Hydrogen (H)
nonmetalGas
Standard Atomic Weight
1.008 u [1.00784, 1.00811]Electron configuration
1s1Melting point
-259.34 °C (13.81 K)Boiling point
-252.87 °C (20.28 K)Density
0.08988 kg/m³Oxidation states
−1, +1Electronegativity (Pauling)
2.2Ionization energy (1st)
Discovery year
1766Atomic radius
25 pmDetails
The lightest chemical element, hydrogen sits above the alkali metals but is a nonmetal under ordinary conditions. Its single proton and one electron make it the simplest atom and a reference point for much of chemistry. On Earth it is usually bound in water and organic matter, while industry uses it as a chemical feedstock, reducing agent, fuel, and energy carrier.
Colourless, odourless gaseous chemical element. Lightest and most abundant element in the universe. Present in water and in all organic compounds. Chemically reacts with most elements. Discovered by Henry Cavendish in 1776.
The name derives from the Greek hydro for "water" and genes for "forming" because it burned in air to form water. Hydrogen was discovered by the English physicist Henry Cavendish in 1766.
Scientists had been producing hydrogen for years before it was recognized as an element. Written records indicate that Robert Boyle produced hydrogen gas as early as 1671 while experimenting with iron and acids. Hydrogen was first recognized as a distinct element by Henry Cavendish in 1766. Composed of a single proton and a single electron, hydrogen is the simplest and most abundant element in the universe. It is estimated that 90% of the visible universe is composed of hydrogen.
Hydrogen is the raw fuel that most stars 'burn' to produce energy. The same process, known as fusion, is being studied as a possible power source for use on earth. The sun's supply of hydrogen is expected to last another 5 billion years.
