Scandium (Sc)
transition-metalSolid
Standard Atomic Weight
44.955908 uElectron configuration
[Ar] 4s2 3d1Melting point
1540.85 °C (1814 K)Boiling point
2835.85 °C (3109 K)Density
2990 kg/m³Oxidation states
0, +1, +2, +3Electronegativity (Pauling)
1.36Ionization energy (1st)
Discovery year
1879Atomic radius
160 pmDetails
Scandium is a light transition metal with chemistry dominated by the +3 oxidation state. It is chemically similar to yttrium and the lanthanides, but its small ionic radius gives some distinct coordination behavior. The element is widely dispersed in minerals and rarely occurs in rich, easily worked ores. Its technological importance is concentrated in specialty aluminum alloys, high-intensity lighting, and research materials rather than large-volume metal use.
Scandium is a silver-white metal which develops a slightly yellowish or pinkish cast upon exposure to air. A relatively soft element, scandium resembles yttrium and the rare-earth metals more than it resembles aluminum or titanium.
It is a very light metal and has a much higher melting point than aluminum, making it of interest to designers of spacecraft. Scandium is not attacked by a 1:1 mixture of HNO3 and 48% HF.
Chemically it is one of the alkaline earth elements; it readily forms a white coating of nitride in air, reacts with water, burns with a yellow-red flame.
The name derives from the Latin scandia for Scandinavia, where the mineral was found. It was discovered by the Swedish chemist Lars-Fredrik Nilson in 1879 in an ytterbium sample. In the same year, the Swedish chemist Per Theodore Cleve proved that scandium was Mendeleev's predicted "eka-boron".
Scandium was discovered by Lars Fredrik Nilson, a Swedish chemist, in 1879 while attempting to produce a sample of pure ytterbia from 10 kilograms of the mineral euxenite ((Y, Ca, Er, La, Ce, U, Th)(Nb, Ta, Ti)2O6). Scandium can be obtained from the minerals thortveitite ((Sc, Y)2Si2O7), bazzite (Be3(Sc, Al)2Si6O18) and wiikite, but is usually obtained as a byproduct of refining uranium. Metallic scandium was first produced in 1937 and the first pound (0.45 kilograms) of pure scandium was produced in 1960. Scandium is a soft, light metal that might have applications in the aerospace industry. With a cost of $270 per gram ($122,500 per pound), scandium is too expensive for widespread use.
From the Latin word Scandia, Scandinavia. On the basis of the Periodic System, Mendeleev predicted the existence of ekaboron, which would have an atomic weight between 40 of calcium and 48 of titanium. The element was discovered by Nilson in 1878 in the minerals euxenite and gadolinite, which had not yet been found anywhere except in Scandinavia. By processing 10 kg of euxenite and other residues of rare-earth minerals, Nilson was able to prepare about 2g of highly pure scandium oxide. Later scientists pointed out that Nilson's scandium was identical with Mendeleev's ekaboron.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
