Potassium (K)
alkali-metalSolid
Standard Atomic Weight
39.0983 uElectron configuration
[Ar] 4s1Melting point
63.38 °C (336.53 K)Boiling point
758.85 °C (1032 K)Density
890 kg/m³Oxidation states
−1, +1Electronegativity (Pauling)
0.82Ionization energy (1st)
Discovery year
1807Atomic radius
220 pmDetails
Potassium is an alkali metal and a major rock-forming and biological element. It occurs naturally only in compounds, chiefly as K⁺ in salts, feldspars, micas, and clay minerals. The metal is highly electropositive, reacts vigorously with water, and is stored away from air and moisture. In living cells, potassium is the principal intracellular cation and is central to osmotic balance and electrical signaling.
It is one of the most reactive and electropositive of metals. Except for lithium, it is the lightest known metal. It is soft, easily cut with a knife, and is silvery in appearance immediately after a fresh surface is exposed. It rapidly oxidizes in air and must be preserved in a mineral oil such as kerosene.
As with other metals of the alkali group, it decomposes in water with the evolution of hydrogen. It catches fire spontaneously on water. Potassium and its salts impart a violet color to flames.
The name derives from the English "potash" or "pot ashes" because it is found in caustic potash (KOH). The symbol K derives from the Latin kalium via the Arabic qali for alkali. It was first isolated by the British chemist Humphry Davy in 1807 from electrolysis of potash (KOH).
Although potassium is the eighth most abundant element on earth and comprises about 2.1% of the earth's crust, it is a very reactive element and is never found free in nature. Metallic potassium was first isolated by Sir Humphry Davy in 1807 through the electrolysis of molten caustic potash (KOH). A few months after discovering potassium, Davy used the same method to isolate sodium. Potassium can be obtained from the minerals sylvite (KCl), carnallite (KCl·MgCl2·6H2O), langbeinite (K2Mg2(SO4)3) and polyhalite (K2Ca2Mg(SO4)4·2H2O). These minerals are often found in ancient lake and sea beds. Caustic potash, another important source of potassium, is primarily mined in Germany, New Mexico, California and Utah. Pure potassium is a soft, waxy metal that can be easily cut with a knife. It reacts with oxygen to form potassium superoxide (KO2) and with water to form potassium hydroxide (KOH), hydrogen gas and heat. Enough heat is produced to ignite the hydrogen gas. To prevent it from reacting with the oxygen and water in the air, samples of metallic potassium are usually stored submerged in mineral oil.
