Cobalt (Co)
transition-metalSolid
Standard Atomic Weight
58.933194 uElectron configuration
[Ar] 4s2 3d7Melting point
1494.85 °C (1768 K)Boiling point
2926.85 °C (3200 K)Density
8860 kg/m³Oxidation states
−3, −1, 0, +1, +2, +3, +4, +5Electronegativity (Pauling)
1.88Ionization energy (1st)
Discovery year
1735Atomic radius
135 pmDetails
Cobalt is a hard transition metal of group 9, best known for stable high-temperature alloys, magnetic materials, rechargeable battery cathodes, and intensely colored blue pigments. It occurs in nature chiefly as a minor constituent of sulfide and arsenide minerals and is commonly recovered with copper or nickel. Chemically it is dominated by the +2 and +3 oxidation states, with coordination chemistry that includes biologically important corrinoids.
Cobalt is a brittle, hard metal, resembling iron and nickel in appearance. It has a metallic permeability of about two thirds that of iron. Cobalt tends to exist as a mixture of two allotropes over a wide temperature range. The transformation is sluggish and accounts in part for the wide variation in reported data on physical properties of cobalt.
The name derives from the German Kobold for "evil spirits" or "goblins", who were superstitiously thought to cause trouble for miners because the mineral contained arsenic that injured their health and the metallic ores did not yield metals when treated with the normal methods. Cobalt was discovered in 1735 by the Swedish chemist Georg Brandt.
Cobalt was discovered by Georg Brandt, a Swedish chemist, in 1739. Brandt was attempting to prove that the ability of certain minerals to color glass blue was due to an unknown element and not to bismuth, as was commonly believed at the time. Cobalt's primary ores are cobaltite (CoAsS) and erythrite (Co3(AsO4)2). Cobalt is usually recovered as a byproduct of mining and refining nickel, silver, lead, copper and iron.
From the German word Kobald, goblin or evil spirit; also from the Greek cobalos, mine. George Brandt discovered cobalt in 1735.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
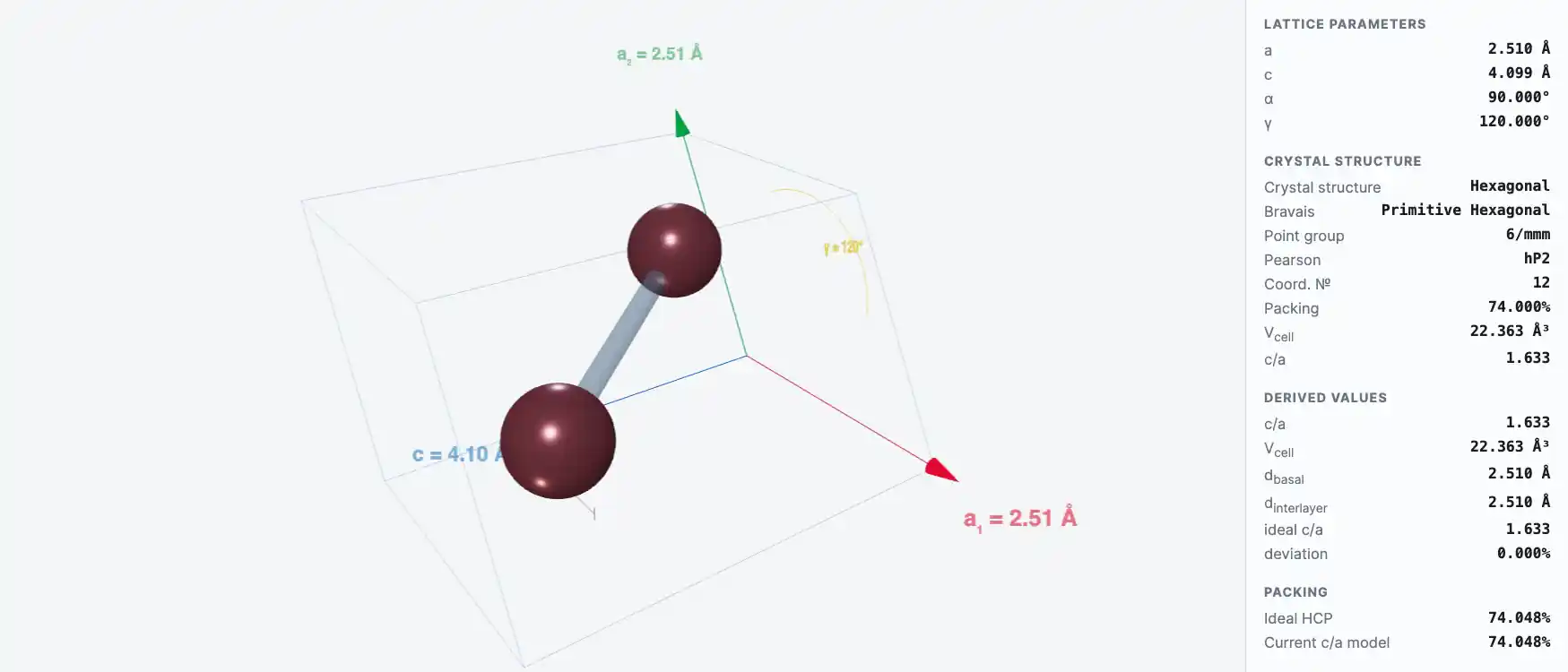
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Co: 3d⁷ 4s²[Ar] 3d⁷ 4s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁷ 4s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 59 Stable | 58.93319429 ± 0.00000056 | 100.0000% | Stable |
Phase / State
Reason: 1469.8 °C below melting point (1494.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 27 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Co I | 0 | 420 | 338 | 338 |
| Co II | +1 | 3168 | 2761 | 3168 |
| Co III | +2 | 2064 | 1957 | 2064 |
| Co IV | +3 | 9 | 0 | 0 |
| Co V | +4 | 55 | 0 | 0 |
| Co VIII | +7 | 165 | 150 | 165 |
| Co IX | +8 | 48 | 19 | 48 |
| Co X | +9 | 22 | 5 | 22 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Co I | 0 | 330 |