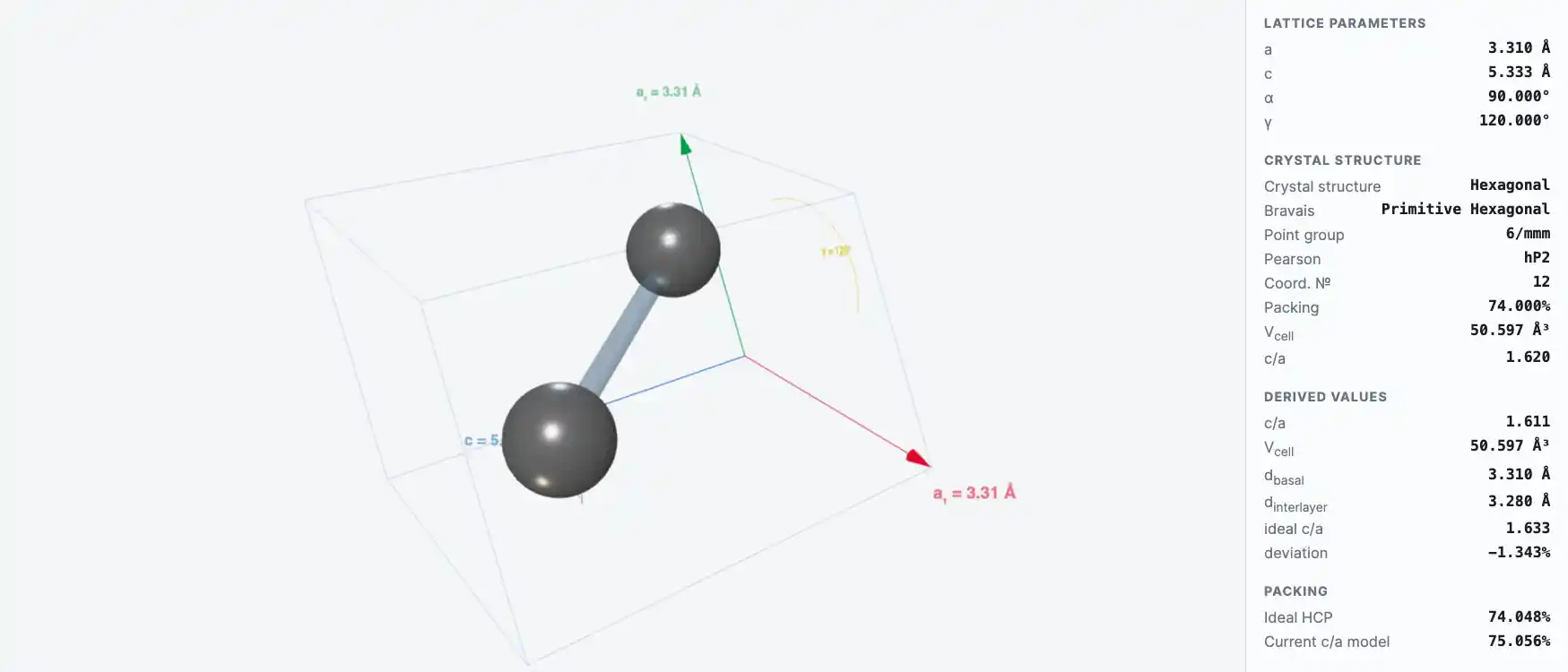
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
Sc: 3d¹ 4s²[Ar] 3d¹ 4s²1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹ 4s²Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 45 Stable | 44.95590828 ± 0.00000077 | 100.0000% | Stable |
Phase / State
Reason: 1515.8 °C below melting point (1540.85 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Atomic Spectra
Showing 10 of 21 Atomic Spectra. Sorted by ion charge (ascending).
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| Sc I | 0 | 2198 | 260 | 1682 |
| Sc II | +1 | 829 | 139 | 829 |
| Sc III | +2 | 133 | 97 | 133 |
| Sc IV | +3 | 408 | 4 | 408 |
| Sc V | +4 | 456 | 16 | 456 |
| Sc VI | +5 | 79 | 12 | 75 |
| Sc VII | +6 | 70 | 37 | 70 |
| Sc VIII | +7 | 75 | 48 | 75 |
| Sc IX | +8 | 42 | 22 | 42 |
| Sc X | +9 | 99 | 29 | 99 |
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| Sc I | 0 | 478 |
| Sc II | +1 | 169 |
| Sc III | +2 | 44 |
| Sc IV | +3 | 129 |
| Sc V | +4 | 119 |
| Sc VI | +5 | 40 |
| Sc VII | +6 | 35 |
| Sc VIII | +7 | 27 |
| Sc IX | +8 | 27 |
| Sc X | +9 | 68 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +3 | 6 | N/A | 74.5 pm |
| +3 | 8 | N/A | 87 pm |
Compounds
Isotopes (1)
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 45 Stable | 44.95590828 ± 0.00000077 | 100.0000% | Stable | stable |
Spectral Lines
Showing 50 of 946 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 683.5026 nm | 640 | Sc I | emission | 3d2.(3P).4s 2P → 3d2.(3P).4p 2S* | Measured | NIST | |
| 681.9491 nm | 485 | Sc I | emission | 3d.4s.(1D).4p 2F* → 3d.4s.(3D).5s 2D | Measured | NIST | |
| 673.7872 nm | 465 | Sc I | emission | 3d.4s.(3D).4p 2F* → 3d.4s.(3D).4d 2G | Measured | NIST | |
| 673.945 nm | 360 | Sc I | emission | 3d.4s.(3D).4p 2F* → 3d.4s.(3D).4d 2G | Measured | NIST | |
| 681.7117 nm | 345 | Sc I | emission | 3d2.(3P).4s 2P → 3d2.(3P).4p 2S* | Measured | NIST | |
| 682.9509 nm | 335 | Sc I | emission | 3d.4s.(1D).4p 2F* → 3d.4s.(3D).5s 2D | Measured | NIST | |
| 406.8661 nm | 100 | Sc III | emission | 3p6.4d 2D → 3p6.4f 2F* | Measured | NIST | |
| 744.9141 nm | 90 | Sc III | emission | 3p6.5s 2S → 3p6.5p 2P* | Measured | NIST | |
| 406.121 nm | 80 | Sc III | emission | 3p6.4d 2D → 3p6.4f 2F* | Measured | NIST | |
| 625.6013 nm | 80 | Sc III | emission | 3p6.4d 2D → 3p6.5p 2P* | Measured | NIST | |
| 503.2072 nm | 60 | Sc III | emission | 3p6.5p 2P* → 3p6.5d 2D | Measured | NIST | |
| 630.7603 nm | 60 | Sc III | emission | 3p6.4d 2D → 3p6.5p 2P* | Measured | NIST | |
| 499.2886 nm | 50 | Sc III | emission | 3p6.5p 2P* → 3p6.5d 2D | Measured | NIST | |
| 652.5571 nm | 40 | Sc I | emission | 3d.4s.(3D).4p 2D* → 3d.4s.(3D).4d 2D | Measured | NIST | |
| 671.4599 nm | 40 | Sc I | emission | 3d.4s.(3D).4p 2D* → 3d.4s.(3D).4d 4D | Measured | NIST | |
| 655.7842 nm | 35 | Sc I | emission | 3d.4s.(1D).4p 2F* → 3d3 2D2 | Measured | NIST | |