From the English word, potash - pot ashes; Latin kalium, Arab qali, alkali. Discovered in 1807 by Davy, who obtained it from caustic potash (KOH); this was the first metal isolated by electrolysis.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
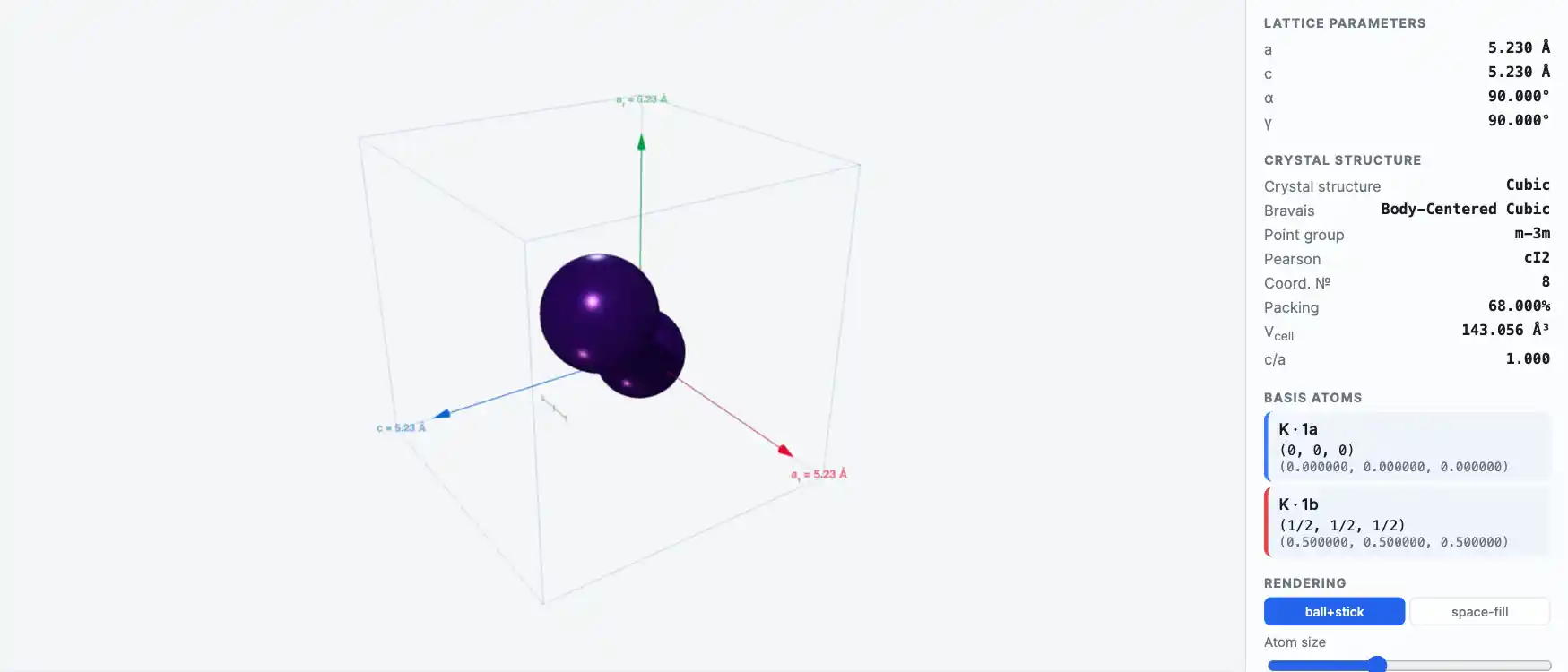
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
K: 4s¹[Ar] 4s¹1s² 2s² 2p⁶ 3s² 3p⁶ 4s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 39 Stable | 38.9637064864 ± 0.0000000049 | 93.2581% | Stable |
| 41 Stable | 40.9618252579 ± 0.0000000041 | 6.7302% | Stable |
Phase / State
Reason: 38.4 °C below melting point (63.38 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Energy required to sublime 1 mol at sublimation point
Density
At standard conditions
At standard conditions
Advanced
Atomic Spectra
Showing 10 of 22 Atomic Spectra. Sorted by ion charge (ascending).
Levels Holdings ?
| Ion | Charge | Levels |
|---|---|---|
| 41K I Isotope | 0 | 8 |
| 40K I Isotope | 0 | 3 |
| 39K I Isotope | 0 | 123 |
| K I | 0 | 299 |
| K II | +1 | 97 |
| K III | +2 | 40 |
| K IV | +3 | 38 |
| K V | +4 | 40 |
| K VI | +5 | 28 |
| K VII | +6 | 81 |
Ionic Radii
| Charge | Coordination | Spin | Radius |
|---|---|---|---|
| +1 | 4 | N/A | 137 pm |
| +1 | 6 | N/A | 138 pm |
| +1 | 7 | N/A | 146 pm |
| +1 | 8 | N/A | 151 pm |
| +1 | 9 | N/A | 155 pm |
| +1 | 10 | N/A | 159 pm |
| +1 | 12 | N/A | 164 pm |
Compounds
Isotopes (2)
Seventeen isotopes of potassium are known. Ordinary potassium is composed of three isotopes, one of which is 40°K (0.0118%), a radioactive isotope with a half-life of 1.28 x 109 years.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 39 Stable | 38.9637064864 ± 0.0000000049 | 93.2581% ± 0.0044% | Stable | stable | |
| 41 Stable | 40.9618252579 ± 0.0000000041 | 6.7302% ± 0.0044% | Stable | stable |
Spectral Lines
Showing 50 of 141 Spectral Lines. Only spectral lines with measured intensity are shown by default.