From the Greek word hydro (water), and genes (forming). Hydrogen was recognized as a distinct substance by Henry Cavendish in 1776. Diagram of a simple hydrogen atom.
Hydrogen is the most abundant of all elements in the universe. The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms.
Images
Properties
Physical
Chemical
Thermodynamic
Nuclear
Abundance
Reactivity
N/A
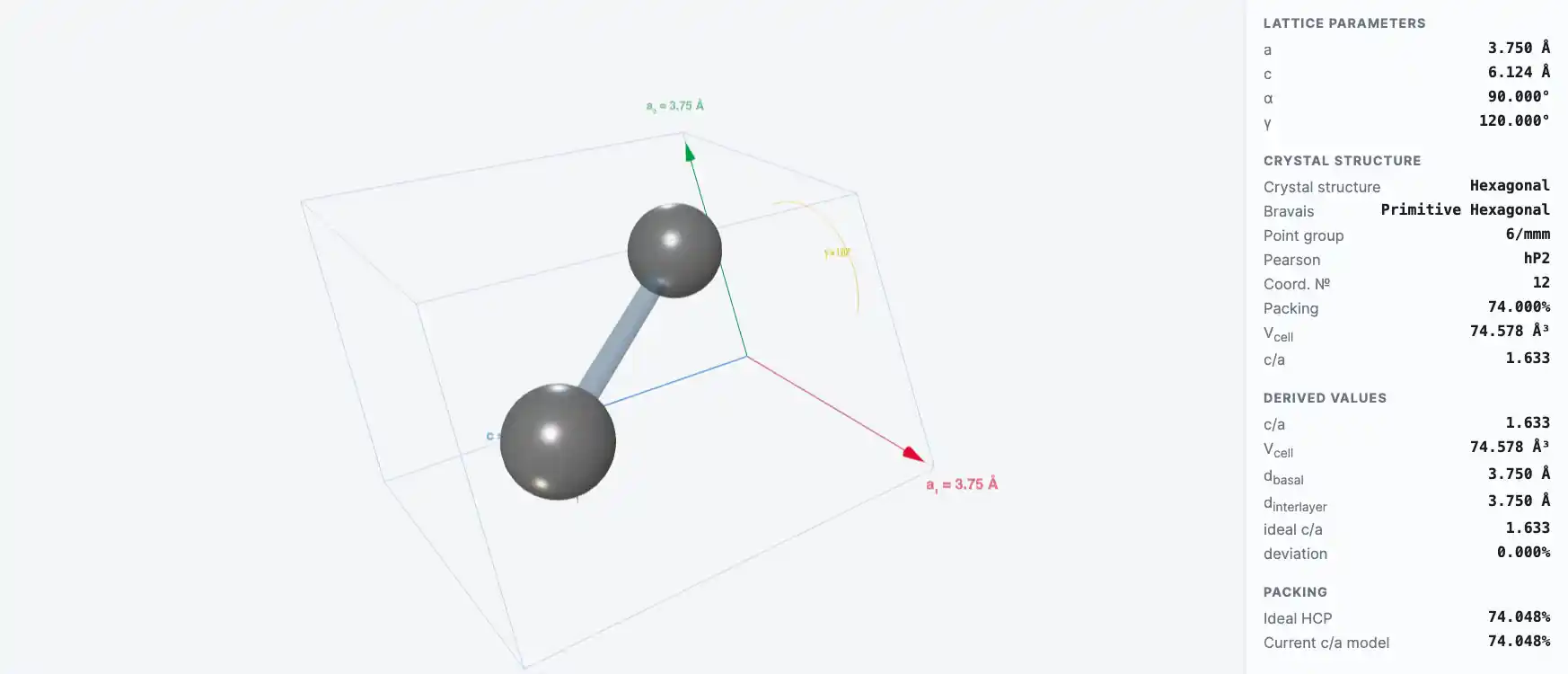
Crystal Structure
Electronic Structure
Identifiers
Electron Configuration Measured
H: 1s¹1s¹1s¹Atomic model
Isotopes change neutron count, mass, and stability — not the electron configuration of a neutral atom.
Schematic atomic model, not to scale.
Atomic Fingerprint
Emission / Absorption Spectrum
Isotope Distribution
| Mass number | Atomic mass (u) | Natural abundance | Half-life |
|---|---|---|---|
| 1 Stable | 1.00782503223 ± 0.00000000009 | 99.9885% | Stable |
| 2 Stable | 2.01410177812 ± 0.00000000012 | 0.0115% | Stable |
Phase / State
Reason: 277.9 °C above boiling point (-252.87 °C)
Schematic, not to scale
Phase transition points
Transition energies
Energy required to melt 1 mol at melting point
Energy required to vaporize 1 mol at boiling point
Density
At standard conditions
Estimated via ideal gas law at current T
Advanced
Atomic Spectra
Lines Holdings ?
| Ion | Charge | Total lines | Transition probabilities | Level designations |
|---|---|---|---|---|
| D I Isotope | 0 | 364 | 161 | 341 |
| H I | 0 | 568 | 441 | 535 |
| T I Isotope | 0 | 11 | 11 | 11 |
Compounds
Isotopes (2)
The ordinary isotope of hydrogen, H, is known as Protium, the other two isotopes are Deuterium (a proton and a neutron) and Tritium (a protron and two neutrons). Hydrogen is the only element whose isotopes have been given different names. Deuterium and Tritium are both used as fuel in nuclear fusion reactors. One atom of Deuterium is found in about 6000 ordinary hydrogen atoms.