| Co II | +1 | 482 |
| Co III | +2 | 288 |
| Co IV | +3 | 297 |
| Co V | +4 | 268 |
| Co VI | +5 | 180 |
| Co VII | +6 | 65 |
| Co VIII | +7 | 73 |
| Co IX | +8 | 40 |
| Co X | +9 | 31 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +2 | 4 | high | 57.99999999999999 pm |
| +2 | 5 | N/A | 67 pm |
| +2 | 6 | low | 65 pm |
| +2 | 6 | high | 74.5 pm |
| +2 | 8 | N/A | 90 pm |
| +3 | 6 | low | 54.50000000000001 pm |
| +3 | 6 | high | 61 pm |
| +4 | 4 | N/A | 40 pm |
| +4 | 6 | high | 53 pm |
Compounds
Isotopes (1)
Cobalt-60, an artificial isotope, is an important gamma ray source, and is extensively used as a tracer and a radiotherapeutic agent.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 59 Stable | 58.93319429 ± 0.00000056 | 100.0000% | Stable | stable |
Spectral Lines
Showing 50 of 738 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 389.4073 nm | 7900 | Co I | emission | 3p6.3d8.(3F).4s a 2F → 3p6.3d8.(3F).4p y 2G* | Measured | NIST | |
| 384.5461 nm | 6900 | Co I | emission | 3p6.3d8.(3F).4s a 2F → 3p6.3d8.(3F).4p y 2G* | Measured | NIST | |
| 399.5302 nm | 6000 | Co I | emission | 3p6.3d8.(3F).4s a 2F → 3p6.3d8.(3F).4p y 4G* | Measured | NIST | |
| 512.92021 nm | 5800 | Co II | emission | 3d7.(4F).4d e 5H → 3d7.(4F<9/2>).4f 2[15/2]* | Measured | NIST | |
| 387.3114 nm | 5500 | Co I | emission | 3p6.3d8.(3F).4s b 4F → 3p6.3d7.(4F).4s.4p.(3P*) z 4D* | Measured | NIST | |
| 412.1311 nm | 4400 | Co I | emission | 3p6.3d8.(3F).4s a 2F → 3p6.3d7.(4F).4s.4p.(3P*) z 2G* | Measured | NIST | |
| 516.315 nm | 4100 | Co II | emission | 3d7.(4F).4d e 3H → 3d7.(4F<7/2>).4f 2[13/2]* | Measured | NIST | |
| 521.43464 nm | 3900 | Co II | emission | 3d7.(4F).4d e 5H → 3d7.(4F<9/2>).4f 2[15/2]* | Measured | NIST | |
| 505.07089 nm | 3800 | Co II | emission | 3d7.(4F).4d e 5G → 3d7.(4F<9/2>).4f 2[13/2]* | Measured | NIST | |
| 517.06829 nm | 3200 | Co II | emission | 3d7.(4F).4d e 5H → 3d7.(4F<7/2>).4f 2[13/2]* | Measured | NIST | |
| 387.3955 nm | 2800 | Co I | emission | 3p6.3d8.(3F).4s b 4F → 3p6.3d7.(4F).4s.4p.(3P*) z 4D* | Measured | NIST | |
| 411.8767 nm | 2800 | Co I | emission | 3p6.3d8.(3F).4s a 2F → 3p6.3d7.(4F).4s.4p.(3P*) z 2G* | Measured | NIST | |
| 519.95128 nm | 2800 | Co II | emission | 3d7.(4F).4d e 3H → 3d7.(4F<5/2>).4f 2[11/2]* | Measured | NIST | |
| 513.56812 nm | 2700 | Co II | emission | 3d7.(4F).4d e 5H → 3d7.(4F<5/2>).4f 2[11/2]* | Measured | NIST | |
| 509.92115 nm | 2500 | Co II | emission | 3d7.(4F).4d e 3G → 3d7.(4F<7/2>).4f 2[11/2]* | Measured | NIST | |
| 523.11044 nm | 2300 | Co II | emission | 3d7.(4F).4d e 3H → 3d7.(4F<3/2>).4f 2[9/2]* | Measured | NIST | |
| 496.41682 nm | 2200 | Co II | emission | 3d7.(4F).4d f 5F → 3d7.(4F<9/2>).4f 2[11/2]* | Measured | NIST | |