| 688.5119 nm | 27 | Sc I | emission | 3d2.(3F).4p 4F* → 3d2.(3F).4d 4G | Measured | NIST | |
| 716.9083 nm | 27 | Sc I | emission | 3d.4s.(3D).4p 2D* → 3d.4s.(3D).4d 2F | Measured | NIST | |
| 688.1012 nm | 26 | Sc I | emission | 3d2.(3F).4p 4F* → 3d2.(3F).4d 4G | Measured | NIST | |
| 662.0207 nm | 21 | Sc I | emission | 3d.4s.(3D).4p 2F* → 3d3 2F | Measured | NIST | |
| 713.8107 nm | 19 | Sc I | emission | 3d.4s.(3D).4p 2D* → 3d.4s.(3D).4d 2F | Measured | NIST | |
| 467.0407 nm | 18 | Sc II | emission | 3p6.3d2 1D → 3p6.3d.4p 1F* | Measured | NIST | |
| 673.0754 nm | 18 | Sc I | emission | 3d2.(3F).4p 4D* → 4P | Measured | NIST | |
| 687.7343 nm | 18 | Sc I | emission | 3d2.(3F).4p 4F* → 3d2.(3F).4d 4G | Measured | NIST | |
| 431.4083 nm | 17 | Sc II | emission | 3p6.3d2 3F → 3p6.3d.4p 3D* | Measured | NIST | |
| 503.1021 nm | 17 | Sc II | emission | 3p6.3d2 1D → 3p6.3d.4p 1P* | Measured | NIST | |
| 680.4611 nm | 17 | Sc I | emission | 3d2.(3F).4p 4F* → 3d2.(3F).4d 4D | Measured | NIST | |
| 437.4457 nm | 16 | Sc II | emission | 3p6.3d2 3F → 3p6.3d.4p 3F* | Measured | NIST | |
| 523.9813 nm | 16 | Sc II | emission | 3p6.4s2 1S → 3p6.3d.4p 1P* | Measured | NIST | |
| 552.679 nm | 16 | Sc II | emission | 3p6.3d2 1G → 3p6.3d.4p 1F* | Measured | NIST | |
| 430.5714 nm | 15 | Sc II | emission | 3p6.3d2 3F → 3p6.3d.4p 3D* | Measured | NIST | |
| 432.0732 nm | 15 | Sc II | emission | 3p6.3d2 3F → 3p6.3d.4p 3D* | Measured | NIST | |
| 478.0863 nm | 15 | Sc III | emission | 3p6.5p 2P* → 3p6.6s 2S | Measured | NIST | |
| 565.7896 nm | 15 | Sc II | emission | 3p6.3d2 3P → 3p6.3d.4p 3P* | Measured | NIST | |
| 624.5637 nm | 15 | Sc II | emission | 3p6.3d2 3P → 3p6.3d.4p 3D* | Measured | NIST | |
| 577.1538 nm | 14 | Sc IV | emission | 3s2.3p5.(2P*<3/2>).5s 2[3/2]* → 3s2.3p5.(2P*<3/2>).5p 2[5/2] | Measured | NIST | |
| 637.0486 nm | 14 | Sc II | emission | 3p6.3d.4d 1F → 3p6.3d.4f 1G* | Measured | NIST | |
| 660.4601 nm | 14 | Sc II | emission | 3p6.3d2 1D → 3p6.3d.4p 1D* | Measured | NIST | |
| 680.3677 nm | 14 | Sc I | emission | 3d.4s.(3D).4p 2F* → 3d.4s.(3D).4d 2G | Measured | NIST | |
| 725.7589 nm | 14 | Sc I | emission | 3d2.(3F).4p 4F* → 3d.(2D).4p2.(3P) 4F | Measured | NIST | |
| 401.4484 nm | 13 | Sc II | emission | 3p6.3d.4s 1D → 3p6.3d.4p 3F* | Measured | NIST | |
| 429.4767 nm | 13 | Sc II | emission | 3p6.3d2 3F → 3p6.3d.4p 3D* | Measured | NIST | |
| 432.4996 nm | 13 | Sc II | emission | 3p6.3d2 3F → 3p6.3d.4p 3D* | Measured | NIST | |
| 564.1001 nm | 13 | Sc II | emission | 3p6.3d2 3P → 3p6.3d.4p 3P* | Measured | NIST | |
| 565.8361 nm | 13 | Sc II | emission | 3p6.3d2 3P → 3p6.3d.4p 3P* | Measured | NIST | |
| 566.9042 nm | 13 | Sc II | emission | 3p6.3d2 3P → 3p6.3d.4p 3P* | Measured | NIST | |
| 687.4193 nm | 13 | Sc I | emission | 3d2.(3F).4p 4F* → 3d2.(3F).4d 4G | Measured | NIST | |
| 385.9595 nm | 12 | Sc II | emission | 3p6.3d.4p 1F* → 3p6.3d.5s 1D | Measured | NIST | |