| Mass number | Atomic mass (u) | Natural abundance | Half-life | Decay mode | |
|---|---|---|---|---|---|
| 1 Stable | 1.00782503223 ± 0.00000000009 | 99.9885% ± 0.0070% | Stable | stable | |
| 2 Stable | 2.01410177812 ± 0.00000000012 | 0.0115% ± 0.0070% | Stable | stable |
Spectral Lines
| Wavelength (nm) | Intensity | Ion stage | Type | Transition | Accuracy | Source | |
|---|---|---|---|---|---|---|---|
| 383.5355892 nm | N/A | H I | emission | 2p 2P* → 9d 2D | Measured | NIST | |
| 383.5356424 nm | N/A | H I | emission | 2p 2P* → 9s 2S | Measured | NIST | |
| 383.53587 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 383.5361082 nm | N/A | H I | emission | 2s 2S → 9p 2P* | Measured | NIST | |
| 383.5361616 nm | N/A | H I | emission | 2s 2S → 9s 2S | Measured | NIST | |
| 383.5361673 nm | N/A | H I | emission | 2s 2S → 9p 2P* | Measured | NIST | |
| 383.53839 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 383.53909 nm | 30000 | H I | emission | 2 → 9 | Measured | NIST | |
| 383.5409535 nm | N/A | H I | emission | 2p 2P* → 9d 2D | Measured | NIST | |
| 383.54096 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 383.5409732 nm | N/A | H I | emission | 2p 2P* → 9d 2D | Measured | NIST | |
| 383.5410263 nm | N/A | H I | emission | 2p 2P* → 9s 2S | Measured | NIST | |
| 388.9019815 nm | N/A | H I | emission | 2p 2P* → 8d 2D | Measured | NIST | |
| 388.9020595 nm | N/A | H I | emission | 2p 2P* → 8s 2S | Measured | NIST | |
| 388.90227 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 388.9024865 nm | N/A | H I | emission | 2s 2S → 8d 2D | Measured | NIST | |
| 388.9025152 nm | N/A | H I | emission | 2s 2S → 8p 2P* | Measured | NIST | |
| 388.9025154 nm | N/A | H I | emission | 2s 2S → 8d 2D | Measured | NIST | |
| 388.9025933 nm | N/A | H I | emission | 2s 2S → 8s 2S | Measured | NIST | |
| 388.9026017 nm | N/A | H I | emission | 2s 2S → 8p 2P* | Measured | NIST | |
| 388.90486 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 388.90557 nm | 70000 | H I | emission | 2 → 8 | Measured | NIST | |
| 388.9074883 nm | N/A | H I | emission | 2p 2P* → 8d 2D | Measured | NIST | |
| 388.90749 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 388.9075172 nm | N/A | H I | emission | 2p 2P* → 8d 2D | Measured | NIST | |
| 388.9075951 nm | N/A | H I | emission | 2p 2P* → 8s 2S | Measured | NIST | |
| 397.0041763 nm | N/A | H I | emission | 2p 2P* → 7d 2D | Measured | NIST | |
| 397.0042976 nm | N/A | H I | emission | 2p 2P* → 7s 2S | Measured | NIST | |
| 397.00448 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 397.0047325 nm | N/A | H I | emission | 2s 2S → 7p 2P* | Measured | NIST | |
| 397.0048539 nm | N/A | H I | emission | 2s 2S → 7s 2S | Measured | NIST | |
| 397.004867 nm | N/A | H I | emission | 2s 2S → 7p 2P* | Measured | NIST | |
| 397.00719 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 397.00788 nm | 30000 | H I | emission | 2 → 7 | Measured | NIST | |
| 397.0099002 nm | N/A | H I | emission | 2p 2P* → 7d 2D | Measured | NIST | |
| 397.00991 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 397.009945 nm | N/A | H I | emission | 2p 2P* → 7d 2D | Measured | NIST | |
| 397.0100663 nm | N/A | H I | emission | 2p 2P* → 7s 2S | Measured | NIST | |
| 410.1702284 nm | N/A | H I | emission | 2p 2P* → 6d 2D | Measured | NIST | |
| 410.1704339 nm | N/A | H I | emission | 2p 2P* → 6s 2S | Measured | NIST | |
| 410.17056 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 410.1707462 nm | N/A | H I | emission | 2s 2S → 6d 2D | Measured | NIST | |