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 693.8764 nm | 20 | K I | emission | 3p6.4p 2P* → 3p6.6s 2S | Measured | NIST | |
| 691.10815 nm | 19 | K I | emission | 3p6.4p 2P* → 3p6.6s 2S | Measured | NIST | |
| 404.41422 nm | 18 | K I | emission | 3p6.4s 2S → 3p6.5p 2P* | Measured | NIST | |
| 404.72132 nm | 17 | K I | emission | 3p6.4s 2S → 3p6.5p 2P* | Measured | NIST | |
| 580.17662 nm | 17 | K I | emission | 3p6.4p 2P* → 3p6.7s 2S | Measured | NIST | |
| 583.18899 nm | 17 | K I | emission | 3p6.4p 2P* → 3p6.5d 2D | Measured | NIST | |
| 578.23999 nm | 16 | K I | emission | 3p6.4p 2P* → 3p6.7s 2S | Measured | NIST | |
| 581.21521 nm | 15 | K I | emission | 3p6.4p 2P* → 3p6.5d 2D | Measured | NIST | |
| 535.95761 nm | 14 | K I | emission | 3p6.4p 2P* → 3p6.6d 2D | Measured | NIST | |
| 533.96873 nm | 13 | K I | emission | 3p6.4p 2P* → 3p6.8s 2S | Measured | NIST | |
| 511.225448 nm | 12 | K I | emission | 3p6.4p 2P* → 3p6.7d 2D | Measured | NIST | |
| 532.32786 nm | 12 | K I | emission | 3p6.4p 2P* → 3p6.8s 2S | Measured | NIST | |
| 534.29693 nm | 12 | K I | emission | 3p6.4p 2P* → 3p6.6d 2D | Measured | NIST | |
| 693.62861 nm | 12 | K I | emission | 3p6.4p 2P* → 3p6.4d 2D | Measured | NIST | |
| 696.46903 nm | 12 | K I | emission | 3p6.4p 2P* → 3p6.4d 2D | Measured | NIST | |
| 464.23725 nm | 11 | K I | emission | 3p6.4s 2S → 3p6.3d 2D | Measured | NIST | |
| 509.717137 nm | 11 | K I | emission | 3p6.4p 2P* → 3p6.7d 2D | Measured | NIST | |
| 509.920005 nm | 11 | K I | emission | 3p6.4p 2P* → 3p6.9s 2S | Measured | NIST | |
| 464.1875 nm | 10 | K I | emission | 3p6.4s 2S → 3p6.3d 2D | Measured | NIST | |
| 496.503213 nm | 10 | K I | emission | 3p6.4p 2P* → 3p6.8d 2D | Measured | NIST | |
| 508.423399 nm | 10 | K I | emission | 3p6.4p 2P* → 3p6.9s 2S | Measured | NIST | |
| 482.924 nm | 9 | K II | emission | 3p5.4s 3P* → 3p5.4p 3S | Measured | NIST | |
| 486.975897 nm | 9 | K I | emission | 3p6.4p 2P* → 3p6.9d 2D | Measured | NIST | |
| 495.081801 nm | 9 | K I | emission | 3p6.4p 2P* → 3p6.8d 2D | Measured | NIST | |
| 495.614802 nm | 9 | K I | emission | 3p6.4p 2P* → 3p6.10s 2S | Measured | NIST | |
| 389.7896 nm | 8 | K II | emission | 3p5.4s 3P* → 3p5.4p 1D | Measured | NIST | |
| 418.6232 nm | 8 | K II | emission | 3p5.4s 3P* → 3p5.4p 3D | Measured | NIST | |
| 460.849 nm | 8 | K II | emission | 3p5.4s 1P* → 3p5.4p 1D | Measured | NIST | |
| 480.43395 nm | 8 | K I | emission | 3p6.4p 2P* → 3p6.10d 2D | Measured | NIST | |
| 485.609209 nm | 8 | K I | emission | 3p6.4p 2P* → 3p6.9d 2D | Measured | NIST | |
| 486.348075 nm | 8 | K I | emission | 3p6.4p 2P* → 3p6.11s 2S | Measured | NIST | |
| 500.564 nm | 8 | K II | emission | 3p5.4s 3P* → 3p5.4p 3S | Measured | NIST | |
| 612.028 nm | 8 | K II | emission | 3p5.3d 3F* → 3p5.4p 3D | Measured | NIST | |
| 381.7547 nm | 7 | K II | emission | 3p5.4p 3D → 3p5.(2P*<3/2>).5s 2[3/2]* | Measured | NIST | |
| 400.122 nm | 7 | K II | emission | 3p5.4s 3P* → 3p5.4p 3P | Measured | NIST | |
| 413.4705 nm | 7 | K II | emission | 3p5.4s 3P* → 3p5.4p 3D | Measured | NIST | |
| 422.296 nm | 7 | K II | emission | 3p5.4s 1P* → 3p5.4p 3P | Measured | NIST | |
| 422.566 nm | 7 | K II | emission | 3p5.3d 3P* → 3p5.4p 1D | Measured | NIST | |
| 426.334 nm | 7 | K II | emission | 3p5.4s 3P* → 3p5.4p 3D | Measured | NIST | |
| 430.498 nm | 7 | K II | emission | 3p5.3d 3P* → 3p5.4p 1D | Measured | NIST | |
| 430.911 nm | 7 | K II | emission | 3p5.4s 1P* → 3p5.4p 3P | Measured | NIST | |
| 438.816 nm | 7 | K II | emission | 3p5.4s 1P* → 3p5.4p 1P | Measured | NIST | |