| 505.7416 nm | 1700 | Co II | emission | 3d7.(4F).4d e 5G → 3d7.(4F<9/2>).4f 2[11/2]* | Measured | NIST | |
| 393.5959 nm | 1500 | Co I | emission | 3p6.3d8.(3F).4s a 2F → 3p6.3d8.(3F).4p y 4F* | Measured | NIST | |
| 384.2046 nm | 1400 | Co I | emission | 3p6.3d8.(3F).4s a 2F → 3p6.3d7.(4F).4s.4p.(3P*) z 2D* | Measured | NIST | |
| 509.52694 nm | 1400 | Co II | emission | 3d7.(4F).4d e 3G → 3d7.(4F<5/2>).4f 2[9/2]* | Measured | NIST | |
| 510.75362 nm | 1400 | Co II | emission | 3d7.(4F).4d e 5G → 3d7.(4F<7/2>).4f 2[9/2]* | Measured | NIST | |
| 506.70997 nm | 1200 | Co II | emission | 3d7.(4F).4d e 5P → 3d7.(4F<9/2>).4f 2[3/2]* | Measured | NIST | |
| 396.31 nm | 1100 | Co II | emission | 3d7.(4F).5p 5F* → 3d7.(4F).6d 5G | Measured | NIST | |
| 496.23566 nm | 1100 | Co II | emission | 3d7.(4F).4d f 5F → 3d7.(4F<9/2>).4f 2[9/2]* | Measured | NIST | |
| 517.6949 nm | 1100 | Co II | emission | 3d7.(4F).4d e 5G → 3d7.(4F<9/2>).4f 2[13/2]* | Measured | NIST | |
| 657.13038 nm | 1100 | Co II | emission | 3d7.(4F).5p 5G* → 3d7.(4F).5d 5H | Measured | NIST | |
| 657.62238 nm | 1100 | Co II | emission | 3d7.(4F).5p 3G* → 3d7.(4F).5d 3H | Measured | NIST | |
| 502.59107 nm | 990 | Co II | emission | 3d7.(4F).4d e 5D → 3d7.(4F<7/2>).4f 2[11/2]* | Measured | NIST | |
| 399.79 nm | 970 | Co I | emission | 3p6.3d8.(3F).4s a 2F → 3p6.3d8.(3F).4p * | Measured | NIST | |
| 512.9972 nm | 960 | Co II | emission | 3d7.(4F).4d e 3G → 3d7.(4F<3/2>).4f 2[9/2]* | Measured | NIST | |
| 743.9418 nm | 960 | Co II | emission | 3d7.(4F).5p 5D* → 3d7.(4F).6s 5F | Measured | NIST | |
| 637.37856 nm | 920 | Co II | emission | 3d7.(4F).5p 5F* → 3d7.(4F).5d 5G | Measured | NIST | |
| 495.82966 nm | 900 | Co II | emission | 3d7.(4F).4d f 5F → 3d7.(4F<9/2>).4f 2[11/2]* | Measured | NIST | |
| 502.25161 nm | 900 | Co II | emission | 3d7.(4F).4d e 3D → 3d7.(4F<3/2>).4f 2[7/2]* | Measured | NIST | |
| 642.58717 nm | 900 | Co II | emission | 3d7.(4F).5p 5F* → 3d7.(4F).5d 5F | Measured | NIST | |
| 510.45696 nm | 890 | Co II | emission | 3d7.(4F).4d e 5D → 3d7.(4F<5/2>).4f 2[9/2]* | Measured | NIST | |
| 502.36685 nm | 860 | Co II | emission | 3d7.(4F).4d f 5F → 3d7.(4F<9/2>).4f 2[9/2]* | Measured | NIST | |
| 620.5716 nm | 860 | Co II | emission | 3d7.(4F).5p 5D* → 3d7.(4F).5d 5F | Measured | NIST | |
| 409.2384 nm | 830 | Co I | emission | 3p6.3d8.(3F).4s a 2F → 3p6.3d7.(4F).4s.4p.(3P*) z 2F* | Measured | NIST | |
| 508.31892 nm | 780 | Co II | emission | 3d7.(4F).4d e 5D → 3d7.(4F<3/2>).4f 2[5/2]* | Measured | NIST | |
| 502.6664 nm | 680 | Co II | emission | 3d7.(4F).4d e 5G → 3d7.(4F<5/2>).4f 2[7/2]* | Measured | NIST | |
| 517.75201 nm | 680 | Co II | emission | 3d7.(4F).4d e 3H → 3d7.(4F<7/2>).4f 2[11/2]* | Measured | NIST | |
| 662.16287 nm | 680 | Co II | emission | 3d7.(4F).5p 3G* → 3d7.(4F).5d 3H | Measured | NIST | |