| 410.1708218 nm | N/A | H I | emission | 2s 2S → 6p 2P* | Measured | NIST | |
| 410.1710277 nm | N/A | H I | emission | 2s 2S → 6s 2S | Measured | NIST | |
| 410.1710499 nm | N/A | H I | emission | 2s 2S → 6p 2P* | Measured | NIST | |
| 410.17346 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 410.17415 nm | 70000 | H I | emission | 2 → 6 | Measured | NIST | |
| 410.17631 nm | N/A | H I | emission | 2p 2P* → 6d 2D | Measured | NIST | |
| 410.17632 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 410.176386 nm | N/A | H I | emission | 2p 2P* → 6d 2D | Measured | NIST | |
| 410.1765915 nm | N/A | H I | emission | 2p 2P* → 6s 2S | Measured | NIST | |
| 434.0426937 nm | N/A | H I | emission | 2p 2P* → 5d 2D | Measured | NIST | |
| 434.04309 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 434.0430904 nm | N/A | H I | emission | 2p 2P* → 5s 2S | Measured | NIST | |
| 434.0433568 nm | N/A | H I | emission | 2s 2S → 5p 2P* | Measured | NIST | |
| 434.0437554 nm | N/A | H I | emission | 2s 2S → 5s 2S | Measured | NIST | |
| 434.0437982 nm | N/A | H I | emission | 2s 2S → 5p 2P* | Measured | NIST | |
| 434.04634 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 434.0471 nm | 90000 | H I | emission | 2 → 5 | Measured | NIST | |
| 434.0494419 nm | N/A | H I | emission | 2p 2P* → 5d 2D | Measured | NIST | |
| 434.04947 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 434.0495889 nm | N/A | H I | emission | 2p 2P* → 5d 2D | Measured | NIST | |
| 434.0499857 nm | N/A | H I | emission | 2p 2P* → 5s 2S | Measured | NIST | |
| 486.1278624 nm | N/A | H I | emission | 2p 2P* → 4d 2D | Measured | NIST | |
| 486.1283363 nm | N/A | H I | emission | 2s 2S → 4d 2D | Measured | NIST | |
| 486.12841 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 486.1286949 nm | N/A | H I | emission | 2s 2S → 4p 2P* | Measured | NIST | |
| 486.128837 nm | N/A | H I | emission | 2p 2P* → 4s 2S | Measured | NIST | |
| 486.1296711 nm | N/A | H I | emission | 2s 2S → 4s 2S | Measured | NIST | |
| 486.1297761 nm | N/A | H I | emission | 2s 2S → 4p 2P* | Measured | NIST | |
| 486.1325 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 486.1333 nm | 180000 | H I | emission | 2 → 4 | Measured | NIST | |
| 486.1361516 nm | N/A | H I | emission | 2p 2P* → 4d 2D | Measured | NIST | |
| 486.13622 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 486.1365118 nm | N/A | H I | emission | 2p 2P* → 4d 2D | Measured | NIST | |
| 486.1374864 nm | N/A | H I | emission | 2p 2P* → 4s 2S | Measured | NIST | |
| 656.27097 nm | N/A | H I | emission | 2p 2P* → 3d 2D | Measured | NIST | |
| 656.2714 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 656.2722 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 656.272483 nm | N/A | H I | emission | 2s 2S → 3p 2P* | Measured | NIST | |
| 656.275181 nm | N/A | H I | emission | 2p 2P* → 3s 2S | Measured | NIST | |
| 656.276701 nm | N/A | H I | emission | 2s 2S → 3s 2S | Measured | NIST | |
| 656.277 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 656.277153 nm | N/A | H I | emission | 2s 2S → 3p 2P* | Measured | NIST | |
| 656.2795 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 656.2819 nm | 500000 | H I | emission | 2 → 3 | Measured | NIST | |
| 656.285177 nm | N/A | H I | emission | 2p 2P* → 3d 2D | Measured | NIST | |
| 656.28533 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 656.2854 nm | N/A | H I | emission | N/A | Measured | NIST | |