| 475.737719 nm | 7 | K I | emission | 3p6.4p 2P* → 3p6.11d 2D | Measured | NIST | |
| 479.104132 nm | 7 | K I | emission | 3p6.4p 2P* → 3p6.10d 2D | Measured | NIST | |
| 484.98645 nm | 7 | K I | emission | 3p6.4p 2P* → 3p6.11s 2S | Measured | NIST | |
| 505.625 nm | 7 | K II | emission | 3p5.3d 3P* → 3p5.4p 3S | Measured | NIST | |
| 630.728 nm | 7 | K II | emission | 3p5.3d 3F* → 3p5.4p 3D | Measured | NIST | |
| 696.41712 nm | 7 | K I | emission | 3p6.4p 2P* → 3p6.4d 2D | Measured | NIST | |
| 380.0162 nm | 6 | K II | emission | 3p5.4p 3D → 3p5.(2P*<3/2>).5s 2[3/2]* | Measured | NIST | |
| 381.657 nm | 6 | K II | emission | 3p5.4p 3P → 3p5.4d 3P* | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Supply Risk & Economics
Phase Transitions & Allotropes
| Melting point | 336.65 K |
| Boiling point | 1032.15 K |
| Critical point (temperature) | 2223.15 K |
| Critical point (pressure) | 16 MPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (6)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0.5105 |
| 2 | p | 3.9728 |
| 2 | s | 5.9938 |
| 3 | p | 11.2744 |
| 3 | s | 10.3201 |
| 4 | s | 15.5048 |
Crystal Radii Detail (7)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 1 | IV | 151 | ||
| 1 | VI | 152 | ||
| 1 | VII | 160 | ||
| 1 | VIII | 165 | ||
| 1 | IX | 169 | ||
| 1 | X | 173 | ||
| 1 | XII | 178 |
Isotope Decay Modes (53)
| Isotope | Mode | Intensity |
|---|---|---|
| 31 | 3p | 100% |
| 32 | p | — |
| 33 | p | — |
| 34 | p | — |
| 35 | B+ | 100% |
| 35 | B+p | 0.4% |
| 36 | B+ | 100% |
| 36 | B+p | 0% |
| 36 | B+A | 0% |
| 37 | B+ | 100% |
X‑ray Scattering Factors (503)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0.03426 |
| 10.1617 | — | 0.03529 |
| 10.3261 | — | 0.03635 |
| 10.4931 | — | 0.03744 |
| 10.6628 | — | 0.03856 |
| 10.8353 | — | 0.03972 |
| 11.0106 | — | 0.04091 |
| 11.1886 | — | 0.04214 |
| 11.3696 | — | 0.0434 |
| 11.5535 | — | 0.04471 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
2.09×104 milligrams per kilogram
References (1)
- [5] Potassium https://education.jlab.org/itselemental/ele019.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
3.99×102 milligrams per liter
References (1)
- [5] Potassium https://education.jlab.org/itselemental/ele019.html
Sources
Sources of this element.
The metal is the seventh most abundant and makes up about 2.4% by weight of the earth's crust. Most potassium minerals are insoluble and the metal is obtained from them only with great difficulty.
Certain minerals, however, such as sylvite, carnallite, langbeinite, and polyhalite are found in ancient lake and sea beds and form rather extensive deposits from which potassium and its salts can readily be obtained. Potash is mined in Germany, New Mexico, California, Utah, and elsewhere. Large deposits of potash, found at a depth of some 3000 ft in Saskatchewan, promise to be important in coming years.
Potassium is also found in the ocean, but is present only in relatively small amounts, compared to sodium.
References (1)
- [6] Potassium https://periodic.lanl.gov/19.shtml
Production
Production of this element (from raw materials or other compounds containing the element).
Potassium is never found free in nature, but is obtained by electrolysis of the hydroxide, much in the same manner as prepared by Davy's first process. Thermal methods also are commonly used to produce potassium (such as by reduction of potassium compounds with CaC2, C, Si, or Na).
References (1)
- [6] Potassium https://periodic.lanl.gov/19.shtml
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Potassium.
The element property data was retrieved from publications.