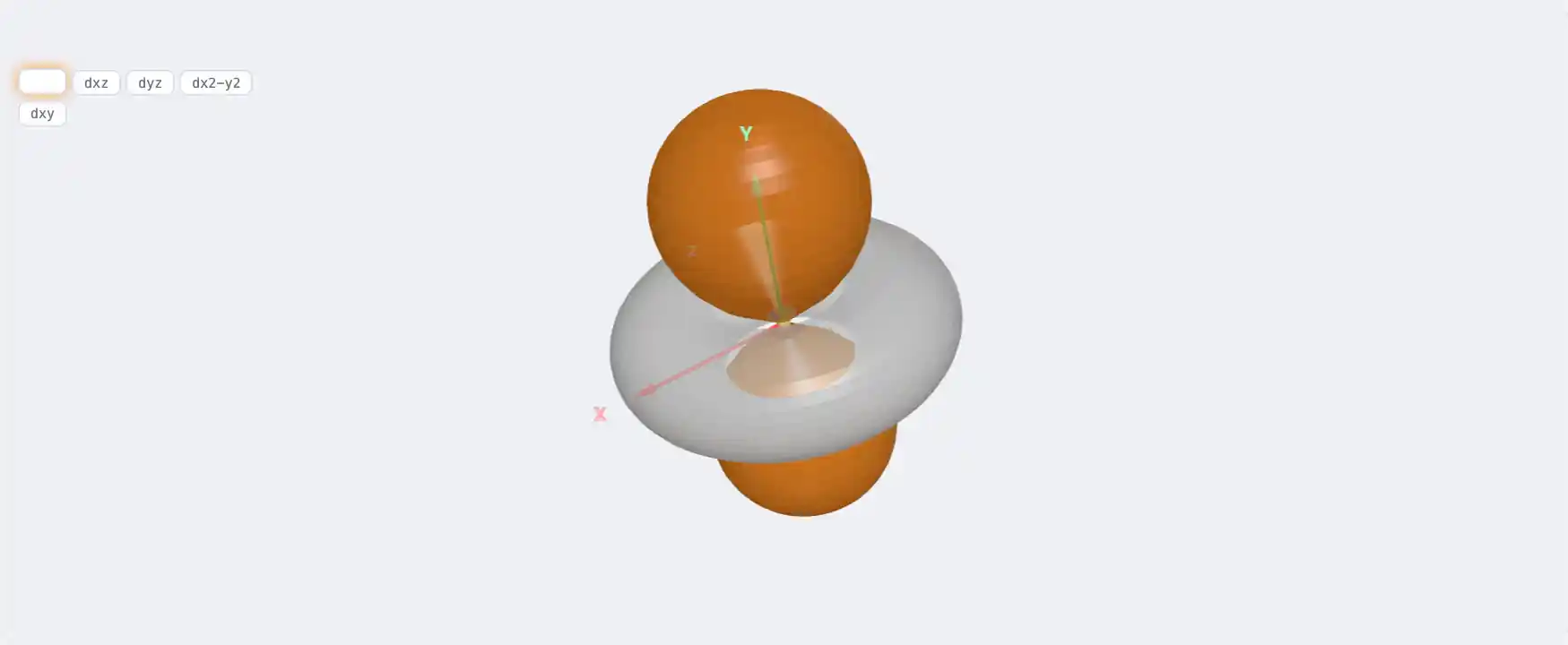
Electronic Structure
Identifiers
Electron Configuration Measured
Zn: 3d¹⁰ 4s²[Ar] 3d¹⁰ 4s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 66 Stable | 65.92603381 ± 0.00000094 | 27.7300% | Stable |
| 67 Stable | 66.92712775 ± 0.00000096 | 4.0400% | Stable |
| 68 Stable | 67.92484455 ± 0.00000098 | 18.4500% | Stable |
Phase / State
Reason: 394.5 °C below melting point (419.53 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 30 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Zn I | 0 | 570 | 16 | 564 |
| Zn II | +1 | 96 | 22 | 90 |
| Zn III | +2 | 39 | 0 | 0 |
| Zn IV | +3 | 119 | 0 | 0 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Zn I | 0 | 380 |
| Zn II | +1 | 94 |
| Zn III | +2 | 316 |
| Zn IV | +3 | 245 |
| Zn V | +4 | 158 |
| Zn VI | +5 | 193 |
| Zn VII | +6 | 134 |
| Zn VIII | +7 | 5 |
| Zn IX | +8 | 2 |
| Zn X | +9 | 2 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 4 | N/A | 60 pm |
| +2 | 5 | N/A | 68 pm |
| +2 | 6 | N/A | 74 pm |
| +2 | 8 | N/A | 90 pm |
Compounds
Isotopes (3)
Naturally occurring zinc contains five stable isotopes. Sixteen other unstable isotopes are recognized.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 66 Stable | 65.92603381 ± 0.00000094 | 27.7300% ± 0.9800% | Stable | stable | |
| 67 Stable | 66.92712775 ± 0.00000096 | 4.0400% ± 0.1600% | Stable | stable | |
| 68 Stable | 67.92484455 ± 0.00000098 | 18.4500% ± 0.6300% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 387.9141 nm | N/A | Zn I | emission | 3d10.4s.4p 1P* → 3d10.4s.7d 1D | Measured | NIST | |
| 396.543 nm | 78000 | Zn I | emission | 3d10.4s.4p 1P* → 3d10.4s.8s 1S | Measured | NIST | |
| 411.31114 nm | 81000 | Zn I | emission | 3d10.4s.4p 1P* → 3d10.4s.6d 1D | Measured | NIST | |
| 429.2883 nm | 32000 | Zn I | emission | 3d10.4s.4p 3P* → 3d10.4s.5s 1S | Measured | NIST | |
| 429.8325 nm | 49000 | Zn I | emission | 3d10.4s.4p 1P* → 3d10.4s.7s 1S | Measured | NIST | |
| 455.326 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.30p 1P* | Measured | NIST | |
| 455.548 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.29p 1P* | Measured | NIST | |
| 455.795 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.28p 1P* | Measured | NIST | |
| 456.073 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.27p 1P* | Measured | NIST | |
| 456.388 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.26p 1P* | Measured | NIST | |
| 456.745 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.25p 1P* | Measured | NIST | |
| 457.155 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.24p 1P* | Measured | NIST | |
| 457.623 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.23p 1P* | Measured | NIST | |
| 458.167 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.22p 1P* | Measured | NIST | |
| 458.796 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.21p 1P* | Measured | NIST | |
| 459.541 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.20p 1P* | Measured | NIST | |
| 460.423 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.19p 1P* | Measured | NIST | |
| 461.482 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.18p 1P* | Measured | NIST | |
| 462.768 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.17p 1P* | Measured | NIST | |
| 462.980809 nm | 390000 | Zn I | emission | 3d10.4s.4p 1P* → 3d10.4s.5d 1D | Measured | NIST | |
| 464.351 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.16p 1P* | Measured | NIST | |
| 466.559 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.15p 3P* | Measured | NIST | |
| 468.013589 nm | 540000 | Zn I | emission | 3d10.4s.4p 3P* → 3d10.4s.5s 3S | Measured | NIST | |
| 469.143 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.14p 3P* | Measured | NIST | |
| 472.215691 nm | 1000000 | Zn I | emission | 3d10.4s.4p 3P* → 3d10.4s.5s 3S | Measured | NIST | |