| 656.286734 nm | N/A | H I | emission | 2p 2P* → 3d 2D | Measured | NIST | |
| 656.290944 nm | N/A | H I | emission | 2p 2P* → 3s 2S | Measured | NIST |
Extended Properties
Covalent Radii (Extended)
Van der Waals Radii
Atomic & Metallic Radii
Numbering Scales
Electronegativity Scales
Polarizability & Dispersion
Miedema Parameters
Phase Transitions & Allotropes
| Melting point | 13.99 K |
| Boiling point | 20.27 K |
| Critical point (temperature) | 32.94 K |
| Critical point (pressure) | 1.29 MPa |
| Triple point (temperature) | 13.8 K |
| Triple point (pressure) | 7.04 kPa |
Oxidation State Categories
Advanced Reference Data
Screening Constants (1)
| n | Orbital | σ |
|---|---|---|
| 1 | s | 0 |
Crystal Radii Detail (2)
| Charge | CN | Spin | rcrystal (pm) | Origin |
|---|---|---|---|---|
| 1 | I | -24 | ||
| 1 | II | -4 |
Isotope Decay Modes (6)
| Isotope | Mode | Intensity |
|---|---|---|
| 3 | B- | 100% |
| 4 | n | 100% |
| 5 | 2n | 100% |
| 6 | n | — |
| 6 | 3n | — |
| 7 | 2n | — |
X‑ray Scattering Factors (501)
| Energy (eV) | f₁ | f₂ |
|---|---|---|
| 10 | — | 0 |
| 10.1617 | — | 0 |
| 10.3261 | — | 0 |
| 10.4931 | — | 0 |
| 10.6628 | — | 0 |
| 10.8353 | — | 0 |
| 11.0106 | — | 0 |
| 11.1886 | — | 0 |
| 11.3696 | — | 0 |
| 11.5535 | — | 0 |
Additional Data
Estimated Crustal Abundance
The estimated element abundance in the earth's crust.
1.40×103 milligrams per kilogram
References (1)
- [5] Hydrogen https://education.jlab.org/itselemental/ele001.html
Estimated Oceanic Abundance
The estimated element abundance in the earth's oceans.
1.08×105 milligrams per liter
References (1)
- [5] Hydrogen https://education.jlab.org/itselemental/ele001.html
Sources
Sources of this element.
Hydrogen is estimated to make up more than 90% of all the atoms three quarters of the mass of the universe! This element is found in the stars, and plays an important part in powering the universe through both the proton-proton reaction and carbon-nitrogen cycle. Stellar hydrogen fusion processes release massive amounts of energy by combining hydrogens to form helium.
Production of hydrogen in the U.S. alone amounts to about 3 billion cubic feet per year. Hydrogen is prepared by
▸ steam on heated carbon,
▸ decomposition of certain hydrocarbons with heat,
▸ reaction of sodium or potassium hydroxide on aluminum
▸ electrolysis of water, or
▸ displacement from acids by certain metals.
Liquid hydrogen is important in cryogenics and in the study of superconductivity, as its melting point is only 20 degrees above absolute zero.
Tritium is readily produced in nuclear reactors and is used in the production of the hydrogen bomb.
Hydrogen is the primary component of Jupiter and the other gas giant planets. At some depth in the planet's interior the pressure is so great that solid molecular hydrogen is converted to solid metallic hydrogen.
In 1973, a group of Russian experimenters may have produced metallic hydrogen at a pressure of 2.8 Mbar. At the transition the density changed from 1.08 to 1.3 g/cm3. Earlier, in 1972, at Livermore, California, a group also reported on a similar experiment in which they observed a pressure-volume point centered at 2 Mbar. Predictions say that metallic hydrogen may be metastable; others have predicted it would be a superconductor at room temperature.
References (1)
- [6] Hydrogen https://periodic.lanl.gov/1.shtml
Isotopes in Forensic Science and Anthropology
Information on the use of this element's isotopes in forensic science and anthropology.