| 424.6822 nm | 12 | Sc II | emission | 3p6.3d.4s 1D → 3p6.3d.4p 1D* | Measured | NIST | |
| 435.4598 nm | 12 | Sc II | emission | 3p6.3d2 3F → 3p6.3d.4p 3F* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Chemical Affinity
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 1814.15 K |
| Boiling point | 3109.15 K |
Oxidation State Categories
Advanced Reference Data
Screening Constants (7)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.5434 |
| 2 | p | 3.9454 |
| 2 | s | 6.4264 |
| 3 | d | 13.8801 |
| 3 | p | 11.5938 |
| 3 | s | 10.6602 |
| 4 | s | 16.3676 |
Crystal Radii Detail (2)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 3 | VI | 88.5 | from r^3 vs V plots, | |
| 3 | VIII | 101 | from r^3 vs V plots, |
Isotope Decay Modes (52)
| Isotope | Mode | Intensity |
|---|---|---|
| 35 | p | — |
| 36 | p | — |
| 37 | p | — |
| 38 | p | — |
| 39 | p | 100% |
| 40 | B+ | 100% |
| 40 | B+p | 0.4% |
| 40 | B+A | 0% |
| 41 | B+ | 100% |
| 42 | B+ | 100% |
X‑ray Scattering Factors (598)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 1.06978 |
| 10.1617 | — | 1.07987 |
| 10.3261 | — | 1.09005 |
| 10.4931 | — | 1.10033 |
| 10.6628 | — | 1.11071 |
| 10.8353 | — | 1.12118 |
| 11.0105 | — | 1.13176 |
| 11.1886 | — | 1.14243 |
| 11.3696 | — | 1.15321 |
| 11.5535 | — | 1.16408 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
2.2×101 milligrams per kilogram
References (1)
- [5] Scandium https://education.jlab.org/itselemental/ele021.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
6×10-7 milligrams per liter
References (1)
- [5] Scandium https://education.jlab.org/itselemental/ele021.html
Sources
Sources of this element.
Scandium is apparently much more abundant (the 23rd most) in the sun and certain stars than on earth (the 50th most abundant). It is widely distributed on earth, occurring in very minute quantities in over 800 mineral species. The blue color of beryl (aquamarine variety) is said to be due to scandium. It occurs as a principal component in the rare mineral thortveitite, found in Scandinavia and Malagasy. It is also found in the residues remaining after the extraction of tungsten from Zinnwald wolframite, and in wiikite and bazzite.
Most scandium is presently being recovered from thortveitite or is extracted as a by-product from uranium mill tailings. Metallic scandium was first prepared in 1937 by Fischer, Brunger, and Grienelaus who electrolyzed a eutectic melt of potassium, lithium, and scandium chlorides at 700 to 800°C. Tungsten wire and a pool of molten zinc served as the electrodes in a graphite crucible. Pure scandium is now produced by reducing scandium fluoride with calcium metal.
The production of the first pound of 99% pure scandium metal was announced in 1960.
References (1)
- [6] Scandium https://periodic.lanl.gov/21.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Scandium.
The element property data was retrieved from publications.