| 495.25711 nm | 670 | Co II | emission | 3d7.(4F).4d e 5G → 3d7.(4F<5/2>).4f 2[9/2]* | Measured | NIST | |
| 500.77152 nm | 640 | Co II | emission | 3d7.(4F).4d f 5F → 3d7.(4F<9/2>).4f 2[5/2]* | Measured | NIST | |
| 510.63945 nm | 640 | Co II | emission | 3d7.(4F).4d e 5D → 3d7.(4F<3/2>).4f 2[5/2]* | Measured | NIST | |
| 499.59719 nm | 600 | Co II | emission | 3d7.(4F).4d e 5H → 3d7.(4F<7/2>).4f 2[13/2]* | Measured | NIST | |
| 509.2051 nm | 600 | Co II | emission | 3d7.(4F).4d e 5D → 3d7.(4F<3/2>).4f 2[3/2]* | Measured | NIST | |
| 510.07741 nm | 600 | Co II | emission | 3d7.(4F).4d e 3G → 3d7.(4F<7/2>).4f 2[9/2]* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1768.15 K |
| Boiling point | 3200.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (7)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.6332 |
| 2 | p | 3.9076 |
| 2 | s | 7.595 |
| 3 | d | 15.1446 |
| 3 | p | 13.5654 |
| 3 | s | 12.6777 |
| 4 | s | 21.4236 |
Crystal Radii Detail (9)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 2 | IV | HS | 72 | |
| 2 | V | 81 | calculated, | |
| 2 | VI | LS | 79 | from r^3 vs V plots, |
| 2 | VI | HS | 88.5 | from r^3 vs V plots, |
| 2 | VIII | 104 | ||
| 3 | VI | LS | 68.5 | from r^3 vs V plots, |
| 3 | VI | HS | 75 | |
| 4 | IV | 54 | ||
| 4 | VI | HS | 67 | from r^3 vs V plots, |
Isotope Decay Modes (57)
| Isotope | Mode | Intensity |
|---|---|---|
| 47 | p | — |
| 48 | p | — |
| 49 | p | — |
| 50 | B+ | 100% |
| 50 | B+p | 70.5% |
| 50 | 2p | — |
| 51 | B+ | 100% |
| 51 | B+p | 3.8% |
| 52 | B+ | 100% |
| 52 | B+p | — |
X‑ray Scattering Factors (504)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.42071 |
| 10.1617 | — | 1.45925 |
| 10.3261 | — | 1.49884 |
| 10.4931 | — | 1.53949 |
| 10.6628 | — | 1.58125 |
| 10.8353 | — | 1.62415 |
| 11.0106 | — | 1.6682 |
| 11.1886 | — | 1.71345 |
| 11.3696 | — | 1.75993 |
| 11.5535 | — | 1.80767 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
2.5×101 milligrams per kilogram
References (1)
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
2×10-5 milligrams per liter
References (1)
Sources
Sources of this element.
Cobalt occurs in the minerals cobaltite, smaltite, and erythrite, and is often associated with nickel, silver, lead, copper, and iron ores, from which it is most frequently obtained as a by-product. It is also present in meteorites.
Important ore deposits are found in Zaire, Morocco, and Canada. The U.S. Geological Survey has announced that the bottom of the north central Pacific Ocean may have cobalt-rich deposits at relatively shallow depths in water close to the the Hawaiian Islands and other U.S. Pacific territories.
References (1)
- [6] Cobalt https://periodic.lanl.gov/27.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Cobalt.
The element property data was retrieved from publications.