| 472.527 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.13p 3P* | Measured | NIST | |
| 477.071 nm | N/A | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.12p 3P* | Measured | NIST | |
| 481.053206 nm | 1100000 | Zn I | emission | 3d10.4s.4p 3P* → 3d10.4s.5s 3S | Measured | NIST | |
| 506.866 nm | 77000 | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.9p 3P* | Measured | NIST | |
| 506.943 nm | 21000 | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.9p 3P* | Measured | NIST | |
| 506.998 nm | 3300 | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.9p 3P* | Measured | NIST | |
| 518.19819 nm | 120000 | Zn I | emission | 3d10.4s.4p 1P* → 3d10.4s.6s 1S | Measured | NIST | |
| 530.866 nm | 380000 | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.8p 3P* | Measured | NIST | |
| 531.017 nm | 160000 | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.8p 3P* | Measured | NIST | |
| 531.101 nm | 56000 | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.8p 3P* | Measured | NIST | |
| 577.205 nm | 490000 | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.7p 3P* | Measured | NIST | |
| 577.5452 nm | 210000 | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.7p 3P* | Measured | NIST | |
| 577.7033 nm | 85000 | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.7p 3P* | Measured | NIST | |
| 623.78967 nm | 93000 | Zn I | emission | 3d10.4s.4p 1P* → 3d10.4s.4d 3D | Measured | NIST | |
| 623.9169 nm | 38000 | Zn I | emission | 3d10.4s.4p 1P* → 3d10.4s.4d 3D | Measured | NIST | |
| 636.23458 nm | 240000 | Zn I | emission | 3d10.4s.4p 1P* → 3d10.4s.4d 1D | Measured | NIST | |
| 647.9184 nm | 55000 | Zn I | emission | 3d10.4s.5s 1S → 3d10.4s.7p 1P* | Measured | NIST | |
| 692.8295 nm | 40000 | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.6p 3P* | Measured | NIST | |
| 693.8449 nm | 20000 | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.6p 3P* | Measured | NIST | |
| 694.3184 nm | 7000 | Zn I | emission | 3d10.4s.5s 3S → 3d10.4s.6p 3P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 692.68 K |
| Boiling point | 1180.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (7)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.6755 |
| 2 | p | 3.902 |
| 2 | s | 8.172 |
| 3 | d | 16.1217 |
| 3 | p | 14.6307 |
| 3 | s | 13.7808 |
| 4 | s | 24.0348 |
Crystal Radii Detail (4)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | IV | 74 | ||
| 2 | V | 82 | ||
| 2 | VI | 88 | from r^3 vs V plots, | |
| 2 | VIII | 104 | calculated, |
Isotope Decay Modes (49)
| Isotope | Mode | Intensity |
|---|---|---|
| 54 | 2p | 87% |
| 55 | B+ | 100% |
| 55 | B+p | 91% |
| 56 | B+ | 100% |
| 56 | B+p | 88% |
| 57 | B+ | 100% |
| 57 | B+p | 87% |
| 58 | B+ | 100% |
| 58 | B+p | 0.7% |
| 59 | B+ | 100% |
X‑ray Scattering Factors (504)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 2.21675 |
| 10.1617 | — | 2.11915 |
| 10.3261 | — | 2.02585 |
| 10.4931 | — | 1.93665 |
| 10.6628 | — | 1.85138 |
| 10.8353 | — | 1.76986 |
| 11.0106 | — | 1.69194 |
| 11.1886 | — | 1.63293 |
| 11.3696 | — | 1.57784 |
| 11.5535 | — | 1.5246 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
7.0×101 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
4.9×10-3 milligrams per liter
References (1)
Sources
Sources of this element.
The principal ores of zinc are sphalerite (sulfide), smithsonite (carbonate), calamine (silicate), and franklinite (zinc, manganese, iron oxide). One method of zinc extraction involves roasting its ores to form the oxide and reducing the oxide with coal or carbon, with subsequent distillation of the metal.
References (1)
- [6] Zinc https://periodic.lanl.gov/30.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Zinc.
The element property data was retrieved from publications.