Measurements of relative 2H abundances are used to determine the breeding grounds of many species of migrant songbirds. These species of songbirds only grow their feathers before migration, and they grow them on or close to their breeding grounds. Therefore, the isotopic composition of a bird’s feathers correlates to the isotopic composition of the growing season’s precipitation [18] [18] Z. D. Sharp, V. Atudorei, H. O. Panarello, J. Fernández, C. Douthitt. J. Archaeolog. Sci.30, 1709 (2003).[18] Z. D. Sharp, V. Atudorei, H. O. Panarello, J. Fernández, C. Douthitt. J. Archaeolog. Sci.30, 1709 (2003)., [19] [19] K. A. Hobson. Oecologia120, 314 (1999).[19] K. A. Hobson. Oecologia120, 314 (1999)., [20] [20] K. A. Hobson, L. I. Wassenaar. Oecologia.109, 142 (1996).[20] K. A. Hobson, L. I. Wassenaar. Oecologia.109, 142 (1996)..
Measurements of relative 2H abundances of human hair samples collected at archeological sites are used to determine the geographic region in which a subject lived based on the hydrogen isotopic composition of the water they drank. This is possible because hair stores a daily record of the hydrogen isotopic composition of intake water, which correlates to local meteoric water [18] [18] Z. D. Sharp, V. Atudorei, H. O. Panarello, J. Fernández, C. Douthitt. J. Archaeolog. Sci.30, 1709 (2003).[18] Z. D. Sharp, V. Atudorei, H. O. Panarello, J. Fernández, C. Douthitt. J. Archaeolog. Sci.30, 1709 (2003)., [21] [21] T. B. Coplen, H. Qi. Forensic Sci. Int.266, 222 (2016).[21] T. B. Coplen, H. Qi. Forensic Sci. Int.266, 222 (2016)..
References (5)
- [18] Z. D. Sharp, V. Atudorei, H. O. Panarello, J. Fernández, C. Douthitt. J. Archaeolog. Sci.30, 1709 (2003).
- [19] K. A. Hobson. Oecologia120, 314 (1999).
- [20] K. A. Hobson, L. I. Wassenaar. Oecologia.109, 142 (1996).
- [21] T. B. Coplen, H. Qi. Forensic Sci. Int.266, 222 (2016).
- [4] IUPAC Periodic Table of the Elements and Isotopes (IPTEI) https://doi.org/10.1515/pac-2015-0703
References
(9)
Data deposited in or computed by PubChem
The half-life and atomic mass data was provided by the Atomic Mass Data Center at the International Atomic Energy Agency.
Element data are cited from the Atomic weights of the elements (an IUPAC Technical Report). The IUPAC periodic table of elements can be found at https://iupac.org/what-we-do/periodic-table-of-elements/. Additional information can be found within IUPAC publication doi:10.1515/pac-2015-0703 Copyright © 2020 International Union of Pure and Applied Chemistry.
The information are cited from Pure Appl. Chem. 2018; 90(12): 1833-2092, https://doi.org/10.1515/pac-2015-0703.
Thomas Jefferson National Accelerator Facility (Jefferson Lab) is one of 17 national laboratories funded by the U.S. Department of Energy. The lab's primary mission is to conduct basic research of the atom's nucleus using the lab's unique particle accelerator, known as the Continuous Electron Beam Accelerator Facility (CEBAF). For more information visit https://www.jlab.org/
The periodic table at the LANL (Los Alamos National Laboratory) contains basic element information together with the history, source, properties, use, handling and more. The provenance data may be found from the link under the source name.
The periodic table contains NIST's critically-evaluated data on atomic properties of the elements. The provenance data that include data for atomic spectroscopy, X-ray and gamma ray, radiation dosimetry, nuclear physics, and condensed matter physics may be found from the link under the source name. Ref: https://www.nist.gov/pml/atomic-spectra-database
This section provides all form of data related to element Hydrogen.
The element property data was retrieved from publications.